Clinical cases of three pediatric patients who had bilateral spontaneous serous collections and septal hematoma located in the anterior sector of the nasal septum are described.
None had a history of trauma or infection, and all were immunocompetent; one was diagnosed with hemophilia type B.
They were treated with antibiotics and surgical drainage with good evolution.
Septal hematoma, Septal seroma, Surgical treatment, Pediatric
Septal hematoma may occur more frequently after nasal trauma or septal surgery for bleeding from the subseptal artery or its branches originating from the facial artery.
Septal abscess can occur when the hematoma becomes infected or directly from neighboring infections.
Its spontaneous appearance is very rare, there being few cases described in the literature, mostly under the clinical form of abscess.
We have not found in the literature the description of spontaneous septal seromas.
Early diagnosis and treatment are important to avoid cosmetic morbidity and potential intracranial complications that can occur if collections get infected.
Three clinical cases of spontaneous bilateral septal collections are described below.
Male patient, 11-years-old.
He consulted due to bilateral nasal obstruction of 5 days of onset.
He had no fever or pain and on inspection he had no facial erythema or cellulitis.
Had no history of other illnesses, trauma, recent surgeries, sinusitis, dental infections, or nasal boils.
Rhinoscopy revealed an anterior nasal tumor occupying both nostrils, covered by normal mucosa (Figure 1).
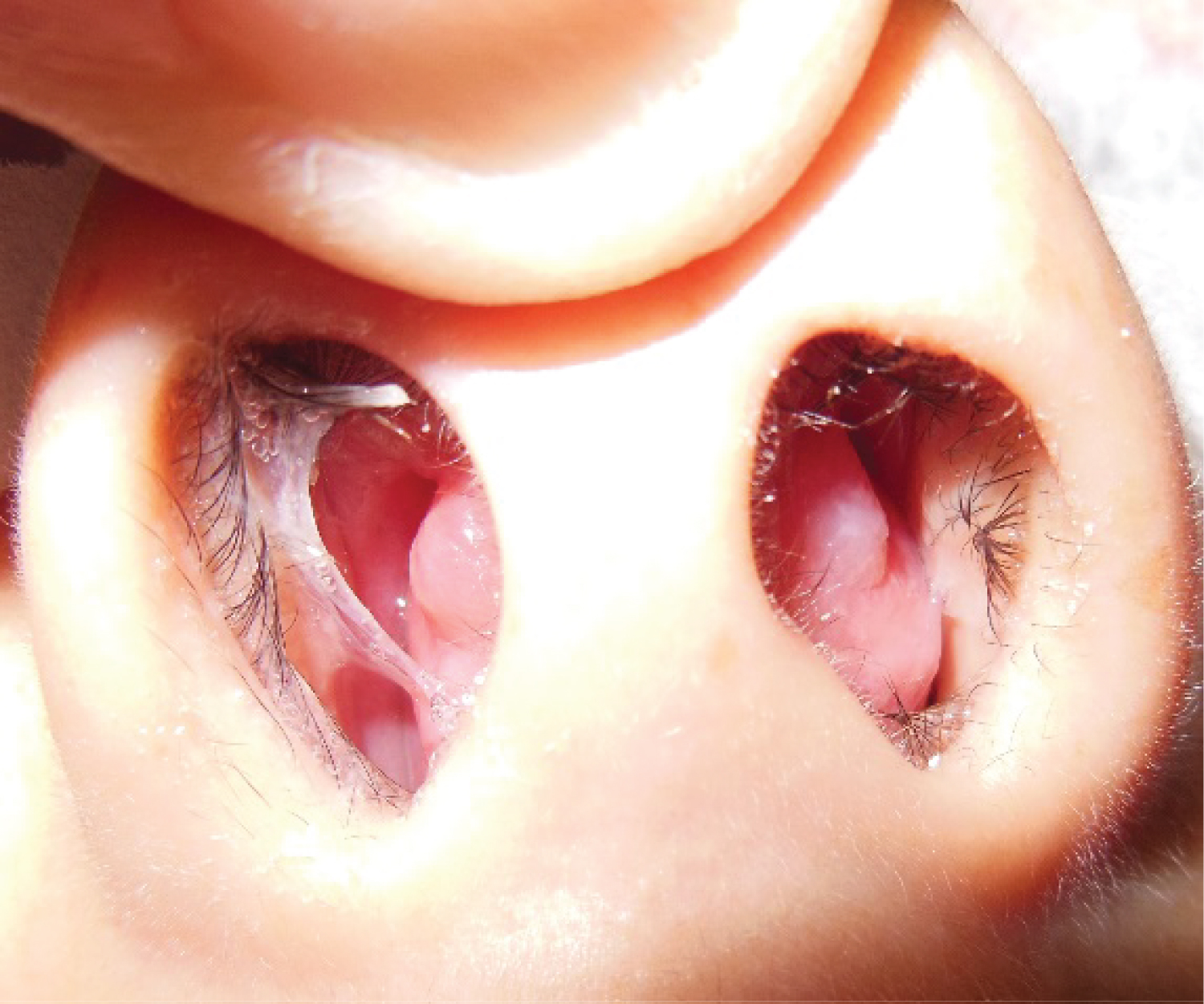 Figure 1: Bilateral anterior septal tumor.
View Figure 1
Figure 1: Bilateral anterior septal tumor.
View Figure 1
Nasal endoscopy was not performed because the endoscope could not be passed through the nostrils.
A computed tomography (CT) revealed a tumor occupying both nasal cavities, located in the anterior sector. Magnetic resonance (MRI) diagnosed a tumor hyperintense on T2, with possible septal origin (Figure 2).
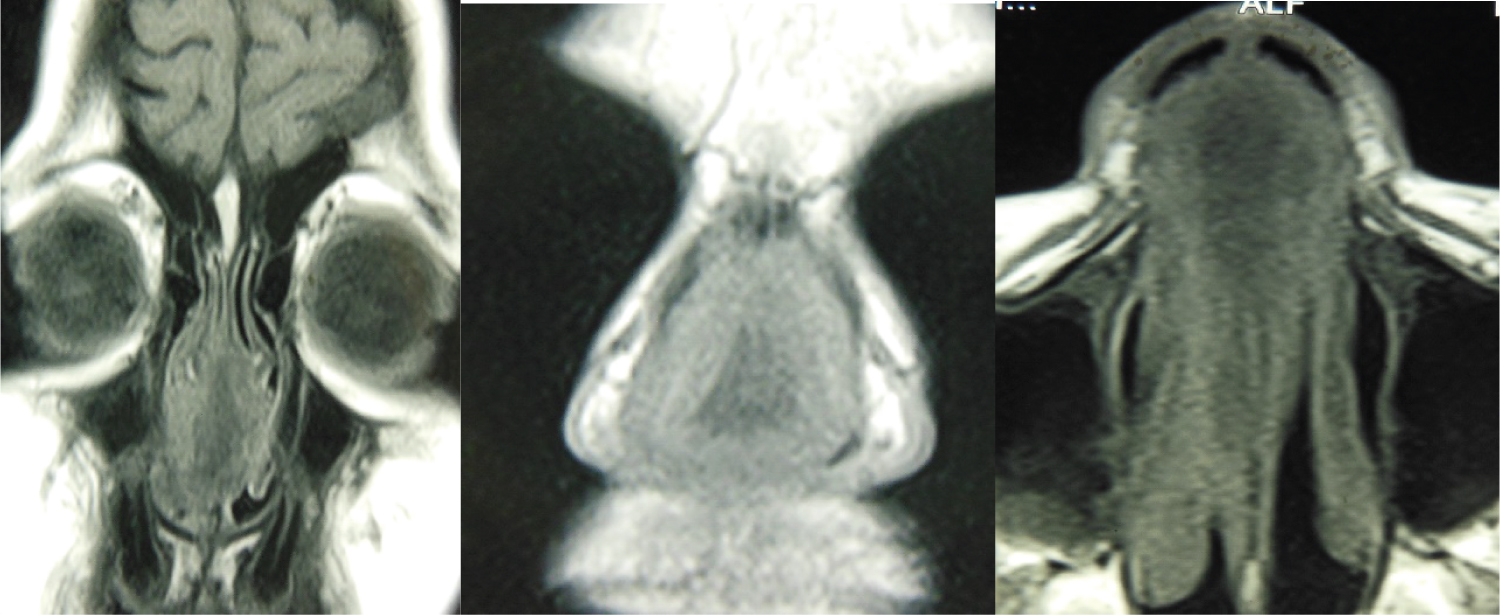 Figure 2: MRI: T2 hyperintense anterior septal tumor.
View Figure 2
Figure 2: MRI: T2 hyperintense anterior septal tumor.
View Figure 2
In the first visit, antibiotics (amoxicillin + clavulanic acid) were prescribed.
Under general anesthesia, through a bilateral hemitransfixing incision in the nasal septum, a mucoperichondrial flap was dissected, and a citrine fluid and a myxoid-looking material were drained and sent for direct bacteriological and mycological study and culture and for histopathological study.
It was found during the intervention that the septal cartilage was intact.
The incision was left open and a bilateral tamponade was placed, which was removed after 3 days.
Bacteriological and mycological studies were negative and the pathology report revealed chronic inflammatory tissue.
The blood count, erythrocyte sedimentation rate, and coagulogram were normal, and the dosage of Wegener-specific antibodies were negative.
The patient evolved well with resolution of the serous collection.
He performed controls for 4 months with no evidence of recurrence and no other disease was diagnosed.
Seven-year-old male patient.
He consulted due to progressive bilateral nasal obstruction of 7 days of onset.
He had no fever or pain.
Rhinoscopy revealed a bilateral tumor located in the anterior sector of the nostrils, covered by normal mucosa (Figure 3).
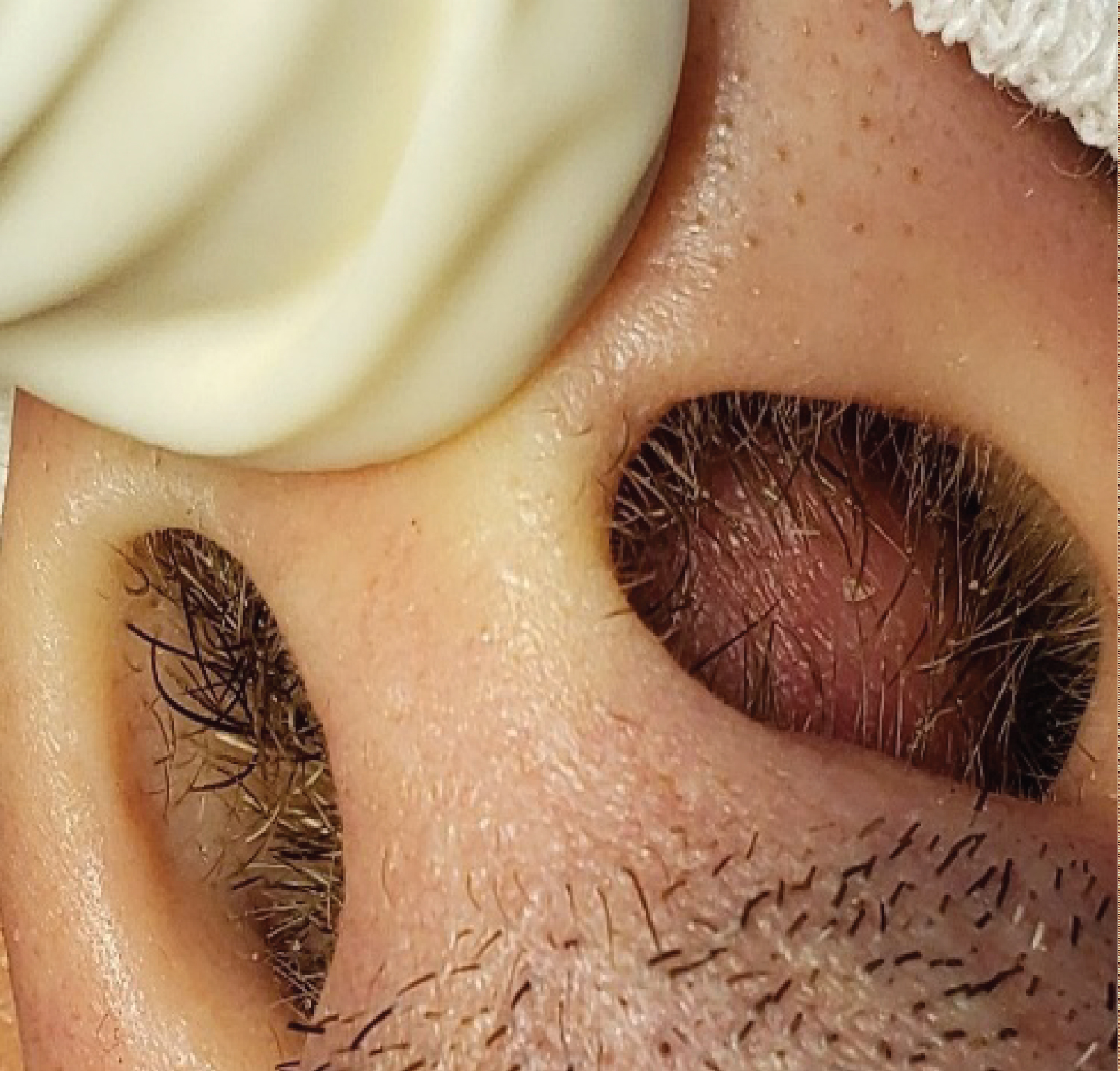 Figure 3: Anterior septal tumor.
View Figure 3
Figure 3: Anterior septal tumor.
View Figure 3
He had no history of trauma, nasal surgery, or other illnesses.
He was previously treated at another center with antibiotics (amoxicillin + clavulanic acid).
CT revealed a tumor located in the anterior sector of the nasal septum that occupied both nasal cavities, and the MRI diagnosed a hyperintense lesion on T2 (Figure 4).
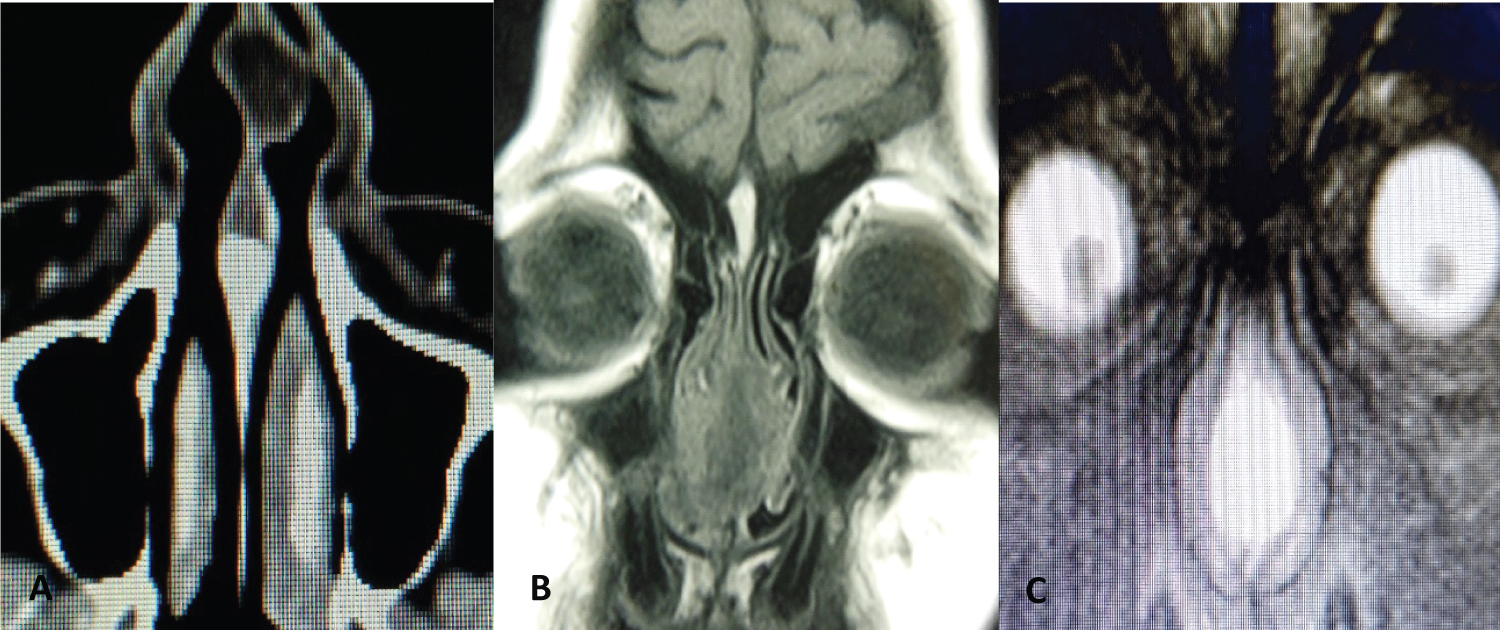 Figure 4: A) CT: Axial section showing an anterior septal tumor; B) MRI: Hyperintense anterior septal tumor on T2.
View Figure 4
Figure 4: A) CT: Axial section showing an anterior septal tumor; B) MRI: Hyperintense anterior septal tumor on T2.
View Figure 4
Fine needle aspiration drainage was performed in the office under topical nasal anesthesia.
Citrine liquid was obtained.
In the control at 48 hours, a bilateral collection was detected again in the anterior sector of the nasal septum.
A bilateral interseptocolumellar hemitransfixing incision was made under local anesthesia and a bilateral mucoperichondrial flap was dissected, draining citrine fluid. The septal cartilage was intact.
A bilateral anterior tamponade was placed and removed after three days.
Direct examination and cultures of the fluid obtained by puncture and during surgery were negative for fungi and bacteria. No other diseases were detected.
He had a good evolution and in the controls for 2 months he did not present recurrences.
A 12-month-old boy consulted for a tumor in the left nostril that had appeared 4 days ago.
He had no history of fever or trauma.
Anterior rhinoscopy revealed a bilateral bulging of the nasal septum, greater to the left, which was erythematous and with fluctuating content (Figure 5).
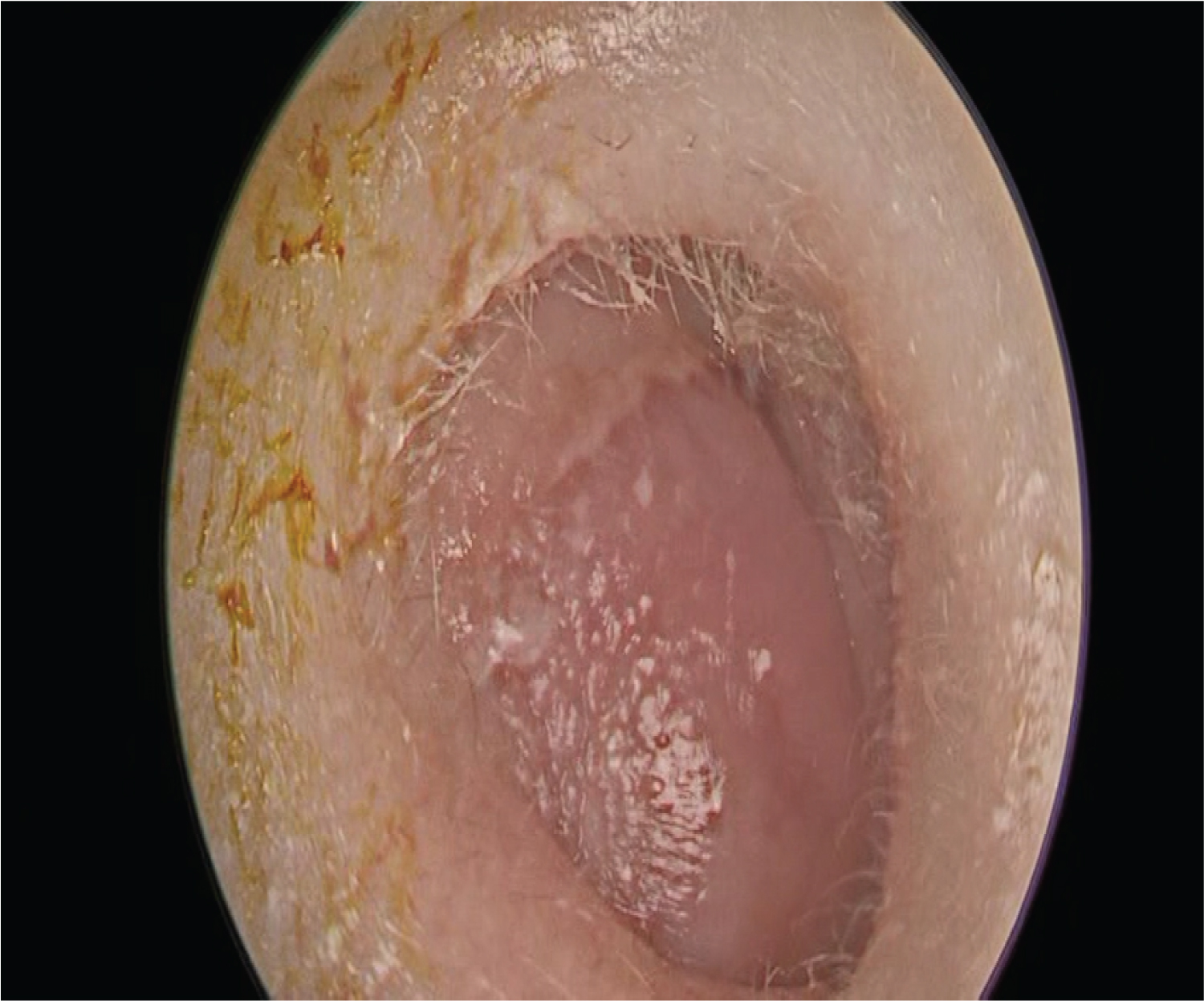 Figure 5: Anterior rhinoscopy: Septal tumor occupying the anterior sector of the nasal cavity.
View Figure 5
Figure 5: Anterior rhinoscopy: Septal tumor occupying the anterior sector of the nasal cavity.
View Figure 5
A puncture was performed under topical nasal anesthesia in office, draining blood content and then an incision was made with a scalpel in the lower sector of the nasal septum coinciding with the point of greatest protrusion. Spongostan was placed in the nasal cavities and he was medicated with antibiotics. He had partial improvement, for which a CT was requested. The report was: expansive lesion with epicenter in the left nostril in close contact with the nasal septum, the lesion was heterogeneous, predominantly hyperdense, with no evidence of contrast enhancement. A solution of continuity was observed in the left cribriform plate (Figure 6).
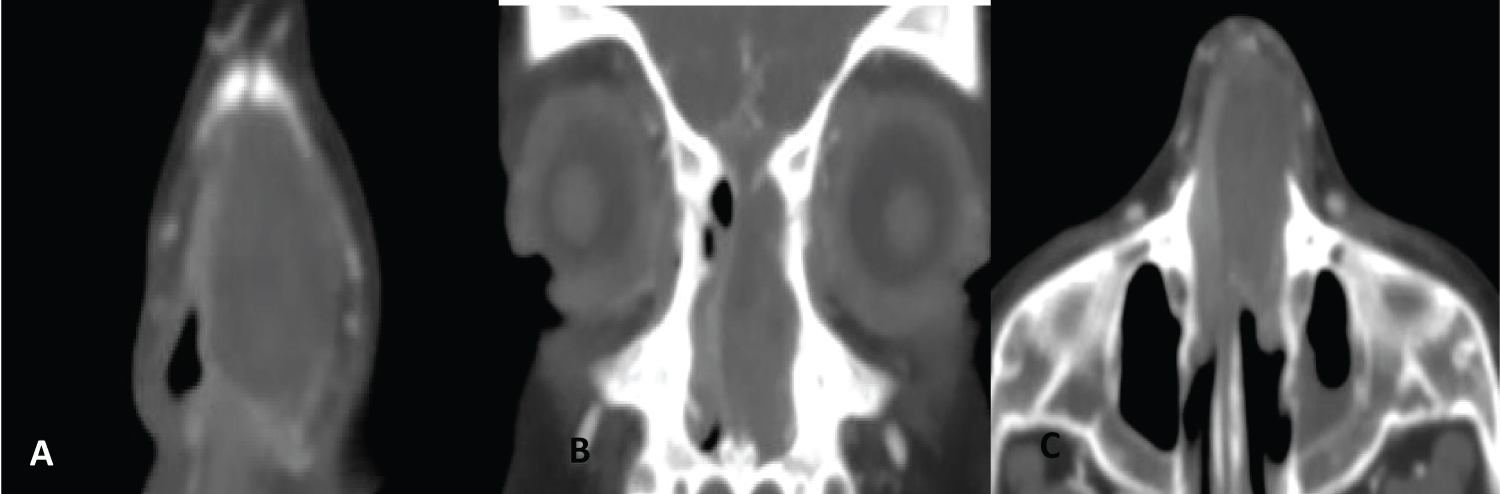 Figure 6: A) Coronal CT slices: Heterogeneous and hyperdense expansive lesion in the anterior nasal septum; B) C-axial CT slice: Lesion occupying the anterior and middle third of the nasal septum.
View Figure 6
Figure 6: A) Coronal CT slices: Heterogeneous and hyperdense expansive lesion in the anterior nasal septum; B) C-axial CT slice: Lesion occupying the anterior and middle third of the nasal septum.
View Figure 6
MRI was requested, which diagnosed a hypointense lesion at the level of the nasal septum on T2 and slightly hyperintense on T1, no solution of continuity was observed at the anterior skull base. The lesion was compatible with a septal hematoma (Figure 7).
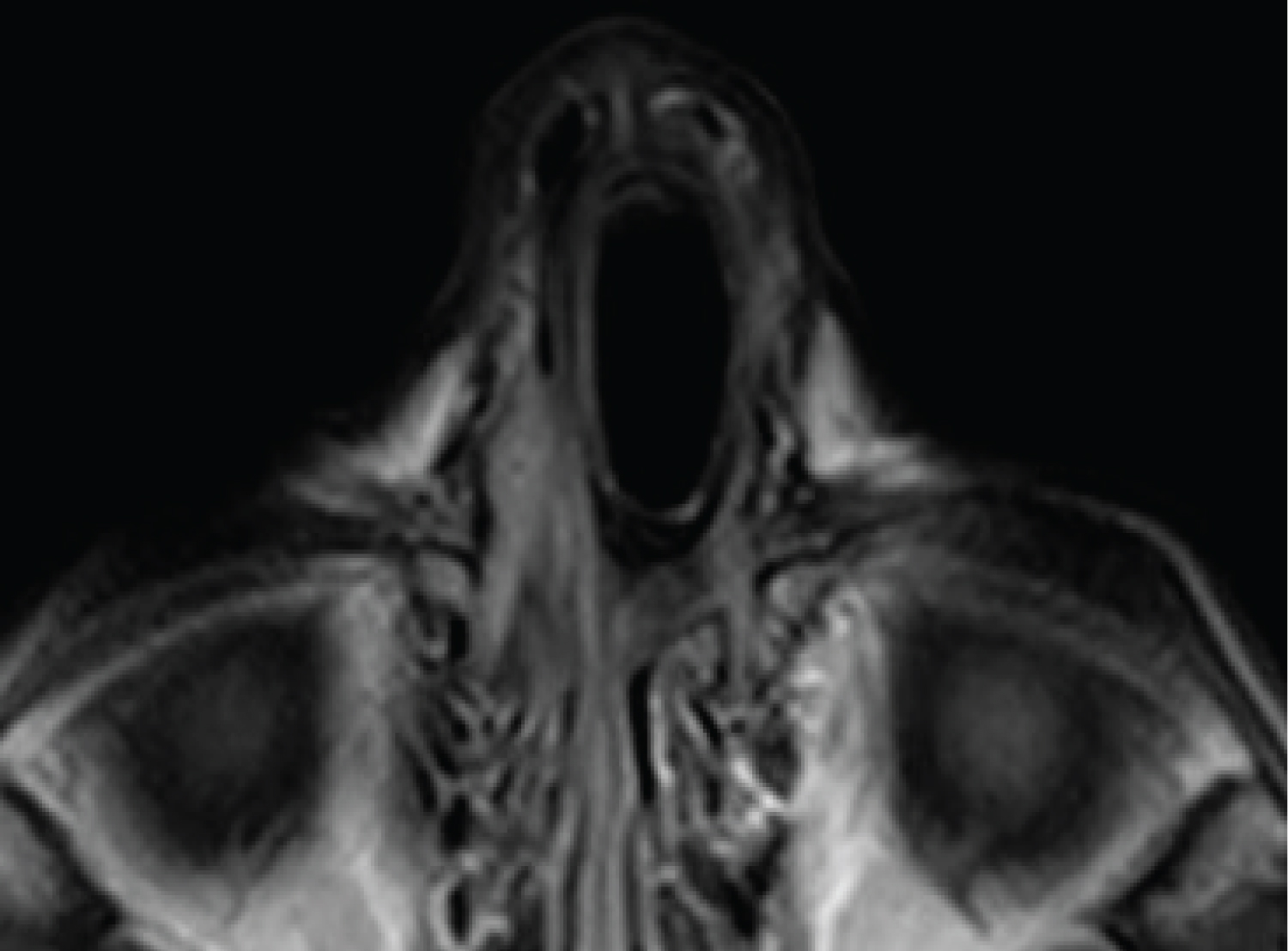 Figure 7: MRI: hypointense septal lesion on T2 and slightly hyperintense on T1.
View Figure 7
Figure 7: MRI: hypointense septal lesion on T2 and slightly hyperintense on T1.
View Figure 7
The controls in the office showed a progression of the collection, for which a pre-surgical evaluation was requested to drain the hematoma under general anesthesia in the operating room.
The coagulogram showed an activated partial thromboplastin time of 61 and factor IX deficiency (2%), for which hematology consultation was performed.
The diagnosis was moderate hemophilia B.
Pre-surgical prophylaxis was performed with intravenous recombinant factor IX 500 U/day.
Drainage of the septal hematoma was performed under general anesthesia 15 days after the initial consultation through a left anterior septal hemitransfixing incision. The mucoperichondrium was dissected with a dissector-aspirator and clots were extracted, confirming the necrosis of a sector of the septal cartilage which was resected. Transfixing sutures were given in the nasal septum and spongostan was placed in the nostrils (Figure 8).
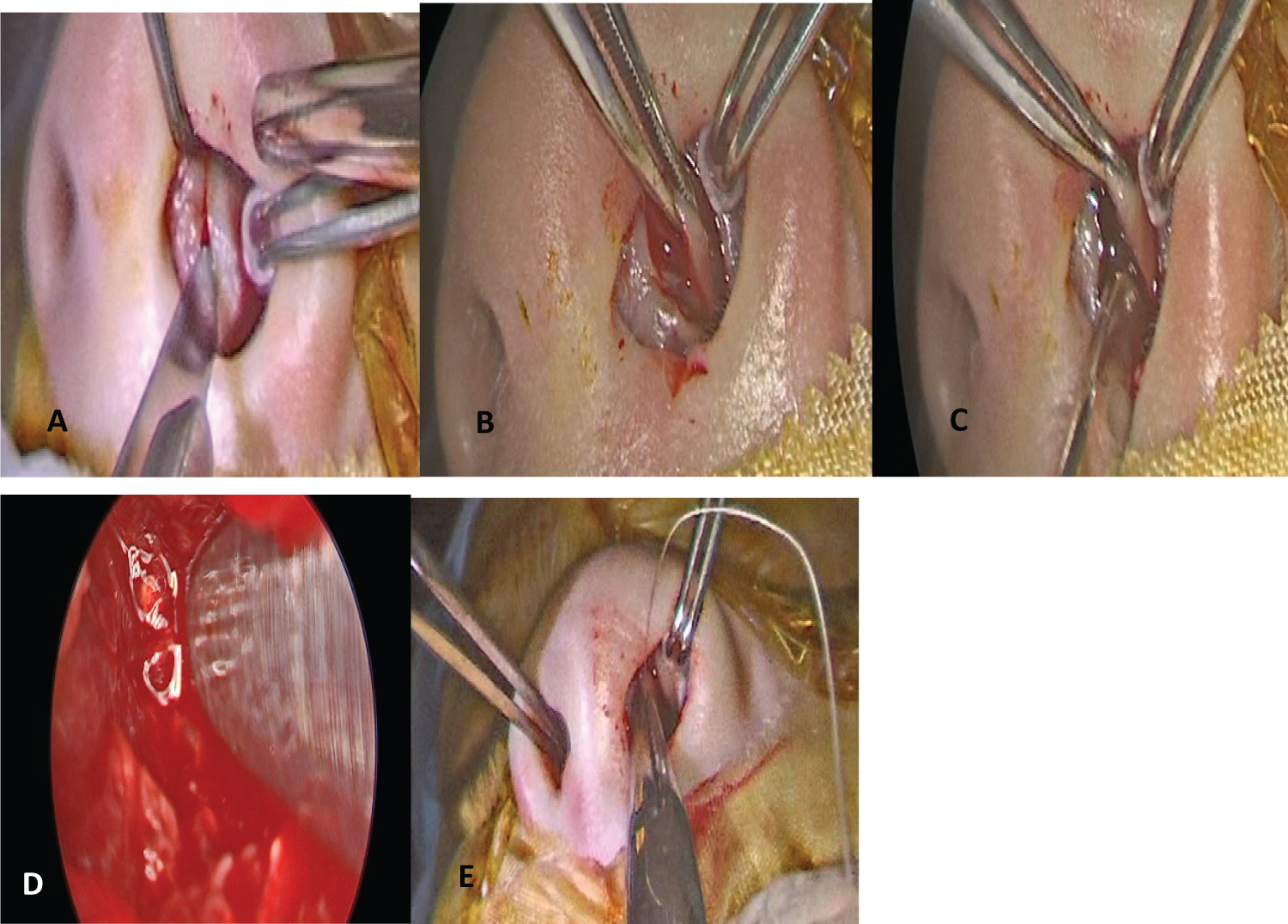 Figure 8: Surgical drainage of bilateral septal hematoma. A) Left septal hemitransfixing incision; B,C and D) Subperichondrial dissection and removal of bilateral hematoma and necrotic cartilage debris; E) Septal transfixing sutures.
View Figure 8
Figure 8: Surgical drainage of bilateral septal hematoma. A) Left septal hemitransfixing incision; B,C and D) Subperichondrial dissection and removal of bilateral hematoma and necrotic cartilage debris; E) Septal transfixing sutures.
View Figure 8
He did well and continued with factor IX infusion for 10 days after surgery.
The septal cartilage receives its blood supply from a network of blood vessels that originate from the overlying mucosa and penetrate the mucoperichondrium through vascular channels located at the chondromaxillary junction. Disruption of the mucoperichondrium on both sides alters its blood supply and produces cartilage necrosis.
Septal hematoma is caused by the rupture of small blood vessels that supply the nasal septum, more frequently after trauma. The blood collection separates the mucoperichondrium from the cartilage, and the cartilage undergoes ischemic necrosis.
This medium is prone to colonization by bacteria and subsequent formation of a septal abscess [1]. Other etiologies of abscesses and hematomas may include sinusitis [2], boils, dental infections, facial cellulitis, septal surgeries, and poor immune status.
The incidence of septal abscesses is unknown; retrospective studies describe 16 cases in a 16-year period in one hospital and 43 cases in an 8-year period in another center [3].
In another study, 46 patients with septal hematomas were reported; in most cases (65.6%) the cause was unknown [4].
Nwosu, et al. describe 53 patients with septal hematomas/abscesses in a 10-year period, the peak incidence was between 10-18 years and then between 1-9 years.
The hematoma/abscess was bilateral in 46 cases. Some type of trauma was identified as an etiological factor in 35 (66.4%) and in 14 (26.4%) the etiology was not identified.
Cases of hematomas were diagnosed within 10 days of symptom onset, and abscesses after two weeks [5].
In our cases, the patients did not have pain or fever or other signs that could indicate infection.
The complete blood count in two patients was normal and the other had a prolonged partial thromboplastin time.
The most frequently isolated bacteria in the drained material are: staphylococcus aureus, streptococcus pneumoniae, B-hemolytic streptococcus, and haemophilus influenzae.
In the patients described, material was sent for culture and no germs were isolated, possibly due to the previous antibiotic treatment that they received.
It is not always necessary in patients with hematomas or septal abscesses to request an image since the diagnosis can be established clinically or by the material obtained by puncture.
In some cases with unclear etiologies, suspicion of Wegener's, sarcoma, lymphomas and especially in spontaneous cases, in immunosuppressed patients or when medical/surgical treatment fails, it may be necessary to request CT and/or MRI [6].
In the cases described, we requested images since in two the etiology could not be established and in the other the cause of the hematoma could not be determined initially and the CT diagnosed a possible disruption of the skull base.
We did not find in the literature indexed in Pub-Med using the keywords: Septal seroma, nasoseptal seroma, spontaneous septal seroma studies that described this form of spontaneous septal collection.
Possibly some minor trauma not perceived by the patients or their parents has produced the serous collection, and the rapid prescription of antibiotics has prevented infection and the production of an abscess.
If the treatment is delayed, the collection can evolve towards the formation of an abscess and necrosis or perforations of the septal cartilage can occur with aesthetic and/or functional alterations. The infection can spread and cause serious complications such as orbital cellulitis, meningitis or thrombophlebitis of the cavernous sinus. Intracranial extension can occur through the superficial venous system or the periosteum. The veins of the anterior septum communicate with the veins of the upper lip and palate and through them with the valveless angular and ophthalmic venous system.
These intracranial complications are more frequent in immunocompromised patients [7,8].
In our patients, the delay in performing surgical drainage from the first consultation was an average of 16 days.
If an abscess is suspected, it is advisable to hospitalize the patient and prescribe parenteral antibiotics.
The definitive treatment of septal hematomas/abscesses is surgical drainage, in children it is better that the procedure be done under general anesthesia.
The surgery consists of making a bilateral hemitransfixing incision, dissecting the mucoperichondrium of the cartilage and draining the collection if it is bilateral. If there is necrosis of the cartilage, drainage and resection of the necrotic tissue can be done through of a unilateral incision.
Transfixing sutures between the mucoperichondrium and the cartilage can be given or the incisions can be left open and a bilateral tamponade can be placed to put pressure on the mucoperichondrial flaps and prevent recurrences. Most leave the tamponage between 48 to 72 hours [9].
We prefer this last procedure in older children, in the youngest of 12 months we gave transseptal transfixing sutures with resorbable material.
If there is an associated infection such as boils or sinusitis, it should be treated at the same surgical time as the collection drainage.
Some prefer to reconstruct the cartilaginous nasal septum if it is compromised by infection between 8 and 12 weeks after drainage, others prefer to perform acute reconstruction, at the same surgical time as abscess drainage [10].
Reconstructive techniques can use residual posterior septal cartilage or the bony septum (exchange technique), can remodel the cartilaginous septum with small fragments of residual cartilage fixed with fibrin glue (mosaicoplasty), or using preserved costal cartilage if no septal cartilage is available (homograft) [11].
Some studies reported the spontaneous regeneration of the cartilaginous septum from infected and necrotic cartilage remains [12].
In a study carried out on 17 rabbits, they performed a submucosal resection of the septal cartilage.
At 7 months they performed a histological examination of the septum and found new cartilage between the mucoperichondrial flaps. They concluded that when the overlying mucoperichondrium is healthy, septal cartilage can regenerate [13].
Early diagnosis of septal collections is important to avoid aesthetic alterations and intracranial complications that can occur if an abscess forms.
Antibiotic therapy and surgical drainage are essential to resolve the pathology, while acute or delayed reconstructions of the nasal septum are controversial.
We do not declare conflicts of interest.