Herpes zoster (HZ, shingles) is caused by reactivated varicella zoster virus, that travels along the peripheral branches from its dormant location in neurons of spinal and cranial sensory ganglia. HZ is well known for its skin vesicular eruptions and severe sensory neuropathy. Over the years, reports have described the occurrence of segmental paresis of skeletal muscles and the involvement of the large and small intestine, clinically resembling intestinal obstruction.
The spread of the virus to the motor and autonomic nervous systems is discussed here based on the neuroanatomical pathways and with reference to the unfamiliar manifestations observed in a recently hospitalised HZ case.
A 66-year-old male of Egyptian descent, initially developed muscle aches in the right loin, altered touch sensation in the overlying skin and bouts of stabbing pain. These were followed on day two by the appearance of an abdominal wall swelling, 10 × 12 cm, in the right lumbar region. On day four, he complained of nausea, bloating, abdominal distension, and constipation. Examination revealed reduced intestinal sounds, and CT scans showed distended intestine with gas and fluid levels. On day five, vesicular skin rash characteristic of HZ appeared along dermatomes of the right spinal nerves T10 to L1.
Surgeons and medical practitioners should be made aware of these unfamiliar presentations of HZ, especially as these may precede the archetypal skin rash, to evade unnecessary surgical intervention.
Herpes zoster, Dermatomes, Dorsal root ganglion, Abdominal pseudohernia, Intestinal pseudo-obstruction, Blood-nerve barrier, Pia mater, Autonomic nervous system
BNB: Blood-Nerve Barrier; DRG: Dorsal Root Ganglion/Ganglia; HZ: Herpes Zoster; Herpes Zoster Virus (HZV); SGC: Satellite Ganglion Cells; VZV: Varicella Zoster Virus
Susceptible individuals who have contracted chickenpox infection may develop herpes zoster (HZ, shingles) later in life. The agent responsible for both illnesses is the varicella zoster virus (VZV), a dermotropic and neurotropic double-strand DNA virus [1,2]. After recovering from chickenpox, the virus may remain dormant in sensory ganglia of the cranial nerves and spinal nerves (dorsal root ganglia, DRG) [2]. A human post-mortem study using polymerase chain reaction (PCR) detected latent VZV DNA in 12 DRG out of 14 cases, and in 10 trigeminal ganglia out of 11 cases [3]. Reactivation of the dormant virus may occur under stress conditions or weakened immunity [2]. The virus is thought to travel along the sensory nerves causing skin rash in the affected dermatomes, severe pain, and may cause postherpetic neuralgia. HZ is well-known for these sensory manifestations. However, the virus may also affect the motor and the autonomic nervous systems, leading to unfamiliar manifestations including skeletal muscles paresis [4], and temporary intestinal paralysis [5]. The neuroanatomical basis for the spread of the virus to motor neurons and to the autonomic and enteric nervous systems are discussed here with reference to a recent case, which presented with motor paresis and visceral neuropathy prior to the development of the skin rash.
A 66-year-old male, presented with deep muscle aches in the right loin and hypo-aesthesia in the overlying skin. This was followed within hours by an altered sensation to touch. Touching the skin in the right lumbar region generated an unpleasant sensation. The patient complained of bouts of lancinating sharp pain in the right side of the trunk, alternating with burning discomfort. HZ was suspected as the likely diagnosis and the skin areas involved were checked regularly anticipating the appearance of skin rash.
On day two, a localised protrusion of 10 × 12 cm appeared in the right lumbar region of the abdominal wall (Figure 1), particularly when the patient was asked to strain to increase the intra-abdominal pressure. Superficial abdominal reflexes were absent over the protrusion. On day four, gastrointestinal tract (GIT) symptoms started. The patient complained of bloating, nausea, and inability to open the bowel. On hospital admission, the patient was afebrile, blood pressure was 140/85 mmHg, and complete blood count and blood chemistry were within normal levels. The abdomen was generally distended, and intestinal sounds were minimal. CT scan of the abdomen showed gas collection in the small and large intestine with fluid levels. All other organs including the liver, spleen, kidneys, bladder and the pancreas, and the peritoneal cavity were unremarkable.
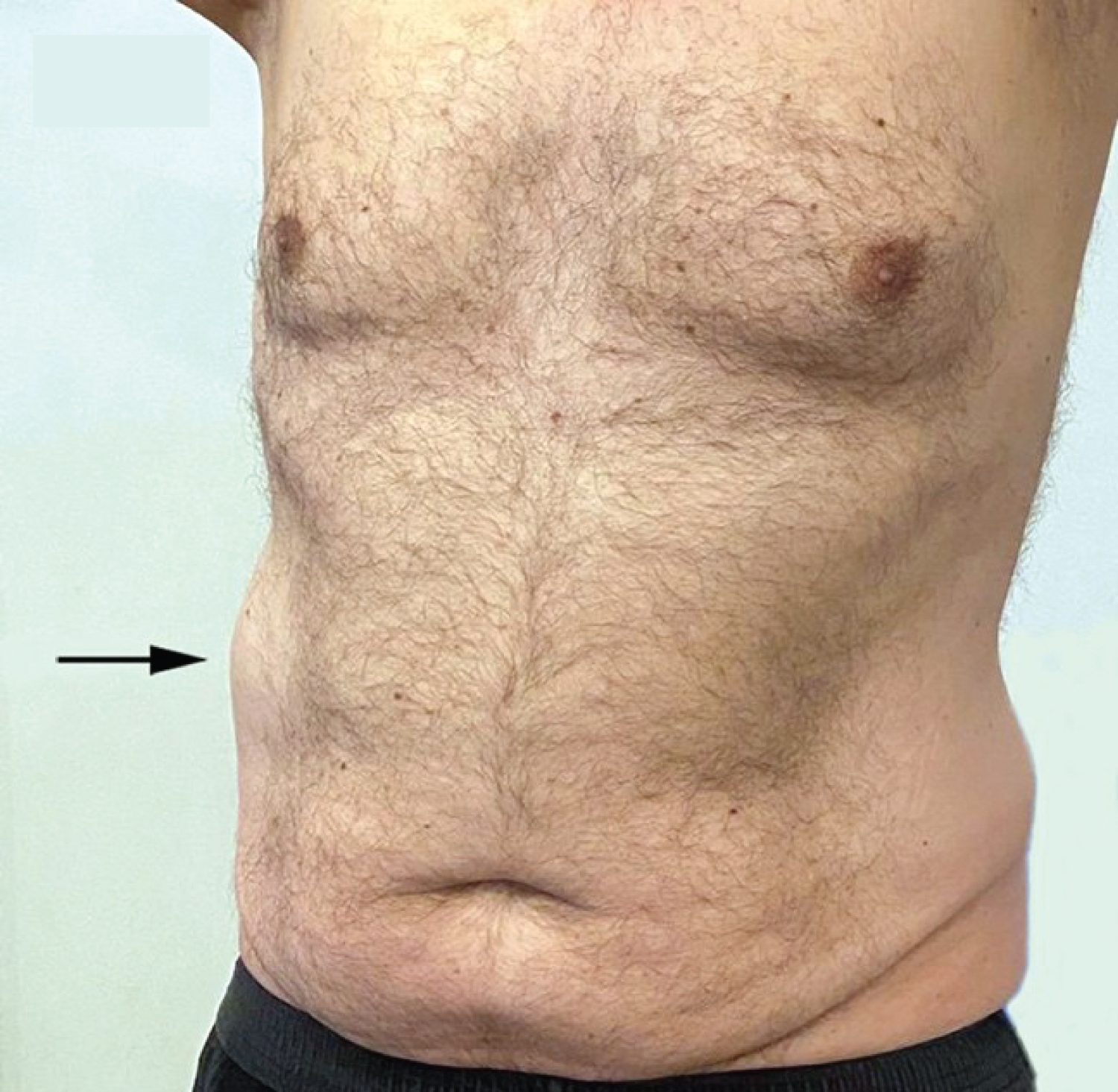 Figure 1: Photograph of the abdominal wall showing a swelling in the right lumbar region (arrow), which appeared three days before the skin rash.
View Figure 1
Figure 1: Photograph of the abdominal wall showing a swelling in the right lumbar region (arrow), which appeared three days before the skin rash.
View Figure 1
On day five, the anticipated skin rash appeared on the right side of the body, on the posterior thoracic wall, flank, and anterior abdominal wall from the level of the umbilicus to the suprapubic region (Figure 2). Crops of cloudy fluid-filled vesicles on erythematous skin patches developed over hours. Oral antiviral Acyclovir 800 mg, 5 hourly was administered for 10 days, alongside supportive therapy including analgesics and topical Acyclovir. On day six, the GIT symptoms and signs stabilized. On day seven, with opening of the bowel, the abdominal discomfort improved, oral fluids were slowly introduced, and the patient was discharged. The rash crusted and the scabs disappeared by four weeks and skin demarcations at the eruption sites disappeared by six weeks. On follow up at five months, all signs and symptoms had resolved apart from the right lumbar region protrusion.
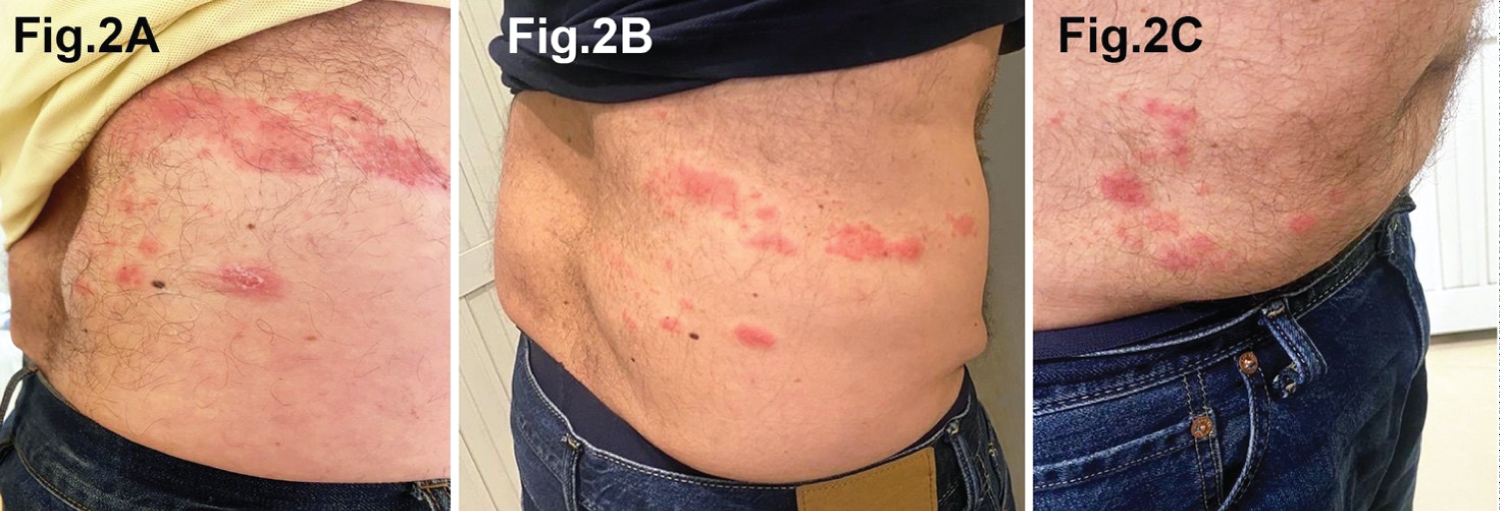 Figure 2: Photographs showing the skin rash at different stages of development in the posterior (A), lateral (B) and anterior (C) abdominal walls. The rash predominantly corresponds to the dermatomes of T10 to L1 spinal nerves.
View Figure 2
Figure 2: Photographs showing the skin rash at different stages of development in the posterior (A), lateral (B) and anterior (C) abdominal walls. The rash predominantly corresponds to the dermatomes of T10 to L1 spinal nerves.
View Figure 2
The reactivated HZV in the neurons of sensory ganglia travels along the peripheral branches of the neurons, reaching the skin where it produces painful vesicular rash in the involved dermatomes. Microscopy of skin lesions shows viral inclusions in epidermal cells, and cell necrosis [1,2]. The DRG of spinal nerves (Figure 3 and Figure 4) contain unipolar neurons surrounded by satellite ganglion cells (SGC), which are modified Schwann cells, and share some features with astrocyte glial cells of the CNS [6]. It is of interest to note that ganglion neurons, SGC, Schwann cells and astrocytes share a common embryonic origin from the neural crest [7]. The SGC play a major role in homeostasis of the DRG neurons. The ganglion neurons (20-100 µm in diameter) are the first order neurons in the neuroanatomical sensory pathways for the transmission of discriminative touch and proprioception (large size neurons), crude/light touch (intermediate size neurons), and pain and thermal sensations (medium size neurons for fast pain, and small size neurons for slow pain and thermal sensations) [6,8,9].
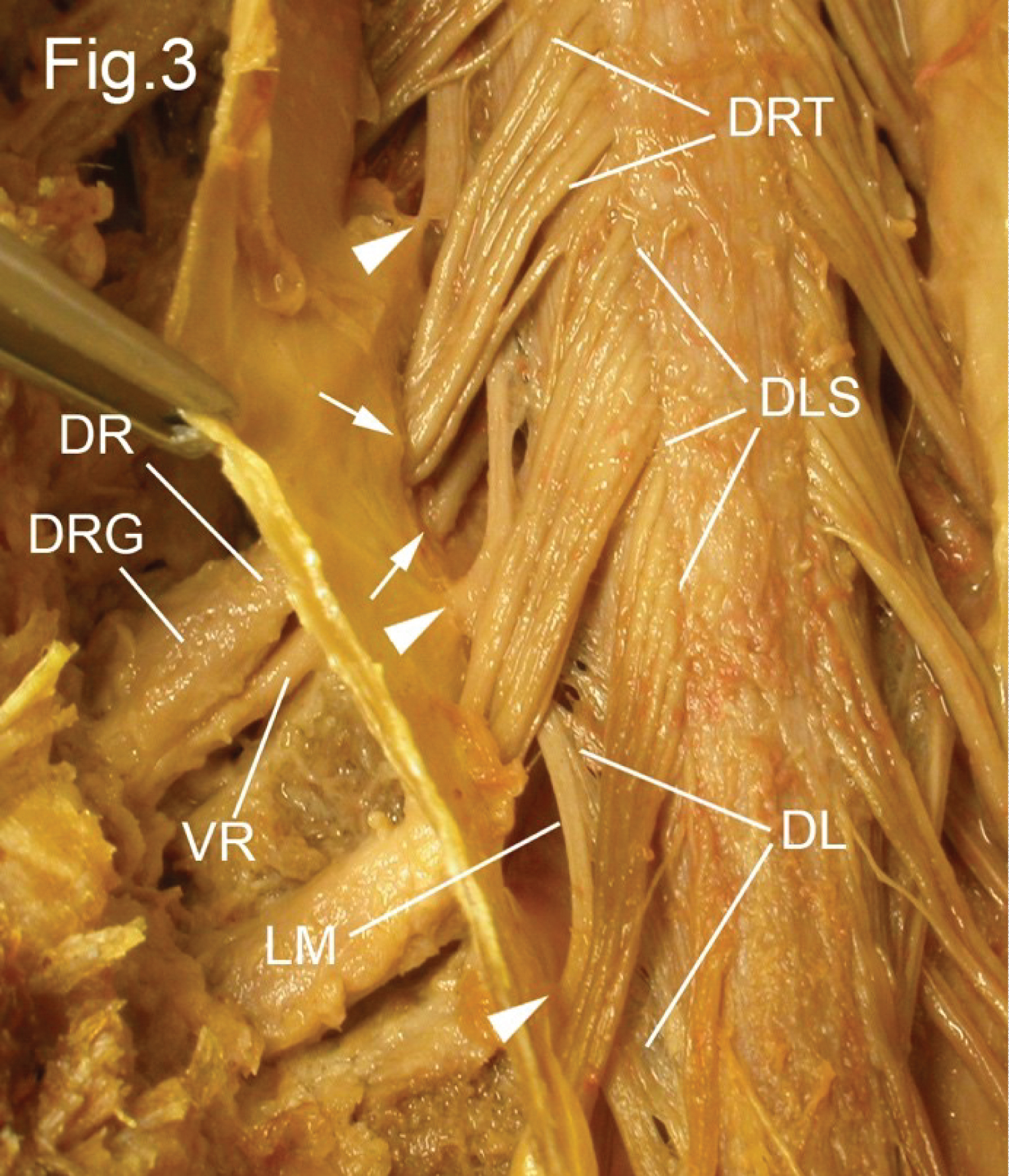 Figure 3: Dissection of the dorsal aspect of the spinal cord. The dura and arachnoid maters were cut longitudinally and reflected, thus opening the subarachnoid compartment, which in the living contains the cerebrospinal fluid. The spinal cord and spinal nerves are seen through the delicate pia mater which intimately invests the cord and nerves. The dorsal roots (DR) split vertically into dorsal rootlets (DRT) which enter the spinal cord along the dorsolateral sulcus (DLS). The dorsal root shows a swelling, the dorsal root ganglion (DRG). The dorsal root (DR) and ventral root (VR) pierce the arachnoid and dura separately (arrows) and acquire a meningeal sleeve from each. At the lateral aspect of the spinal cord the pia mater is pulled laterally to form a double membrane, the denticulate ligament (DL), the free lateral margin (LM) of which has teeth-like projections (arrowheads) which pierce the arachnoid mater and fuse with the dura mater to support the spinal cord.
View Figure 3
Figure 3: Dissection of the dorsal aspect of the spinal cord. The dura and arachnoid maters were cut longitudinally and reflected, thus opening the subarachnoid compartment, which in the living contains the cerebrospinal fluid. The spinal cord and spinal nerves are seen through the delicate pia mater which intimately invests the cord and nerves. The dorsal roots (DR) split vertically into dorsal rootlets (DRT) which enter the spinal cord along the dorsolateral sulcus (DLS). The dorsal root shows a swelling, the dorsal root ganglion (DRG). The dorsal root (DR) and ventral root (VR) pierce the arachnoid and dura separately (arrows) and acquire a meningeal sleeve from each. At the lateral aspect of the spinal cord the pia mater is pulled laterally to form a double membrane, the denticulate ligament (DL), the free lateral margin (LM) of which has teeth-like projections (arrowheads) which pierce the arachnoid mater and fuse with the dura mater to support the spinal cord.
View Figure 3
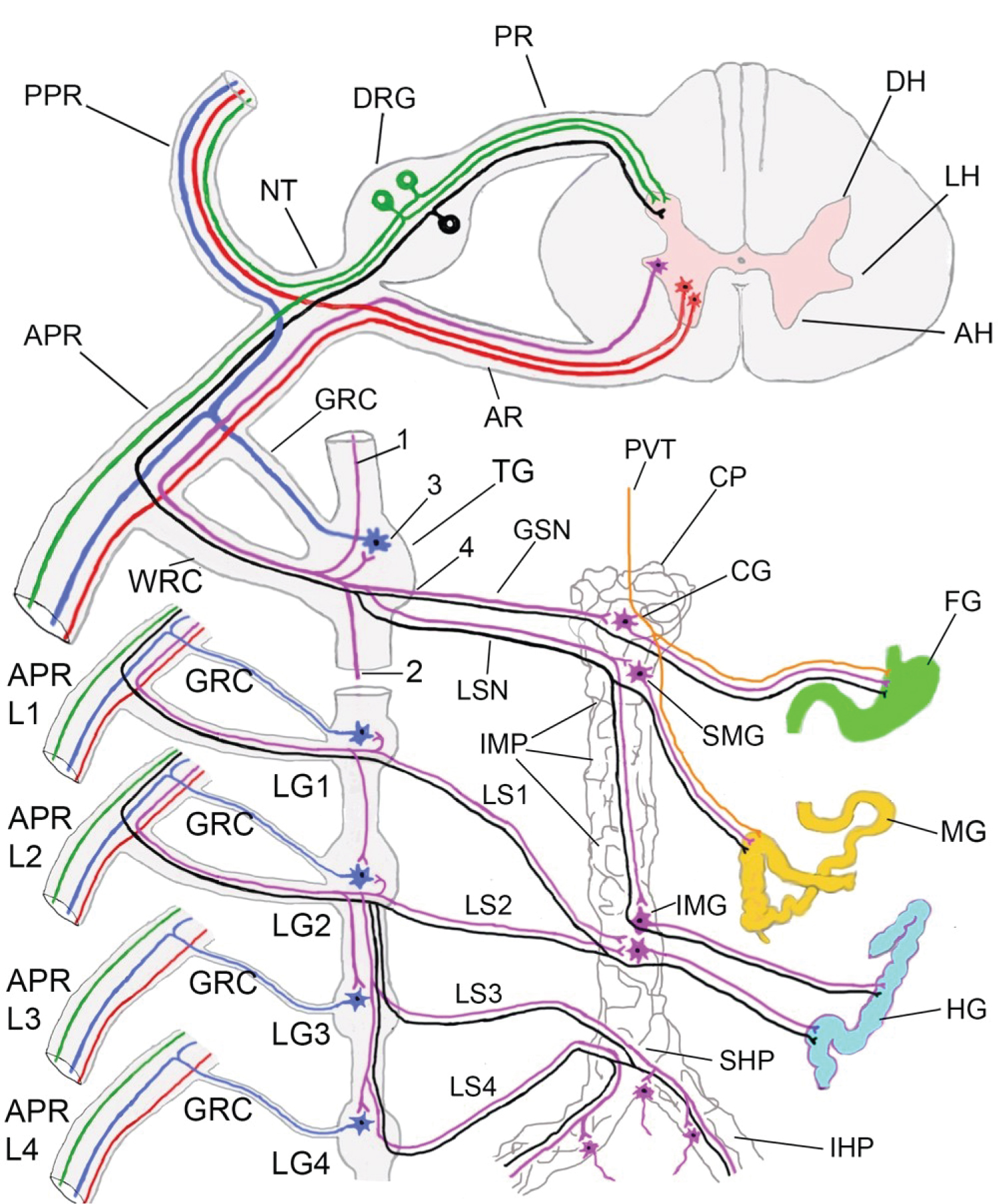 Figure 4: Diagram of a transverse section of a thoracic spinal cord segment, with a spinal nerve attached via a posterior root (PR) and an anterior root (AR). To simplify the diagram one thoracic sympathetic ganglion is depicted (TG) to represent the sympathetic ganglia T5 to T11. The four lumbar sympathetic ganglia (LG1 to LG4) are depicted.
Figure 4: Diagram of a transverse section of a thoracic spinal cord segment, with a spinal nerve attached via a posterior root (PR) and an anterior root (AR). To simplify the diagram one thoracic sympathetic ganglion is depicted (TG) to represent the sympathetic ganglia T5 to T11. The four lumbar sympathetic ganglia (LG1 to LG4) are depicted.
Motor neuronal cell bodies (red colour) reside in the anterior horn (AH) of spinal cord grey matter. Their axons exit the cord via the anterior root (AR), pass through the nerve trunk (NT) and enter the posterior primary ramus (PPR) or the anterior primary ramus (APR) to reach their target skeletal muscles.
The cell bodies of preganglionic sympathetic neurons (purple colour) reside in the lateral horn (LH) of grey matter of spinal segments T1 to L2 inclusive. Their myelinated axons exit the cord via the anterior root, nerve trunk (NT), and anterior primary ramus (APR) then enter the nearest sympathetic ganglion via the white ramus communicans (WRC). The axons may either (1) Ascend in the sympathetic trunk and synapse in a higher ganglion, or (2) Descend in the sympathetic trunk and synapse in a lower ganglion, or (3) Synapse with postganglionic sympathetic neurons (blue colour), or (4) Exit the sympathetic ganglion from its medial aspect without relay and contribute to the splanchnic nerves. The unmyelinated axons of the postganglionic sympathetic neurons (blue colour) enter the anterior primary ramus (APR) via the grey ramus communicans (GRC) to reach the body wall and skin to supply blood vessels, sweat glands, and arrector pili muscles.
The greater splanchnic nerve (GSN) receives fibres from thoracic sympathetic ganglia T5-T9. The lesser splanchnic nerve (LSN) receives fibres from thoracic sympathetic ganglia T9 and T10. Both splanchnic nerves enter the coeliac plexus (CP), which is continuous distally with the inter- mesenteric plexus (IMP), superior hypogastric plexus (SHP) and the inferior hypogastric plexus (IHP). Most of the preganglionic sympathetic fibres in the GSN and LSN synapse with postganglionic sympathetic neurons in the coeliac ganglion (CG) and the superior mesenteric ganglion (SMG). Postganglionic fibres are distributed to derivatives of the foregut (FG) and the midgut (MG) along the branches of coeliac and superior mesenteric arteries respectively. Some preganglionic fibres descend in the inter-mesenteric plexus (IMP) and synapse with postganglionic neurons in the inferior mesenteric ganglion (IMG).
Lumbar splanchnic nerves (LS1 and LS2) originate from lumbar sympathetic ganglia LG1 and LG2 as preganglionic fibres, join the inter-mesenteric plexus and synapse with neurons in the inferior mesenteric ganglion (IMG). Postganglionic fibres supply derivatives of the hindgut (HG) along the branches of the inferior mesenteric artery. The third and fourth lumbar splanchnic nerves (LS3 and LS4) leave the lumbar sympathetic ganglia (LG3 and LG4) and join the superior hypogastric plexus (SHP) and the inferior hypogastric plexuses (IHP). They synapse with postganglionic neurons in discrete ganglia along the pathway to pelvic organs as they follow branches of the internal iliac artery.
Sensory fibres that follow the posterior vagal trunk (PVT, orange colour) enter the coeliac plexus (CP) and reach the foregut (FG) and midgut (MG) via the branches around the coeliac and superior mesenteric arteries.
The unipolar first order somatic sensory neurons for pain, touch, and proprioception (green colour) reside in the dorsal root ganglia (DRG). Each neuron has a single process which splits into a peripheral branch that runs in the posterior or anterior primary rami, and a central branch that enters the cord via the posterior root (PR). The first order visceral sensory neurons for pain (black colour) reside in the DRG of T1 to L2 spinal nerves. Their central branches enter the cord via the posterior root (PR) and their peripheral branches reach the viscera via the nerve trunk (NT), anterior primary ramus (APR), white ramus communicans (WRC), splanchnic nerves, and the coeliac, superior mesenteric, and inferior mesenteric, superior hypogastric and inferior hypogastric plexuses to reach the gut and pelvic organs.
Note: there are only 14 pairs of white rami communicantes connected to anterior primary rami of spinal nerves T1-L2. However, each of the 31 pairs of spinal nerves receives a grey ramus communicans (GRC) from the nearest sympathetic ganglion.
View Figure 4
The most distressing symptom of HZ is neuropathic pain. In the current case the patient complained of sharp lancinating pain alternating with longer-lasting burning pain. This suggests the reactivated virus in the DRG affected the neurons responsible for the fast and slow pain pathways respectively. The fast pain sensation is transmitted by medium size neurons and their thinly myelinated Aδ nerve fibres. The slow burning sensation is transmitted by small size neurons and their type-C unmyelinated nerve fibres [6,10]. In the faster system, painful stimuli are perceived at conscious level in the cerebral cortex via a series of three neurons. The first order neurons are those in the DRG, whose central branches synapse with second order neurons in the dorsal horn of the spinal cord grey matter. Axons of second order neurons cross the midline anterior to the central canal, in the anterior white commissure and ascend in the anterolateral column of the spinal cord white matter, as the spinothalamic tract where they synapse with third order neurons in the ventral posterior lateral nucleus of the thalamus. Axons of neurons of this thalamic nucleus project via the posterior limb of the internal capsule of the brain to reach conscious level in the cortical sensory area in the postcentral gyrus of the brain. In the slow pain pathway, axons of the second order neurons ascend bilaterally in the anterolateral columns of the spinal cord white matter. They establish multiple synapses in the brainstem reticular formation before ascending further and synapsing with neurons of the mediodorsal and intralaminar thalamic nuclei. The neurons of these thalamic nuclei project widely to cortical association areas, the anterior cingulate gyrus, and the frontal lobe [6,10], which may explain the heightened and emotional response of patients to pain.
Crude touch sensation is transmitted to the cortex via a similar series of three order neurons [6,10]. The altered sensation to touch whereby an innocuous stimulus generates pain (allodynia) may be initiated locally. Activation of great numbers of type-C pain afferents leads to the release of inflammatory mediators in the tissues and the sensitization of mechanoreceptors [11]. The release of inflammatory mediators in the spinal cord dorsal grey horn at the end of central branches of first order neurons may lead to sensitization of secondary order neurons, and if sustained may permanently alter synaptic organization in the spinal cord leading to chronic pain. A study of a 76-year-old male who had herpetic rash in the right C4-C6 dermatomes with right segmental paresis of the deltoid and shoulder lateral rotators showed hyperintensity of the right posterior horn of the spinal cord grey matter using MRI, indicating direct spread of the virus to the spinal cord and a close relationship to neuralgia [12].
Experimental studies using human DRG xenografts in immunodeficient mice showed that VZV exhibits specific tropism for SGC and for subtypes of DRG neurons [13]. This experimental system demonstrated the inter-fusion of DRG neurons, fusion of DRG neurons with satellite cells, the presence of VZV genomic DNA in the cells, and the production of viral proteins in neurons and satellite cells. Disruption of the structure and function of satellite cells and neurons in the DRG may contribute to the VZV-related ganglionopathy [13,14].
The distribution of the skin rash in the current case indicates the involvement of dermatomes T10 to L1 [8,11]. Involvement of multiple dermatomes suggests a severe disease [2]. The thoracic nerves are the most-commonly involved nerves [2] and males are four times more likely to develop HZ than females [2,4]. The skin band level with the umbilicus is innervated by T10 intercostal nerve, while the suprapubic skin is innervated by the subcostal nerve and the iliohypogastric nerve. The latter nerve is a branch from L1 anterior primary ramus, and it pierces the external oblique aponeurosis 2-3 cm above the superficial inguinal ring to reach and supply the skin of the suprapubic region [8,9,15].
Cases of motor system involvement with skeletal muscles paresis have been reported [4,16,17]. Upper and lower limb weakness, particularly of the proximal muscles, in association with the skin rash in corresponding dermatomes, have been described [18,19]. More serious weakness of the diaphragm, which is innervated by cervical nerves C3, C4 and C5, has been reported following HZ [20]. Although intercostal nerves are most-commonly affected in HZ, a review of the literature found only 36 cases with segmental abdominal wall paresis [4]. Another study of 1,210 patients with HZ found segmental motor limb involvement in 61 patients. However, only 2 patients had abdominal wall paresis [16]. Despite the above stated motor involvement, HZ has generally been thought of as a disease of the sensory system and many clinicians may not be familiar with motor deficits, because subtle muscle weakness can only be detected using electromyography [21] and the severity of pain may not allow adequate motor examination [19].
In the current case the right lumbar region showed localized abdominal wall bulging suggesting segmental paresis of the flat abdominal muscles. This manifestation is known as abdominal pseudohernia because abdominal wall defects were not detected by scanning [4,17,22,23]. The abdominal muscles are supplied segmentally by spinal nerves T7 to L1, T7 being close to the xiphoid process, and T10 at the level of the umbilicus. The lowest muscle fibres of the transversus abdominis and the internal oblique of the abdomen receive innervation from the subcostal nerve, and L1 spinal nerve via its branches (iliohypogastric and ilioinguinal nerves). Involvement of these nerves by VZV is the likely cause of the lumbar pseudohernia seen in the current case. The lowest muscle fibres of the transversus abdominis and internal oblique fuse medially, become aponeurotic and form the conjoint tendon, which contributes to the medial part of the posterior wall of the inguinal canal. It is located posterior to the superficial inguinal ring of the anterior wall, thereby contributing to the integrity of the inguinal canal [9,15]. Therefore, involvement of the L1 nerve in HZ may lead to pseudo-inguinal hernia. However, no weakness was noticed in the inguinal canal in the current case. Abdominal pseudohernia often follows the appearance of the skin rash [16,24], on average 3.5 weeks after the appearance of the rash [4,22]. However, the current case was unusual as lumbar pseudohernia was seen three days prior to the appearance of the rash, which delayed the diagnosis of HZ. The reported mean period of recovery of segmental abdominal paresis is 4.9 months [4]. In the current case lumbar pseudohernia was still evident when the patient was examined five months after the onset of the disease.
The pathogenesis of motor involvement is generating increasing interest, as it is not clear how the reactivated virus in the DRG is transmitted to motor neurons in the spinal cord or the anterior roots of spinal nerves. DRG lacks the blood-nerve barrier (BNB) that exists in peripheral nerves, such as the sciatic nerve. The BNB is composed of two components: (a) Endothelial cells of endoneurial capillaries, which are joined by tight junctions, thus forming tight capillaries, and (b) The perineurium which bundles groups of nerve fibres in peripheral nerves [6]. The perineurium is formed of collagen fibres and sheets of flat perineurial cells, joined by tight junctions and disposed in consecutive lamellae. The perineurium therefore forms the outer barrier of the bundle, while endoneurial capillaries form the inner barrier. The BNB controls the chemical composition of the endoneurial compartment which surrounds the nerve fibres.
Experimental studies on rodents show that the small molecular weight fluorescent tracer (sodium fluorescein, MW 376; 5 Angstrom diameter) freely enters the DRG after intravenous injection but not the endoneurium of the sciatic nerve [25,26]. Sodium fluorescein also entered the endoneurium of DRG after intrathecal and epidural injections [26]. Another experimental study employing albumin labelled with Evans Blue (MW 69,000) injected intravenously demonstrated entry into the endoneurium of DRG through the leaky capillaries, but not the endoneurium of the sciatic nerve [27]. In peripheral nerves the perineurium controls the entry of molecules and ions into the nerve bundles from the surrounding tissues [25,28]. In the DRG however, the perineurium is replaced by a thin connective tissue sheath with no barrier properties. Experimental studies on pigs demonstrated by light and electron microscopy that the tracer horseradish peroxidase (MW 40,000) injected into the epidural space enters the DRG within 5 mins [29].
Extrapolating from the stated animal experiments, the authors speculate that the DRG in humans lacks a BNB. In the early phases of HZ disease, influenza-like symptoms, and a brief period of viraemia occur [2], possibly via the leaky capillaries of DRG. In a case report, a 68-year-old man developed a painful blistering rash along the C5 and C6 dermatomes with weakness of shoulder abductors and lateral rotator muscles. A PCR test of a sample from his cerebrospinal fluid (CSF) was positive for VZV DNA [10], suggesting that the virus entered the CSF compartment from the infected DRG. Viruses in the CSF might have entered the anterior roots located in the CSF compartment. The pia mater that covers the nerve roots, the DRG, and the spinal cord within the spinal canal is permeable and allows free exchange of nutrients and metabolites between the neuropil of the spinal cord and the endoneurium of the nerve roots on the one hand and the CSF on the other hand [11]. In an experimental study, intrathecally injected fluorescent tracer entered the spinal cord grey and white matter [26]. In an in vivo study also on rodents, macromolecular and ionic tracers injected into the subarachnoid space entered the dorsal and ventral roots indicating that their connective tissue sheath and their pia mater covering are permeable [30]. MRI investigations of HZ patients showed enhancement of ventral and dorsal roots that correspond to segmental muscle paresis and dermatomal distribution of skin rash in the lower limb [31] and the upper limb [32] supporting the view that HZ motor paresis may be due to anterior root radiculopathy.
The intestine has an intricate and complex neural enteric system that can function independently to produce peristaltic movement. The central inputs from the parasympathetic and sympathetic nervous systems modulate the enteric circuits [6]. The enteric nervous system is disposed in two networks within the gut wall; the Meissner's plexus located in the submucosa, and the myenteric (Auerbach's) plexus located between the outer longitudinal and inner circular muscle layers. The two plexuses communicate and have a plethora of neurons: unipolar and bipolar sensory neurons, and ganglionic visceral motor neurons [6]. The components of the network are surrounded by neuroglial cells, like the CNS astrocytes. Visceral neuropathy and pseudo-obstruction of the small and large intestine have been reported as complications of HZ in 19.4% of patients presenting with abdominal pseudohernia [4]. Pseudo-obstruction and abdominal distension often develop concurrent with [5] or after the skin rash [33], thus the diagnosis of HZ is achieved early. In the current case abdominal distension, constipation and signs of intestinal obstruction developed one day prior to the appearance of the skin rash. This knowledge is of particular importance for surgeons as the pseudo-obstruction recovers conservatively within days, and surgical intervention is unnecessary.
The pathogenesis of enteric zoster and how the VZV reaches the intestine is of great interest. Three possible routes are discussed here. Firstly, transmission may be possible via visceral afferent fibres from the intestine (Figure 4). This route resembles viral infection of the skin via somatic afferent fibres. Afferent fibres from the viscera follow either the parasympathetic or the sympathetic nervous system. Those that follow the parasympathetic system are mainly involved in physiological reflexes and they run with the vagus nerve for most of the GIT, or with sacral nerves S2-S4 for the splenic flexure, descending colon, sigmoid colon, and all pelvic organs. The cell bodies of physiological afferents that accompany the vagus nerve are unipolar and located in the nodose (inferior) vagal ganglion, an oval swelling, 2-3 cm long, seen on the vagus nerve just inferior to the skull base. These neurons are classified as general visceral sensory neurons. They regulate blood pressure, heart rate, cough reflex, respiration, GIT motility and secretions. They are distributed via the branches of the vagus nerve: Carotid sinus, internal laryngeal, recurrent laryngeal, cardiac, pulmonary, oesophageal nerves, and the anterior and posterior vagal trunks. The vagal trunks enter the abdomen through the oesophageal hiatus. The posterior trunk joins the coeliac plexus (Figure 4). Sensory fibres then follow the vagal parasympathetic fibres and are distributed via the visceral branches to derivatives of the foregut and midgut (Figure 4) as far as the proximal two-thirds of the transverse colon [6,11]. A smaller number of neurons in the nodose ganglion respond to taste (classified as special visceral sensory neurons). The peripheral branches of these neurons reach a few taste buds in the mucous membranes of the epiglottis and the aryepiglottic folds, at the laryngeal opening. Reactivation of VZV in these neurons leads to mucous membrane eruptions at these sites [34]. The central branches of all the neurons of the nodose ganglion enter the medulla oblongata and synapse with neurons of the nucleus of the solitary tract. A study of human post- mortem samples employing PCR detected VZV DNA in the nodose ganglion [35].
The afferent nerve fibres that transmit pain sensation from the GIT predominantly run with the sympathetic nervous system. They are the peripheral branches of visceral sensory unipolar neurons located in DRG of spinal nerves T5 to L2 inclusive (Figure 4). They share the same ganglia with the somatic sensory afferents [11]. Reactivated VZV in these neurons may reach the intestine via their peripheral branches (Figure 4), and presumably induce oedema and cell necrosis in the wall of the intestine, resembling pathological changes in skin eruptions.
Secondly, VZV may reach The GIT via the efferent sympathetic postganglionic neurons. VZV DNA was detected in post-mortem samples from the coeliac ganglion [35]. The coeliac ganglion is a collection of sympathetic postganglionic neurons found in the coeliac plexus (Figure 4) around the coeliac artery. The coeliac plexus, part of the preaortic plexus, continues distally as the inter-mesenteric plexus, the superior hypogastric plexus, and the inferior hypogastric plexuses (Figure 4). Collections of postganglionic neurons of variable sizes exist in the preaortic plexus. The largest being the coeliac ganglion. Smaller collections form the superior mesenteric ganglion close to the superior mesenteric artery, and the inferior mesenteric ganglion close to the inferior mesenteric artery [6,9] (Figure 4). The axons of these neurons are involved in regulating peristaltic movements by presynaptic inhibition of ganglionic cells in the enteric plexus [6]. Thirdly, it has been suggested that virus-loaded lymphocytes may carry the virus to the viscera [5].
The recovery from abdominal pseudohernia and intestinal pseudo-obstructions have different timeframes. The mean recovery time for abdominal pseudohernia is 4.9 months [4], while the intestinal pseudo-obstruction recovers within days. The different recovery times are likely to reflect the neural pathophysiological changes induced by the virus. According to Seddon's clinico-pathological classification of nerve injury [36], the relatively short time for recovery of neural injury indicates temporary physiological block, without pathological structural damage (neurapraxia). The enteric plexus may become subjected to temporary compression due to viral induced oedema and inflammation in the gut wall.
The long recovery time of abdominal pseudohernia indicates degeneration and regeneration of motor axons, or demyelination and remyelination of motor axons [36]. Studies using highly purified human neuronal cell cultures showed that VZV infection does not induce neuronal apoptosis unlike VZV-induced apoptosis in fibroblasts [37]. However, anterior horn cells overloaded with the virus may become unable to maintain their long axons, leading to axonal degeneration from the terminal towards the cell body (dying back neuropathy) [1]. Axonal degeneration stimulates hypertrophy and hyperplasia of Schwann cells as they breakdown their own myelin sheath. Haematogenous macrophages also become involved in phagocytosis and clearing of the myelin remains and the fragmented axons. The original basal lamina which surrounds individual nerve fibre (axon, myelin sheath and Schwann cells) remains intact [6]. After clearing the remains of the axon and myelin, the dividing Schwann cells packed inside the basal lamina form what is known as the bands of von Bungner. The end of the surviving proximal part of the axon forms a growth cone, which sends advancing processes that grow distally among the Schwann cells, which produce growth promoting factors. The original basal laminae, acting as conduits, guide the growing axons to their targets, the skeletal muscles. Regenerating axons grow at a rate of 2-3 mm per day [1], with some delay at the start until the growth cones are formed, and some delay at the end until the axons establish connections with the target muscle fibres [6]. The resolution of the skin rash takes 3-4 weeks, depending on the immune response and the early administration of antiviral medications. In the current case Acyclovir was prescribed as soon as the rash appeared. Clinical trials showed that oral Acyclovir 800 mg administered five times per day for seven to ten days reduced the duration of pain and the incidence of postherpetic neuralgia [24,38]. Acyclovir was found to be cost-effective by reducing the number of hospital admissions of shingles patients [39].
Surgeons and medical practitioners managing patients presenting with unilateral pain, bulging of the abdominal wall and/or visceral symptoms and signs should suspect herpes zoster, even in the absence of skin rash, to avert unnecessary operative intervention.
The authors declare no conflict of interest and have not received any funding towards this manuscript. The authors have made equal contributions to the manuscript. We would like to thank Dr Stephen Ghabriel for critical reading of the manuscript.