Lemierre's syndrome is a rare disorder of young adults, usually caused by the anaerobic bacterium, Fusobacterium necrophorum and occasionally by other Fusobacterium species (F. nucleatum, F. mortiferum and F. varium etc). The condition is characterized by an oropharyngeal infection leading to inflammation of the internal jugular vein (IJ) and that can lead to septic thrombophlebitis with positive blood cultures, clinical or radiographic evidence of internal jugular vein thrombosis, and at least one metastatic focus. The incidence of Lemierre's syndrome is not known with some reporting one in a million. In the pre-antibiotic era, Lemierre's syndrome followed a fulminant, often fatal course. During the 1960s and 1970s, the syndrome was rarely reported when penicillin was commonly prescribed to treat oropharyngeal infections. However, the appearance of antibiotic-resistant organisms are leading to a rise of the condition.
We reviewed the literature and present a case report of a 30-year-old patient that presented with a peritonsillar abscess and rapidly progressed to develop proptosis, lateral rectus palsy and then devastating ischemic stroke and subsequent brain death.
Our patient was a 30 year old female with no significant past medical history. She presented to the ED with complaints of fever, chills and progressively worsening sore throat for 4-5 days, to the point she could not eat or drink due to the pain and dyspnea on lateral decubitus but not supine. The day of admission she developed intermittent dizziness and blurry vision. She was found to be tachycardic to 190 bpm, febrile with temperature of 102.1 Fahrenheit, blood pressure of 117/91 and respiratory rate of 18. She was able to communicate, but with a hoarse voice. She had left facial edema, left submandibular edema, left eye proptosis, and lateral rectus palsy. In addition to her difficulty communicating, she also had obvious drooling. Mouth opening was limited due to edema. The rest of her examination was unremarkable. These symptoms were most suggestive of peritonsillar abscess. Her family history was only positive for rheumatoid arthritis in her mother. She had never smoked, drank about 12 oz of alcohol per week and denied use illicit drugs. She had no allergies and was taking no medications or supplements. She had no obvious exposures or recent travel history.
Pertinent laboratories showed a potassium of 3.1 mmol/L, chloride 93 mmol/L, total CO2 26 mmol/L, ionized calcium 4.3 mg/dL, BUN 31 mg/dL, creatinine 1.0 mg/dL, white blood cell count of 25.91 with 88% neutrophils, hemoglobin 17.3 g/dL, hematocrit 45.5%, platelet count of 20,000, total bilirubin was 1.3 mg/dL direct bilirubin 0.98 mg/dL, albumin 2.1 g/dL, ASL 114 U/L with normal ALT and total protein, lactic acid 5.7 mmol/L. Her arterial pH was 7.41 with a PaO2 of 61 mmHg and PCO2 35 mmHg.
Her CT scan of the neck (Figure 1 and Figure 2) revealed thrombosis of the left internal jugular vein with focal fluid collection in the retropharyngeal soft tissues extending from the level of the posterior nasopharynx into the level of C6 vertebral body and thrombosis of the left sphenoid sinus. The distal 3 cm of the left IJ vein was also patent.
The initial CT scan of the head did not reveal pathology; however, the initial CT of the chest (Figure 3) did reveal multiple pulmonary masses highly concerning for septic emboli.
She was started on intravenous vancomycin, piperacillin/tazobactam, metronidazole and underwent intubation in the operating room by anesthesiology, and subsequent incision and drainage of her peritonsillar and retropharyngeal space by the oral-maxillofacial surgery team but no purulence was found from the tonsils and posterior pharyngeal wall aspiration. She remained intubated post-operatively due to her airway edema.
A CT venogram was obtained revealing thrombosis of the entire course of the left internal jugular vein with probable thrombosis of the left sigmoid dural venous sinus. There was no contrast opacification of either cavernous sinus, and a small amount of thrombus in the superior aspect of the left internal jugular vein was consistent with thrombus extension from the right inferior petrosal sinus. There were no other dural venous sinus thromboses; the superior sagittal sinus and the straight sinus preferentially drain into the right transverse sinus and, subsequently, into a large right internal jugular vein.
She remained thrombocytopenic in the 20,000 range with a total of 8 unit's platelets given during admission. On the fourth day, she became unresponsive to painful stimuli and had a dilated unresponsive left pupil. This was the reason for which a brain MRI (Figure 4) was obtained, which ultimately revealed multiple acute infarctions in both cerebral hemispheres, most of which were in the expected vascular border zone between the anterior and middle cerebral arteries. There were also acute infarctions in the left occipital lobe (expected vascular distribution of the left posterior cerebral artery) and in the left cerebellar hemisphere. None of these infarctions had an appearance suggestive of venous occlusion as its origin.
An attempt to perform embolectomy was considered but felt that it would produce further septic emboli and probably worsen her overall clinical status; in addition, because of the size of the vein there were concerns with the ability to keep it patent. In addition, because the left internal jugular vein did not contribute significantly to her cerebral venous outflow, and there was no evidence that thrombosis of this vein or of the left sigmoid sinus were associated with her worsening mental status, it was decided not to proceed with the procedure.
Since no obvious explanation for the source of her arterial-appearing infarctions was found, the concern was that it was due to the cavernous sinus septic thrombosis leading to vasospasm of the adjacent internal carotid arteries (Figure 5). There was no evidence of septic emboli to explain the symmetric border-zone infarctions.
She was briefly placed on a heparin drip but given her worsening thrombocytopenia and increased risk for hemorrhage it was later discontinued. On day five, antibiotics were changed to vancomycin, meropenem, and acyclovir for broader coverage adding meningitis to the differential due to her worsening clinical status. On day six, her right pupil became dilated and unresponsive. She was taken for emergent CT head (Figure 6) which showed very large left cerebral infarct with probable underlying punctate hemorrhages and mass effect. Shortly after, she no longer had brainstem reflexes or spontaneous breaths; an apnea test was performed confirming the diagnosis of brain death.
Lemierre's syndrome is a potentially fatal disease associated with oropharyngeal infections. The most common etiology is from Fusobacterium species [1-3]. The classic triad consists of oropharyngeal infections with jugular vein thrombosis and septic emboli (most commonly to the lungs) with the most common initial presenting symptom being sore throat [1-3].
Lemierre's syndrome was initially thought to be a rare disease, however many articles suggest an increasing occurrence (possibly due to antibacterial resistance) [2-6].
Fusobacterium is part of the normal flora of the oral cavity, female genital tract and the gastrointestinal tract. Fusobacterium necrophorum is the most virulent strain and most common pathogen isolated in Lemierre's syndrome [1,2,4]. It is an anaerobic, nonmotile, gram negative bacillus (Figure 7). It is slow growing and can often be difficult to isolate or identify. Fusobacterium strains have a lipopolysaccharide endotoxin with strong biologic activity and produces leucocidin and haemolysin to augment virulent properties. Due to these toxins, Fusobacterium is able to affect previously healthy individuals. Some suggest that Fusobacterium also causes aggregation of platelets, further increasing its virulence [1,2,4]. According to one article, the release of serotonin from platelets accompanied aggregation when fusobacterium was added to a platelet solution. This showed a typical aggregation-degranulation reaction. No avirulent strains of fusobacterium could aggregate whereas virulent A type strains could. These authors suggested platelet aggregation was associated with virulence [7].
Others believe that the hypercoagulable state is related to both endothelial damage from microorganism invasion and a pro-inflammatory immune response to systemic infection (particularly in patients that have hypercoagulable syndromes). Evidence that this is the case comes from studies that show F. necrophorum bacterium's ability to activate the contact system, which is a crucial converging pathway between inflammation and coagulation [8].
Infection of the lateral pharyngeal space may occur from a tonsillar or peritonsillar primary infection and also pharyngitis, parotitis, or otitis media [1-3].
This is due to the anatomical characteristics of the area. The lateral pharyngeal space is divided into the anterior and posterior compartment. Within the posterior compartment is the carotid sheath enclosing the carotid artery and internal jugular vein (along with the vagus nerve). When the posterior compartment is infected, it can extend to cause thrombophlebitis of the jugular vein directly or can result from extension of peritonsillar vein thrombophlebitis [4].
The time of onset from the initial oropharyngeal infection to the development of Lemierre's syndrome is typically less than 1 week [1,3,5]. The development of neck pain and swelling can be due to thrombophlebitis of the internal jugular vein [1]. The most common site of metastasis is the lungs (which can also be associated with pulmonary infiltrates, effusion, empyema, cavitary lesions or acute respiratory distress syndrome (ARDS) [1-5]. Many cases suggest that the next most common sites of metastases are the joints or osteomyelitis [1,3-5]. Other complications include liver abscess formation, soft tissue abscesses and cutaneous pustules and septic shock.
CNS complications are unusual, and most of them include brain abscesses, meningitis, and cavernous sinus thrombosis. Twenty-two cases of Lemierre's syndrome from 1980 to 2010 were identified with CNS complications, and of these only six reported cerebral infarcts [9]. Within this subset, the two that died were an 18 year old male [10], and a 20 year old female [11]. The proposed mechanism suggested for how an infectious focus may lead to cerebral embolic phenomenon is the presence of a bony defect near the infectious focus as a point of entry [12].
Ophthalmic involvement in Lemierre's syndrome is rare with few reports of proptosis [13], endogenous endophthalmitis [14], vitreous hemorrhage [15], and orbitopathy [16,17]. There are reports of isolated fourth and sixth nerve palsies resulting from Lemierre's syndrome [18,19]. Fusobacterium necrophorum is usually sensitive to penicillin, clindamycin and metronidazole. One review suggested a mortality rate of 5-10%, but with much higher rates of morbidity [20]. Extended antibiotic time is suggested due to the vascular nature of the disease [1,2].
The role of anticoagulation remains controversial. Some cases suggest expedited resolution of thrombophlebitis with anticoagulation use, therefore increasing recovery time of septicemia [21]. Another article suggests that the only clear indications for anticoagulant use would be in patients with confirmed sigmoid sinus thrombosis with possible retrograde propagation to the cavernous sinus and inferior vena cava [1,22].
A review by Kuppalli et al. 2012 demonstrated that the characteristics of the very rapid and progressive onset of complications as described in this case, despite empirical antibiotic therapy, are poorly understood and thus may warrant more aggressive therapeutic modalities, including the use of antiplatelet agents, anticoagulants, and even steroids.
Microbial identification can take several days due to the slow growing nature. One case report describes the culture taking 9 days to grow while another took 2 weeks for isolation [5]. Unfortunately, no organism was ever isolated in our case.
Early suspicion and recognition is crucial for recovery. Delay in treatment increases morbidity and mortality [1-5] and therefore no delay in therapy should occur while awaiting microbiological identification. Furthermore failure of conventional therapy and rapid deterioration in patients with oropharyngeal infections should prompt the concern for this potentially lethal condition.
Future studies and evaluation of treatment modalities in the management of Lemierre's syndrome need to be obtained, as more cases are reported (currently more notable in pediatric literature [23,24]) with severe CNS complications including ischemic infarcts and subsequent death.
All authors listed have contributed sufficiently to the project to be included as authors, and all those who are qualified to be authors are listed in the author byline. To the best of our knowledge, no conflict of interest, financial or other, exists.

Figure 1: Initial computed tomography scan, soft tissue neck - 7 × 5 mm small peritonsillar abscess.

Figure 2: Initial computed tomography soft tissue neck, partial thrombosis of the left internal jugular vein.
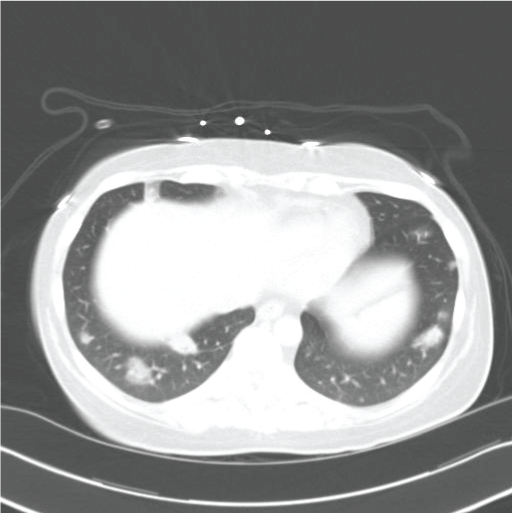
Figure 3: Initial computed tomography scan thorax, lung windows: multiple septic emboli.
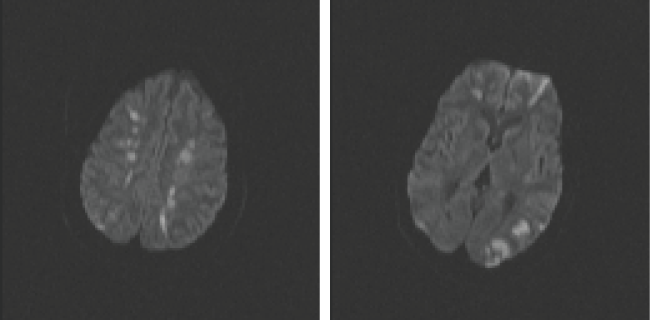
Figure 4: MRI imaging with multiple acute infarctions in both cerebral hemispheres.
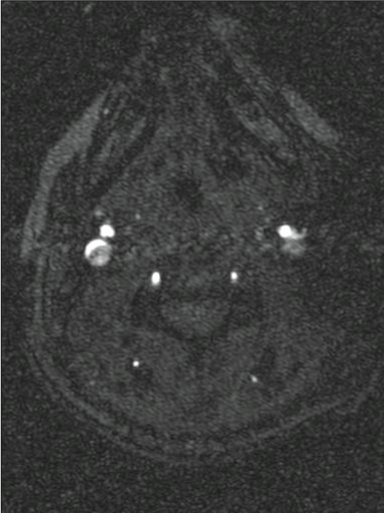
Figure 5: Magnetic resonance arteriogram of the neck showing an abrupt cutoff of the left ICA within a few centimeters of its takeoff with no discernible flow noted within the left cervical, petrous and cavernous internal carotid artery consistent with an embolic occlusion.
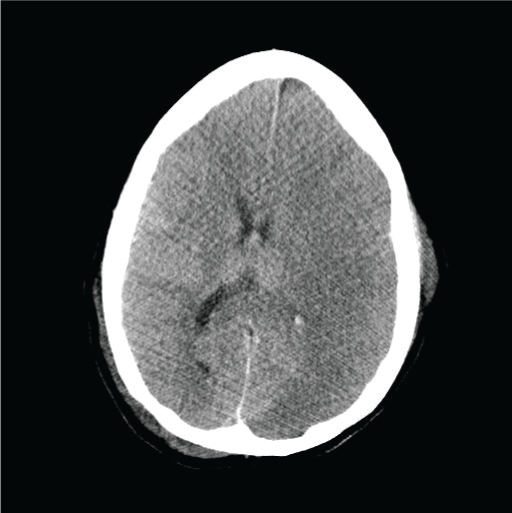
Figure 6: CT head showing a large left cerebral infarct with underlying punctate hemorrhages with mass effect on the ventricular system.

Figure 7: Fusobacterium necrophorum. (Courtesy of Dr. V.R. Dowell, Jr., CDC).