Remifentanil is a mu-receptor agonist chemically related to fentanyl, alfentanil, and sufentanil that was approved for use in general anesthesia in 1996 by the US Food and Drug Administration [1]. It is a piperidine derivative, a 3-(4-methoxycarbonyl-4-[(L-oxopropyl)-phenylamino]-L-piperidine) propanoic acid, methyl ester. The introduction of the methyl ester group onto the N-acyl side chain of the piperidine ring is what makes remifentanil structurally unique among the currently available opioids. The ester structure makes remifentanil susceptible to widespread ester hydrolysis by non-specific plasma and tissue esterases, resulting in a unique short acting pharmacokinetic profile [2] (Figure 1).
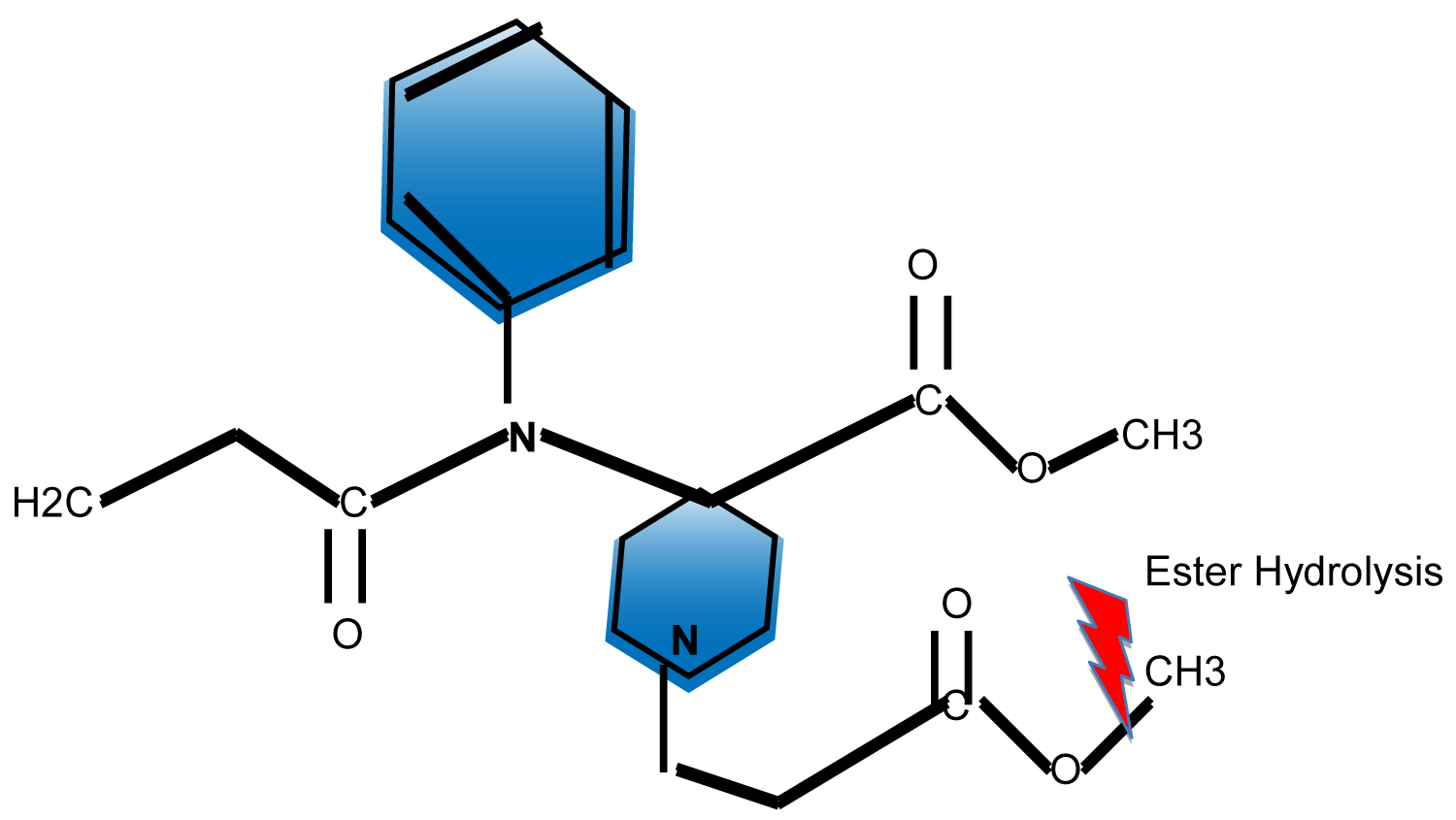 Figure 1: Remifentanil Molecule. View Figure 1
Figure 1: Remifentanil Molecule. View Figure 1
One of the most distinctive features of remifentanil is its favorable context-sensitive half-life or the time required for the drug's plasma concentration to decrease by 50% after cessation of an infusion. The context sensitive half-life for remifentanil is 3.2 minutes and is independent of the duration of the infusion conversely to all other u-agonists [3]. In comparison, after a 3-hour infusion of alfentanil, the context-sensitive half-life was found to be 47.3 minutes. The 50% recovery in drug effect was also compared for remifentanil and alfentanil following a 3-hour infusion. The drug effect, as measured by recovery from respiratory depression, was 5.4 min for remifentanil and 54.0 min for alfentanil [4].
Remifentanil is a weak base with a Pka of 7.07. This chemical property makes it a highly protein-bound drug, approximately 70%, which two-thirds binds to alpha-1 -acid glycoprotein.
Age plays a minimal role in the recovery of remifentanil following a prolonged infusion. The time required for a 50% decrease in effect-site concentration in different populations (20, 50, and 80-year-olds) was examined using complex pharmacokinetic models and was minimally affected by age. All age groups displayed a rapid decrease in concentration but the variability was increased in elderly individuals [5]. Although age was found to play a minimal role in effect-site concentration, age is a significant factor in sensitivity to remifentanil. This is the reason why the remifentanil dose should be decreased in elderly patients to 50% of the dose used in young adults [6].
The significance of body weight on remifentanil pharmacokinetics has been studied. Lean and morbidly obese patients were compared and revealed that remifentanil pharmacokinetic profile is more closely related to lean body weight (LBW) and not total body weight (TBW) [7]. This indicates that obese patients should not receive a weight proportional increase in dosing, doing so may cause significant side effects such as hypotension and bradycardia. One important caveat in this group of patients is that due to their large volume of distribution, an infusion rate started at ideal body weight might take longer to achieve a steady therapeutic plasma concentration, which can be corrected by the administration of a loading dose. The time required to achieve a plasma steady-state of remifentanil by starting a continuous infusion without the administration of boluses is around 15 minutes [8,9].
Unlike existing opioids, remifentanil is metabolized by non-specific esterases that are found in the plasma, interstitial tissues, and red blood cells, whereas the other anilidopiperidine opioids depend upon hepatic biotransformation and renal excretion for elimination. The primary metabolic pathway of remifentanil metabolism results in a carboxylic acid metabolite (GI90291) [10]. We know that the pharmacokinetics and pharmacodynamics of remifentanil are not altered in patients with renal impairment, but the accumulation of its principal metabolite, GI90291, occurs in patients with altered renal clearance mechanisms [11]. Although GI90291 accumulates in patients with moderate/severe renal impairment, its clinical impact is negligible due to its reduced potency [12]. In a study conducted in rats, it was determined that the metabolite is at least three orders of magnitude less potent than the parent drug due to a low affinity to the mu-opioid receptor [13].
Although remifentanil is metabolized by non-specific esterases its half-life is not altered in butyrylcholinesterase-deficient patients. An in-vitro study comparing whole blood, plasma, and red cells between normal and butyrylcholinesterase-deficient volunteers showed no difference in remifentanil kinetics [14]. A case report also demonstrated that in a pseudocholinesterase deficient patient there was not a prolonged response to remifentanil but a prolonged response to succinylcholine was observed in the same patient [15]. Therefore, we can infer from the current data that should be safe to use in this group of patients.
Pharmacodynamically, remifentanil is similar to the other fentanyl congeners. The drug produces analgesia and sedation consistent with potent mu receptor agonist activity. A study using minimum alveolar concentration reduction demonstrated a similar potency between remifentanil and fentanyl [16]. In another study, the potency of remifentanil and alfentanil was compared using the ventilatory response to the rebreathing of carbon dioxide, remifentanil was found to be 40-70 times more potent [17]. In 2008 Maurtua, et al. in a prospective randomized clinical trial determined the degree of potency of different infusion rates of remifentanil in combination with isoflurane IA. They demonstrated that remifentanil at a rate of 0.13 mcg/kg/min had a potency equivalent to half a MAC (One minimum alveolar concentration or one MAC is the concentration of IA in the lungs that is needed to prevent movement in 50% of subjects in response to surgical stimulus). This information is of value to the clinician not only because it allows for a better understanding of dosing and potency but also because it enables the determination of the degree of patient immobility expected at a certain remifentanil dose when combined with IA without the use of muscle relaxants. Avoidance of muscle relaxants is common practice in neuroanesthesia, especially when the patient undergoes intra-operative neuromuscular monitoring through electromyography and or motor evoked potentials monitoring [18].
Another important pharmacodynamic characteristic of remifentanil is its effect on elderly patients. Minto and colleagues investigated the influence of opioid-induced EEG changes based on the spectral edge as a measure of effect. The elderly had a 50% decrease in EC50 compared to younger individuals based on EEG findings. Based on the results we can expect the elderly to require a 50% decrease in infusion rates to what will be clinically indicated in young adults [6].
Remifentanil's adverse effect profile includes respiratory depression, bradycardia, hypotension, muscular rigidity, pruritus, and opioid induce hyperalgesia/opioid tolerance.
The respiratory depression induced by remifentanil is reversible with naloxone. One can expect a prompt spontaneous recovery of the blunted hypoxic responses, shortly after infusion termination [16].
Muscle rigidity is a side effect commonly seen after the administration of many opiate medications. Remifentanil is no exception. However, its occurrence can be decreased by administering remifentanil as a continuous intravenous infusion or over a 30-90 second period [19]. In the intubated patient under anesthesia, the treatment of opioid-induced muscle rigidity is by administering a neuromuscular blocking agent. An alternative treatment is to decrease the infusion rate of remifentanil or to discontinue the infusion. It is yet unclear what dose of remifentanil correlates to the frequency of muscle rigidity however, the incidence of this side effect is less than 1% and is generally dose-dependent.
Hyperalgesia as a side effect of opioid administration has been a subject of debate. In the qualitative systematic review by Angst MS and Clark JD, they mentioned the difficulties found to differentiate between opioid induced hyperalgesia (OIH) vs. acute opioid tolerance (AOT) in several human studies. Their final statement when referring to OIH, stated: "studies in human volunteers involving the short term administration of opioids provide the only currently available direct evidence for the existence of OIH in humans using models of secondary hyperalgesia and cold pressor pain" [20]. In 2009, Fishbain DA, et al. in their evidenced-based structured review declared no sufficient evidence to support or refute opioid induce hyperalgesia except in normal volunteers receiving opioid infusions [21]. Despite these statements about OIH what we do know is that repeated dosing of opioids, as well as the infusion of large doses of remifentanil, will produce tolerance [22]. Also, several human studies showed that when it is about developing OIH and AOT high intraoperative opioid doses are more likely to produce it [18,23-25]. Therefore the clinical approach to these side effects of OIH and AOT, is to foresee the rapid metabolism of remifentanil and one option could be to combine its administration with a longer-acting opiate, especially if we decide to use remifentanil for its rapid onset and offset of action, or to potentiate the effect of a hypnotic agent such as, IA or propofol. Another option is to use opioids with a longer context-sensitive half-life such as fentanyl or sufentanil infusions or hydromorphone or morphine in intermittent boluses, instead of remifentanil, for surgeries where we expect considerable postoperative pain. A third option could be the addition of multimodal analgesia techniques that consists in the administration of acetaminophen, gabapentin and or ketamine and lidocaine infusions that have proved to decrease postoperative opioid requirements [26,27]. Besides, case reports have described that when remifentanil was used in the intensive care unit setting as a sedative, its abrupt discontinuation produced acute withdrawal syndrome [28,29].
Today's use of remifentanil is widespread across anesthesiology. Well-studied areas include maintenance of anesthesia for neurosurgery, ophthalmology, otolaryngology, and obstetrics. One of the most important reasons for the use of opiates in combination with inhalation anesthetics (IA) during neurosurgery, is due to the potentiation they exert on IA, therefore allowing a decrease in IA delivered. This decrease in IA dosing has several advantages in the care of neurosurgical patients. First, there is less IA induced cerebral vasodilation, making the brain more relaxed and less swollen, therefore optimizing surgical conditions, second, cerebral perfusion autoregulation is less affected at lower IA doses, third, after a neurosurgical procedure is performed, patient awakening and neurological exam ideally should be performed with less interference from residual anesthetics. This is where remifentanil excels when compared to other opiates, due to its short context-sensitive half-life. Hemodynamic stability was studied in patients undergoing brain biopsy receiving remifentanil or alfentanil and both shown to elicit better hemodynamic stability compared to fentanyl [30,31].
In pediatric patients remifentanil has been widely used. In the case series reported by Soontrakom T, et al. focusing on neonates undergoing non-cardiac surgical procedures, he reported the benefits of remifentanil’s pharmacokinetic and pharmacodynamic properties in this population, especially the fast awakening and improved respiratory function when compared to fentanyl [32].
In otolaryngology, a comparison of remifentanil with propofol versus a propofol and fentanyl anesthetic for otologic microsurgery demonstrated improved hemodynamic stability, less movement, and faster emergence in the remifentanil group [33].
In obstetric anesthesia, remifentanil patient-controlled analgesia is an alternative in women with contraindications to epidural analgesia or in patients who want to avoid neuraxial analgesia [35]. Several studies have been performed comparing the degree of analgesia, side effects and neonatal outcomes after the administration of IV remifentanil vs epidural analgesia.
Remifentanil has been evaluated against different epidural analgesia local anesthetics including, bupivacaine, ropivacaine and levobupivacaine, in combination with fentanyl. Three studies evaluating these two analgesic techniques, reported higher pain scores in the remifentanil group, while two other studies found no difference in pain scores between patients receiving remifentanil infusions vs epidural analgesia [36-40]. Interesting is to note that despite the apparent remifentanil insufficient analgesia, maternal satisfaction was equivalent in both tested groups.
In terms of side effects such as sedation and oxygen desaturation, they were more common in the remifentanil groups. When evaluating the incidence of instrumental vs spontaneous vaginal deliveries, both analgesic techniques showed the same rate of delivery complications.
Therefore, the conclusion of these findings indicated that despite the superiority of epidural analgesia, the administration of IV remifentanil is still an effective option, however these patients should be continuously and closely monitor to prevent oxygen desaturation and over sedation [36-40].
Kan, et al. studied remifentanil placental transfer, as well as its maternal and neonatal effect, by administering an IV infusion of remifentanil in addition to epidural anesthesia in patients undergoing elective C-sections. They correlated Apgar scores, neurologic and capacity scores, to umbilical vein and artery remifentanil concentrations and maternal arterial remifentanil concentrations. They found that remifentanil crosses the placenta rapidly, but it appears to be metabolized and or re-distributed to a degree that did not affect adversely Apgar scores, neurologic and adaptive scores. Maternal sedation and respiratory changes did occur [41].
In retrobulbar blocks for ophthalmologic surgeries, administering remifentanil over a 30-90 second period before the block can prevent pain during the block [18]. Remifentanil has even been shown to provide pain relief that is superior to propofol [42]. In this study, a downside to this technique of remifentanil mentioned as a sole agent for these blocks is the increased incidence of recall [43]. That said, remifentanil is an opiate, not an hypnotic agent, and therefore expecting no recall from the use of remifentanil alone is unreasonable.
It is important to note that when comparing remifentanil infusions in combination with propofol vs. remifentanil solutions in combination with desflurane IAs, the later combination, tends to allow for faster extubation when the depth of anesthesia is guided by BIS monitoring [44].