Amygdala, Hippocampus, Cytoarchitecture
The human amygdala and hippocampus are key structures for emotion and memory processing. They are involved in various neurological and psychiatric disorders. Since each of both brain regions is structurally and functionally extremely segregated [1], their numerous subdivisions are implicated to different degrees in specific pathological processes, e.g. epilepsy [2,3].
Although modern functional imaging techniques can assign dysfunctions to the whole amygdala or hippocampus after specific impairments [4], the disorder- or lesion-specific involvement of the clearly defineable, and functionally as well as structurally diverse cytoarchitectonic subdivisions remains to be elucidated. Therefore, cytoarchitectonic probability maps are a prerequisite for an anatomically sufficiently precise localization of functional imaging data [1]. This was realized by registration of probability maps of the subdivisions of the amygdala and hippocampus to a stereotaxic reference space, which is also used for the localization of functional imaging data. E.g., a prevailing contribution of the basolateral amygdala was revealed by using this combined approach in impaired acquisition of conditioned fear in patients with Urbach-Wiethe disease [5].
Here we present further advanced cytoarchitectonic parcellations of the amygdala (Figure 1) and the hippocampal formation (Figure 2). The cytoarchitectonic identification of the subdivisions of the hippocampus and amygdala in histological sections is the basis of the fine-grained probability maps, as well as of volumetric measurements.
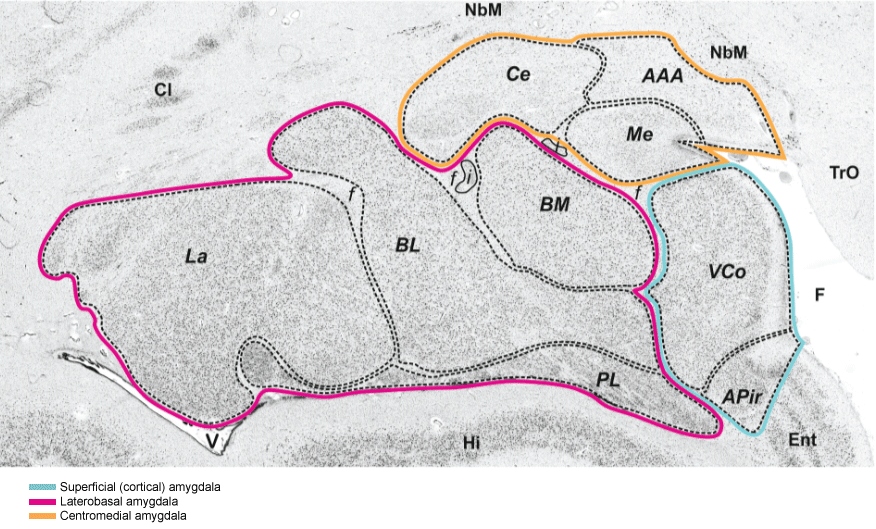
Figure 1: Cytoarchitecture of the amygdala and neighboring cortical and subcortical structures in a coronal section of a human postmortem brain. The centromedial nucleus is labelled by an orange line and the basolateral complex by a red line. The VCo belongs to the superficially located part of the amygdala. AAA anterior amygdaloid area, APir amygdalopiriform transition area, BL basolateral nucleus, BM basomedial nucleus, BV basoventral nucleus, Ce central nucleus, f fiber bundles, i intercalated islands, La lateral nucleus, Me medial nucleus, PL paralaminar nucleus, VCo (ventral) cortical nucleus. Neighbouring structures: Cl Claustrum, Ent entorhinal cortex, F endorhinal sulcus, Hi hippocampus, NbM Nucleus basalis of Meynert, TrO Tractus opticus, V lateral ventricle.
Source: Figure 1 (minimally altered) "Springer Anat Embryol, Cytoarchitectonic mapping of the human amygdala, hippocampal region and entorhinal cortex: Inter subject variability and probability maps, 210, 2005, 343-352, Amunts K, Kedo O, Kindler M, Pieperhoff P, Mohlberg H, Shah NJ, Habel U, Schneider F, Zilles K ©Springer Verlag, "With permission of Springer".
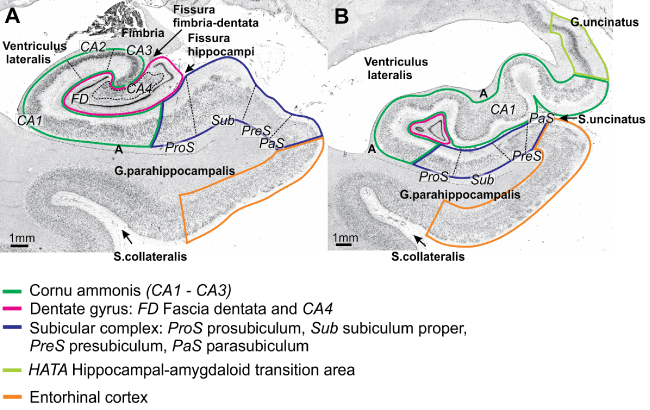
Figure 2: Cytoarchitecture of the mesial temporal lobe at the level of the body of the hippocampus (a), and its head (b). The hippocampus proper with its subdivisions Cornu ammonis, dentate gyrus, the HATA, the subicular complex and the entorhinal cortex are labelled by different colours. Note that cytoarchitectonic borders of the different subdivisions do not coincide with sulci in most cases.
Source: Figure 2 (minimally altered) "Springer Anat Embryol, Cytoarchitectonic mapping of the human amygdala, hippocampal region and entorhinal cortex: Intersubject variability and probability maps, 210, 2005, 343-352, Amunts K, Kedo O, Kindler M, Pieperhoff P, Mohlberg H, Shah NJ, Habel U, Schneider F, Zilles K ©Springer Verlag, "With permission of Springer".