Solitary Pulmonary Cryptococcoma without diffuse infiltration of the lung is an extremely rare manifestation of cryptococcosis, even in immunosuppressed individuals. This report presents an immunocompetent elderly man with a large solitary right lung mass, preoperatively diagnosed as inflammatory lesion with granuloma on CT guided biopsy, later proved to be Cryptococcoma on histopathological examination after Right Postero-lateral Exploratory Thoracotomy and Excision of the Mass.
Lung mass, Cryptococcoma, Thoracotomy
Cryptococcoma is caused by an encapsulated fungus, Cryptococccus sp. C. neoformans and C. gattii are the most common pathogenic species. Immunocompromised individuals are more vulnerable to Cryptococcosis. An estimated 223,000 cases of cryptococcal meningitis occur among people with HIV/AIDS globally each year, resulting in 181,000 deaths annually [1]. Pulmonary cryptococcosis goes mostly unrecognised. The diagnosis of pulmonary cryptococcosis in the immunocompetent is frequently challenging and often misdiagnosed clinically and radiographically. In most cases, diagnosis requires histopathological evaluation.
A 70-year-old male patient, who is a known case of hypertension on anti-hypertensive medications came to our OPD with complaints of dry cough, weight loss and decreased appetite from the past 2 years. There was no history of expectoration, fever, hemoptysis, dyspnea or orthopnea. He was treated initially with oral antibiotics. However, in view of persistent symptoms, HRCT thorax was done (May 2019) which was suggestive of? Inflammatory (Round Pneumonia) with bilateral apical sub-pleural fibrotic lesions with traction bronchiectasis. On the suspicion of TUBERCULOUS EMPYEMA, he was initiated on anti-tubercular therapy in May 2019, following which he improved symptomatically.
In July 2020, he developed a sudden increase in cough with no associated symptoms. HRCT THORAX repeated in October 2020, showed features suggestive of viral pneumonitis, radiologically suspecting COVID infection (COVID-19 RTPCR- negative) and a well circumscribed mass measuring 8.5 × 7.6 × 9.4 cms in the right lower lobe with no evidence of infiltration by the mass, likely Benign aetiology. CT guided Biopsy from the right lung mass was taken; which showed focal areas of necrosis, chronic inflammatory infiltrates and focal sheets of? Foamy macrophages. No granuloma/malignancy.
CECT THORAX was done on 12/11/2020, which showed features suggestive of well-defined benign cystic mass of the posterior mediastinum in the right lower lobe measuring approximately. 10 × 7.7 × 9.9 cm with broad base towards pleura and mediastinum with peripheral smooth enhancing wall with well-maintained margins with lung parenchyma. No evidence of any invasion of parenchymal vessels (Figure 1).
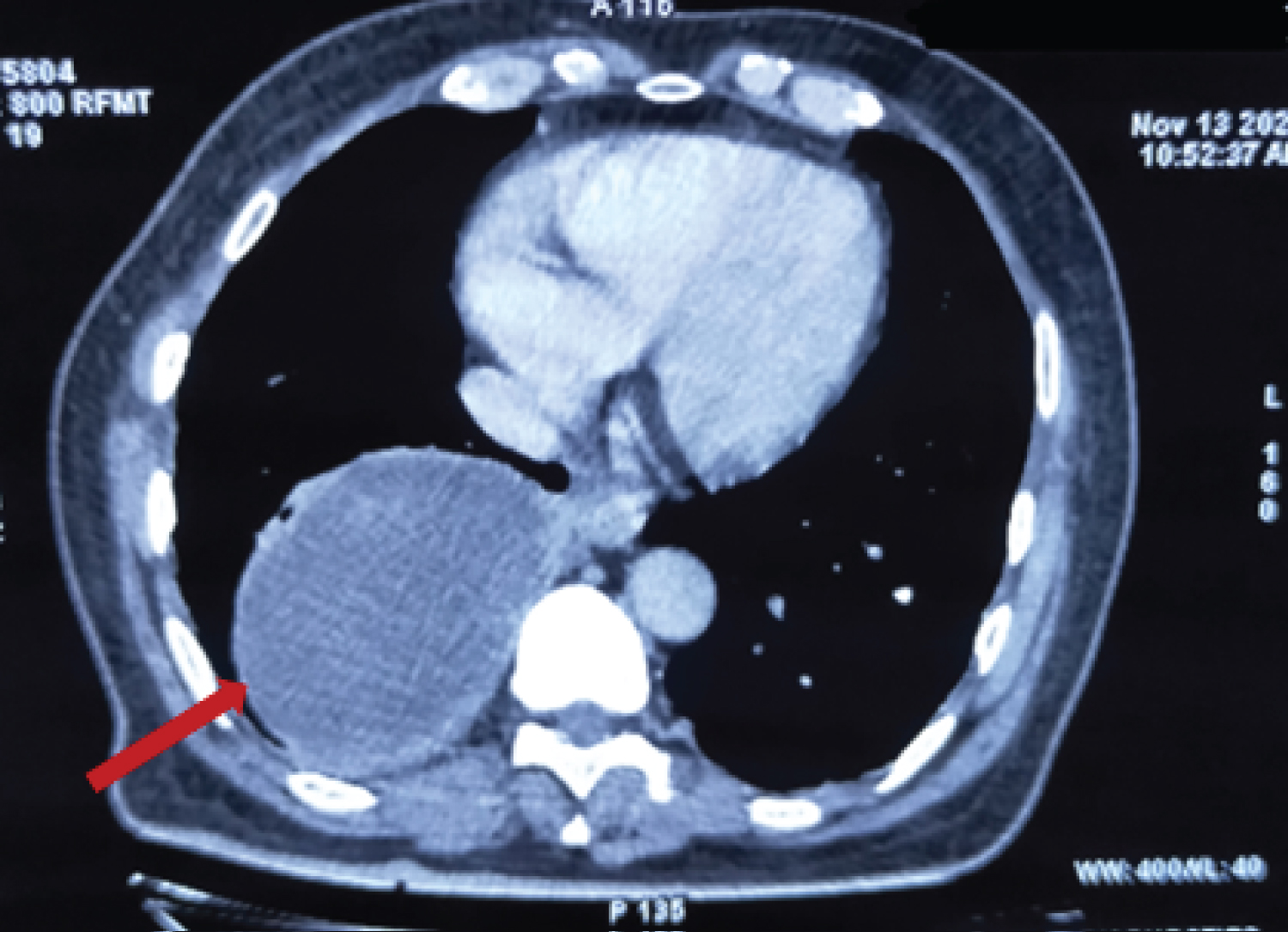 Figure 1: CECT Thorax showing well defined mass in right lower lobe.
View Figure 1
Figure 1: CECT Thorax showing well defined mass in right lower lobe.
View Figure 1
Radiological differential diagnosis -
Esophageal duplication cyst.
Loculated pleural effusion.
Bronchogenic duplication cyst.
Hydatid cyst.
Thereafter, he was hospitalized (3/12/2020) with normal hemodynamic parameters. Breath sounds were reduced in the right lower lung zone. Neurological evaluation was within normal limits. Laboratory work-up was normal, with no immunocompromised status.
After pre-anaesthetic checkup, on 4/12/2020, the patient was taken up for Right Posterolateral Exploratory Thoracotomy. We found a solid mass of size 14 × 12 × 12 cm, minimally adhered to the parietal pleura involving the right lower lobe . A well-defined plane was created between the mass and the rest of the lung tissue. Excision of the mass was done, thereby preserving residual lung tissue (Figure 2), and the post-operative outcome was uneventful.
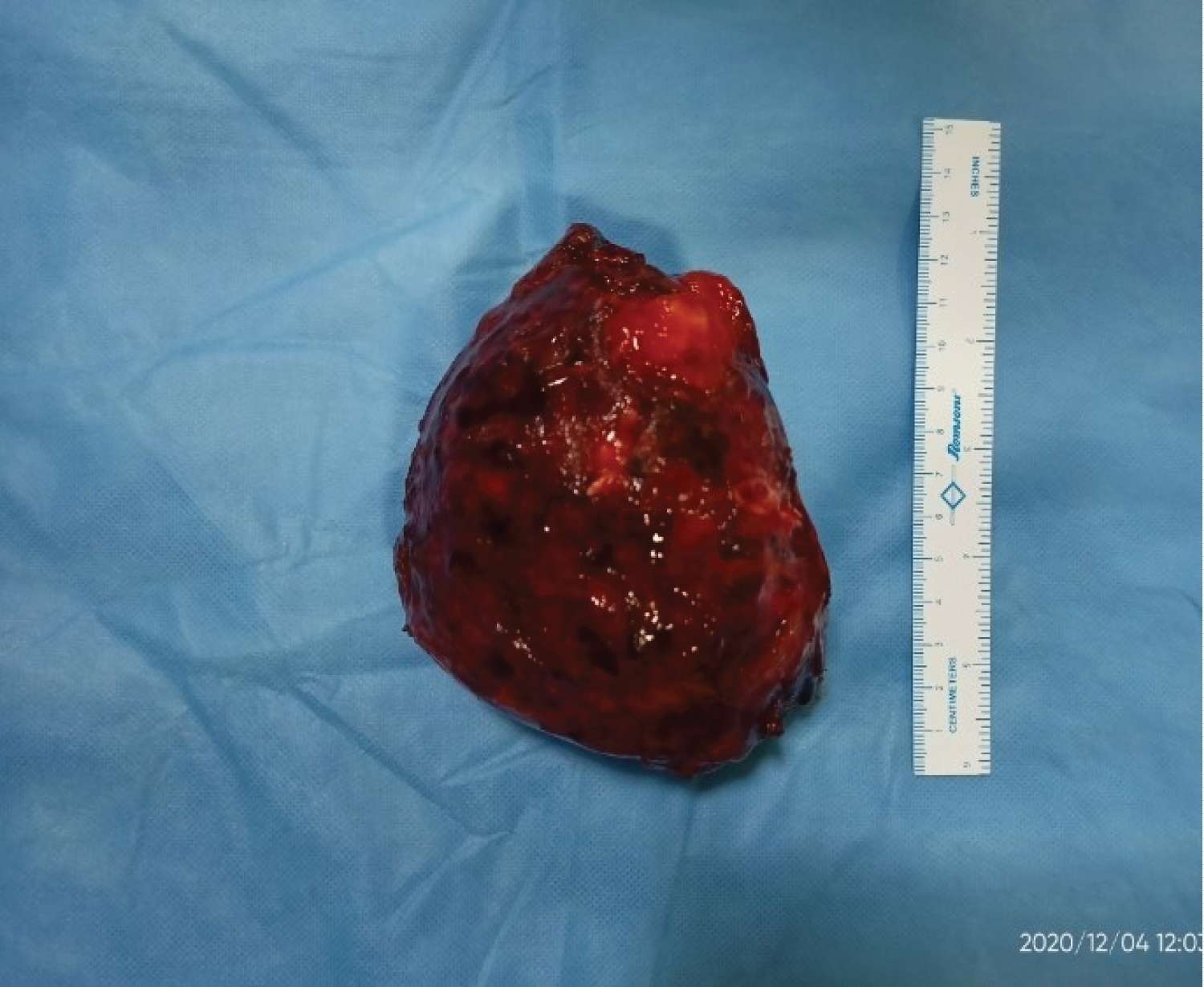 Figure 2: Gross specimen of the mass excised of size about 14 × 12 × 12 cm.
View Figure 2
Figure 2: Gross specimen of the mass excised of size about 14 × 12 × 12 cm.
View Figure 2
The pathological examination revealed a grey white irregular nodular mass, measuring 10 × 6 × 7.5 cm. The cut section showed whitish nodular mucoid and cystic areas (Figure 3A).
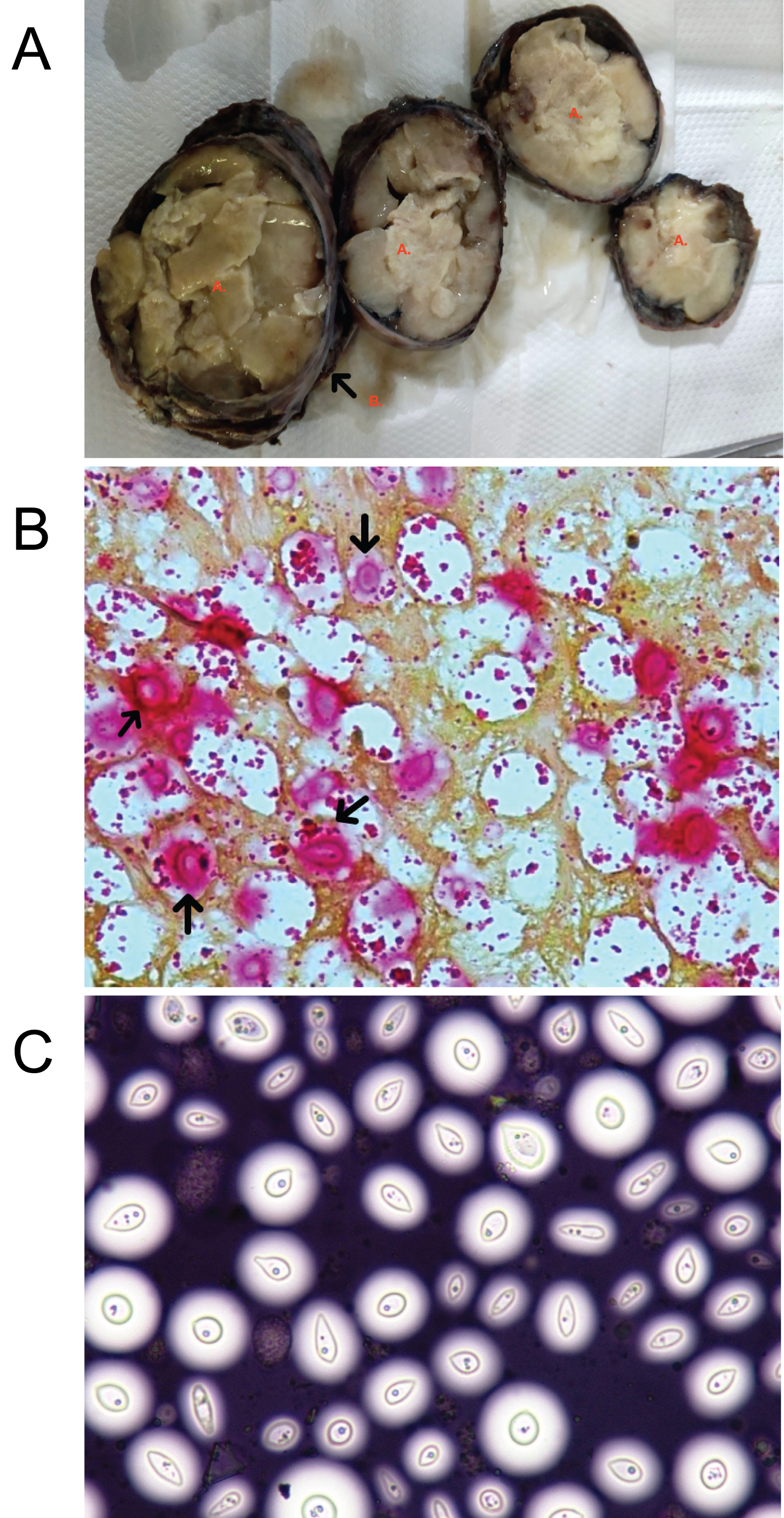 Figure 3: (A) Cut section of the specimen showing whitish nodular and mucoid areas (marked A) with surrounding capsule (marked B); (B) Positive mucicarmine stain - black arrow shows Cryptococcus species; (C) Positive India ink stain showing clear halo around the capsule of the Cryptococcus species.
View Figure 3
Figure 3: (A) Cut section of the specimen showing whitish nodular and mucoid areas (marked A) with surrounding capsule (marked B); (B) Positive mucicarmine stain - black arrow shows Cryptococcus species; (C) Positive India ink stain showing clear halo around the capsule of the Cryptococcus species.
View Figure 3
The histology showed extensive foamy histiocyte infiltration, necrotic areas surrounded by granulomas with epitheliod histiocytes and multinucleated giant cells and presence of numerous round birefringent structures with a peripheral halo morphologically resembling Cryptococcus. The mucicarmine (Figure 3B) and India ink (Figure 3C) staining were positive and diagnosed the structure as Cryptococcus sp. Surgical margins were positive for the infiltration with the organism.
In due course, the patient improved symptomatically. In view of positive surgical margins for the infiltration with the organism, he was started on oral antifungal therapy with FLUCONAZOLE. He was discharged in a hemodynamically stable condition on post-operative day 5. Regular follow up showed no evidence of recurrence clinically or radiographically.
Pulmonary cryptococcoma is a granulomatous mass due to the cavernous destruction of pulmonary tissue [2]. Very seldom, a large solitary cryptococcoma may be the only radiological feature, and with intact cell-mediated defenses, such a presentation is even rarer. Pulmonary involvement varies from interstitial and alveolar infiltrations to large masses, which are frequently first interpreted as lung neoplasm [3].
Patients with cellular immunity dysfunction such as HIV-infection, post-organ transplant patients on immunosuppressants, on long-term corticosteroid therapy, hematologic malignancies, sarcoidosis, or tuberculosis are prone for this fungal infection. Hence, Cryptococcus is an “opportunistic pathogen” [2]. The main route of procurement of human infection is by inhalation of Cryptococcus into the respiratory system [4].
Clinical presentation of pulmonary cryptococcosis is kaleidoscopic, ranging from asymptomatic abnormal radiological findings to symptoms of acute infection like fever, chest pain, cough, expectoration, and weight loss. Meningoencephalitis is the most severe manifestation, due to hematogenous dissemination from the lung.
The most common radiologic features of pulmonary cryptococcosis on computed tomography among immunocompetent patients can be chronicled as well-circumscribed, non-calcified, non-cavitated, multiple, or single nodules of usual diameter < 10 mm with absence of halo, with unilateral or bilateral lung involvement [5]. The usual location is subpleural within the inferior lobes, whereas interstitial and alveolar opacities are present predominantly in immunocompromised patients.
Findings such as consolidation with or without cavitation, lymphadenopathy, pleural effusion, and cryptococcoma are very rarely detected in immunocompetent patients [5].
The differential diagnosis includes primary or metastatic pulmonary neoplasia, granulomatous diseases of the lung, such as fungal or mycobacterial infections and septic embolization [6].
Diagnosis is made by direct detection of the fungus or the cryptococci antigen in the sputum and/or bronchoalveolar lavage. The demonstration of the fungus may be done in the histopathological examination of biopsy specimens or in Bronchoalveolar lavage (BAL) by India ink, Grocott-Gomori, mucicarmine, PAS, and alcian blue staining [3]. In our patient, the histopathological examination of the resected mass yielded the final diagnosis with the aid of mucicarmine and India ink staining.
Treatment of pulmonary cryptococcoma depends on the immunity of the patient and the clinical status [6]. For mild and moderate infections confined to the respiratory tract, treatment should be with oral fluconazole 200 mg/day for 6-12 months and 200-400 mg/day for 12 months respectively, for both immunocompetent and immunocompromised patients. For severe cases, disseminated disease, and CNS involvement, intravenous Amphotericin B and intravenous / oral Flucytosine should be added, followed by oral Fluconazole for up to 12 months in immunocompetent patients and for an undetermined period, or until immune reconstitution in immunocompromised patients [3]. In our patient, since there was no definitive preoperative diagnosis, we proceeded with surgery and postoperatively he was started on anti-fungal therapy as histopathological examination showed infiltration of organism at the margins.
Bumhee Yang, et al. [7] evaluated the role played by surgery in the diagnosis and treatment of pulmonary cryptococcosis (PC) in immunocompetent subjects and concluded that Surgical resection not only afforded reliable diagnosis but also effectively treated pulmonary cryptococcosis. Even additional postoperative antifungal therapy is not required after complete surgical resection.
The diagnosis of pulmonary cryptococcoma in a majority of cases is only made after histopathological examination of the surgical specimen (pulmonary mass), undertaken in the pursuit of neoplasia.
Currently, surgical treatment is the recommended management in case of failure of medical treatment or for the treatment of these pseudotumoral lesions. Based on preoperative radiological and pathological investigations, the etiology in this patient was most likely to be benign, hence considering the age of the patient, only mass was excised , thereby preserving maximum residual lung tissue, followed by anti- fungal therapy based on the surgical margin status. Thus, a synergistic approach for diagnosis and treatment of such cases is required for better patient outcome.
I express my sincere gratitude to Dr. Srinath S. and Dr. Jayanth B. N, consultants in the Department of General Surgery, Apollo BGS Hospitals, Mysuru, for their valuable guidance and thoughtful suggestions in preparing this article.
There is no financial funding received for this article.
None.