Hypothalamus-Pituitary-Adrenal (HPA) may be disturbed by drugs, including opioids. In fact, opioids are a well-recognized cause of hypogonadotropic hypogonadism due to its effects in the hypothalamic-pituitary-gonads axis. Nevertheless, these results are not observed in all the studies and there are numerous contradictions in the literature. Receptor binding selectivity, doses, species used, and administration pathways of opioid derivates may contribute to these different results.
A 36-years-old caucasian man was referred to our hospital with asthenia, facial and distal lower extremity swelling with pitting edema, and vinous red striaes on abdomen for one month. A hormone study was requested finding low levels of cortisol at 8 AM and Adrenocorticotropic Hormone (ACTH) levels. An ACTH stimulation test was performed observing an increase of cortisol levels. With the diagnosis of secondary adrenal insufficiency AI, transdermal Fentanyl 100 mcg per day treatment because of a chronic back pain was suspected as a causal agent. We gradually reduced the fentanyl dosage from 100 to 50 mcg per day and we introduce hydrocortisone 20 mg every 12 h as a treatment. After 10 days, HPA axis was recovered.
Attending to the large number of patients treated with opioids, may be more common than is recognised. The mechanism involved in this disease is not well understood yet, however, there are several contradictions in the modulatory activity of opioids on the HPA axis. According to the possibility of the increase of the prevalence of this pathology, an awareness of this endocrinopathy is essential to the diagnosis and to apply the appropriate treatment with glucocorticoid replacement.
Adrenal insufficiency, Fentanyl, Opioids
Several studies have observed the relationship between drugs and Hypothalamus-Pituitary-Adrenal (HPA) disturbs [1,2]. It has been seen that drug-dependent patients show a neuroadaptative response in underlying elevated stress during an abstinence period. Neuropeptide Corticotropin-Releasing Factor (CRF) is involved in one of these adaptations. It is a molecule which plays a pivotal role in stress and drug withdrawal responses. In humans, it is observed that polymorphisms in these genes that encode CRF receptors have been associated with increased stress responses and propensity to develop drug addiction [3,4]. Due to that, the CRF system has been observed to be a potential target for medication development.
The HPA axis may also be modified in patients affected of chronic pain treated with opioids. In fact, opioids are well known to be a cause of hypogonadotropic hypogonadism [5]. Similar results has been observed in methadone addicts, where previous studies observed decreased plasma cortisol levels as well as an absence of Adrenocorticotrophin Hormone (ACTH) response to naloxone [6]. However, as we described previously, in a rat study, acute injections of morphine without exogenous stress have been found to involve an increased response in Corticotrophin Releasing Hormone (CRH) levels. However, it is observed that exogenous stress as well as repeated daily doses of morphine generated a significant decrease of hypothalamic CRH content, and pituitary and plasma ACTH concentrations levels. Chronic exposure to morphine has also been found to increase corticosteroid-binding globulin implying attenuation of the HPA axis due to a deficit of physiologically active corticosterone [7]. Simililar effects in the HPA axis activity were observed in a rhesus monkeys study using a self-administration of fentanyl model [8].
However, these results are not observed in all the previous studies and there are several contradictions in the literature attending to the modulatory activity of opioids on the HPA axis. Receptor binding selectivity, doses, species used, and administration pathways of opioid derivates may contribute to these different results. [9]. Despite these observations, in many occasions, the HPA axis activity is being taken underrated along the treatment with chronic high-dose opiate medication in clinical practice; that can cause repercussions in the patients.
We report a case of secondary adrenal insufficiency in a patient treated with chronic high-dose of fentanyl.
A 36-years-old caucasian man was referred to our hospital with asthenia, facial and distal lower extremity swelling with pitting edema, and vinous red striaes on abdomen for one month. In addition, the patient observed increased constipation, hair loss as well as difficulty starting urination itchy during this period. A hormone study was requested by his primary care physician, finding low levels of cortisol at 8 AM (2.83 μg/dL, normal values range 5-25 μg/dL). Adrenocorticotropic Hormone (ACTH) levels were also lowers (< 5 pg/ml, normal values range 0-46 pg/ml). The rest of the analytical blood tests values were normal, including electrolytes, TSH, and free T4 levels. He was admitted to the Internal Medicine Department to study a possible adrenal insufficiency.
The personal medical history showed that the patient was allergic to metoclopramide and tramadol. He was intervened of L5-S1 disc herniation with retrolisthesis L5-S1 posteromedial and posterolateral disc protrusion as a sequela of the surgery which causes chronic back pain. Pain was monitorized and treated in the Pain Unit of our hospital. The patient also had chronic diseases such as hypertriglyceridemia, hypercholesterolemia, liver steatosis, hiatal hernia, gastroesophageal reflux, and asthma. The patient also had a history of substance abuse. He smoked approximately 18 packs a year and referred and alcohol intake of 1-2 beers per day for the last 10 years. In addition, until one year ago, he also presented cocaine dependence. The patient's medication list included transdermal Fentanyl 100 mcg (per day), Metamizole 575 mg (every 8 hours), Levosulpiride 25 mg (every 8 hours), Fluoxetine 20 mg (per day), Olanzapine 2.5 mg (at bedtime), and Gabapentin 600 mg (every 8 hours).
Physical exploration was normal except for the vinous red striaes on abdomen as well as the facial and distal lower extremity swelling with pitting edema commented previously. No hepatomegaly or other disturbances were found in the abdominal physical exploration.
During his admission, a chest x-ray was performed without disturbs. Abdominal ultrasound showed normal liver size with an increase of diffuse echogenicity suggestive of fatty infiltration. Brain Magnetic Resonance Imaging (MRI) was also normal.
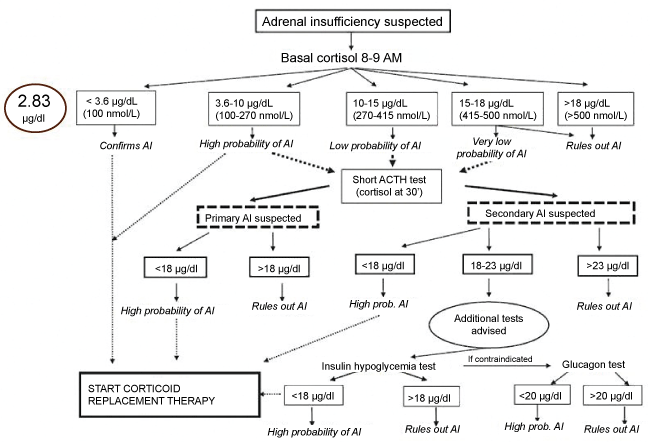 Figure 1: Diagnostic algorithm elaborated by de Miguel Novoa, et al. [10] of adrenal insufficiency. AI was confirmed attending to cortisol values of our patient (2.83 μg/dL).
Figure 1: Diagnostic algorithm elaborated by de Miguel Novoa, et al. [10] of adrenal insufficiency. AI was confirmed attending to cortisol values of our patient (2.83 μg/dL).
AI: Adrenal Insufficiency; ACTH: Adrenocorticotropic Hormone. View Figure 1
According to the diagnostic algorithm elaboration by de Miguel Novoa, et al. [10] (Figure 1), Adrenal Insufficiency (AI) was confirmed. Table 1 summarizes primary and secondary AI etiology [11]. AI etiology, however, considering a possible secondary AI etiology, the patient did not provide a history of cranioencephalic traumatism or glucocorticoid excess intake previously. A normal MRI ruled out a brain tumor as a possible cause of the secondary AI. Reviewing the literature, we observed some case reports where opioid treatments were the causal agent of secondary AI [12,13]. According to this theory, we realized an ACTH stimulation test showing an increase of cortisol levels (6.1 μg/dL). With the diagnosis of secondary AI, we gradually reduced the fentanyl dosage from 100 to 50 mcg per day and we introduce hydrocortisone 20 mg every 12 h as a treatment. After 10 days, HPA axis was recovered showing ACTH levels of 50 pg/ml, and cortisol levels at 8 AM of 8.4 μg/dL.
Table 1: Primary and secondary adrenal insufficiency etiology. AIDS: Acquired Immune Deficiency Syndrome. View Table 1
Opioids are frequently used for the management of painful symptomatology, and their effects on the HPA axis are not well understood yet. In fact, there are several contradictions in the literature attending to the modulatory activity of opioids on the HPA axis. There are described three opioid receptor subtypes: mu, kappa, and delta opioid receptors. In humans, mu opioid analgesics such as fentanyl as well as buprenorphine, have been observed to modulate the HPA activity due to its inhibitory role of mu opioid receptors [9,12-15]. Oltmanns, et al. [12] described a case report of a man affected of secondary adrenocortical insufficiency who has been treated chronically with transdermal fentanyl (200 mcg per day) because of a sciatic pain syndrome. The authors gradually reduced the fentanyl dosage from 200 mcg per day to 100 mcg per day during 1 week. After 1 week, HPA axis function was markedly improved. Similar results were observed by Lee, et al. [13], in which the authors observed an opioid-induced secondary adrenal insufficiency presented as hypercalcaemia. The hypercalcaemia was demonstrated to be generated as a consequence of a secondary adrenal insufficiency, developing as a result of methadone opioid analgesia. After 3 days of treatment with intravenous saline and 10 days of glucocorticoid replacement, the hypercalcemia and the secondary adrenal insufficiency were resolved.
Conversely, in rodents and other non primate species, plasma ACTH and corticosterone levels are observed to be increased due to the activity of mu opioid receptor agonists [9,16,17]. Receptor binding selectivity, doses, and administration pathways of opioid derivatives may contribute to these different results between intraspecies.
In the clinical management of pain, most of the opioid analgesics used are mu opioid receptor agonists. Nevertheless, kappa and delta opioid receptor agonists have been observed to have therapeutic potential effect as analgesics and for other indications [9,18]. In spite of their clinical potential, the modulatory effect of kappa and delta opioid receptor agonists on the HPA axis is not well understood in humans. A human study observed that cortisol release was stimulated due to the activity of a kappa opioid receptor agonist [19]. However, this effect was not determined in this study as a response of a kappa opioid receptor antagonist. Because of that, the authors did not confirm that the observed stimulation was via the kappa opioid receptors. Pascoe, et al. [9] in a study developed in monkeys, used fentanyl, U-50488H, and SNC80 in behaviorally active doses to know the effects of mu, kappa, and delta opioid receptor agonists in the HPA axis. They are highly selective for its receptors, respectively. This study demonstrates that both Mu and delta opioid receptor agonists did not stimulate the HPA axis activity after acute administration of the different treatment in primates. However, synthetic kappa encouraged HPA axis activity.
To our knowledge, there are few human case reports in the literature of opioid-induced secondary adrenal insufficiency. This pathology, attending to the large number of patients treated with opioids for several pathologies, may be more frequent than is diagnosed. This could cause important adverse clinical effects if it is not recognized and treated appropriately. The mechanism involved in this disease are not well understood yet, however, it is observed different effects of opioid treatment between primate and non-primate species as well as the receptors where the different treatments are binded (kappa opioid receptor agonist may stimulate HPA axis in primates where as mu or delta opioid receptor agonists not). The endocrinological scientific societies have not developed a screening program of patients under opioids treatment for possible secondary hypoadrenalism. According to the possibility of the increase of the prevalence of this pathology, an awareness of this endocrinopathy is essential to the diagnosis and to apply the appropriate treatment with glucocorticoid replacement.
No conflict of interest shown in the realization of this writing and no external funding sources and any comments are received.