Bosma arhinia microphthalmia (BAM) syndrome is a rare condition with less than 100 patients reported. The suggested criteria for BAM syndrome diagnosis include congenital arhinia, hypoplastic maxilla, hypogonadotropic hypogonadism, and normal cognition in males. Our patient presented with unique symptomatology of cardiac abnormalities not otherwise described in literature relating to BAM syndrome. Anatomically, our patient had a patent ductus arteriosus and questionable aortic coarctation, a patent foramen ovale, and pulmonary and tricuspid valve insufficiency. Further documentation must be accumulated to investigate deviations from typical phenotypic presentations including respiratory distress syndromes and cardiac abnormalities to aid further diagnosis and treatment.
Bosma arhinia microphthalmia (BAM) syndrome is an extremely rare condition characterized by abnormalities of the nose and eyes as well as dysfunctions with puberty [1]. There have been less than 100 patients reported worldwide in the past century. The absence of a nose, and in some cases hypoplasia of the nose, is the key feature of the syndrome. This leads to impaired olfactory and gustatory sensation [2,3].
Additionally, patients with BAM syndrome can have microphthalmia or anophthalmia leading to severe vision impairment or blindness. Other potential ocular defects include colobamas and cataracts [1].
Individuals with this syndrome also have hypogonadotropic hypogonadism which leads to a decreased production of hormones that directly influence sexual development. Without involvement of endocrinological treatment, this results in delayed puberty. Affected males may also have underdeveloped reproductive tissues and cryptorchidism [1-3].
A 10-week-old Hispanic male with an absent nose presented with his mother for intercostal and subcostal retractions and increased work of breathing for 24 hours. The mother also stated that the patient had increased secretions in the patient's tracheostomy tube. The patient was born full term at 38 weeks via vaginal delivery weighing 6 pounds 2 ounces and was kept in hospital for the first 2 months of life. A tracheostomy tube was put in at 3 weeks of life because of difficulty breathing while feeding. A gastrostomy tube was put in at 1 month of life per the mother's request. The patient's hearing and vision were tested and were unremarkable. The patient also had been diagnosed with a patent ductus arteriosus and questionable aortic coarctation, a patent foramen ovale, and physiologic pulmonary valve and tricuspid valve insufficiency all of which led to left to right shunting of the heart. The mother has an established diagnosis of systemic lupus erythematosus but is not taking medications. She also stated that she received prenatal care, there were no complications during pregnancy, and was unaware of the condition during pregnancy. Socially, the mother worked as a housekeeper, landscaper, and in construction before she knew she was pregnant. The patient has a brother that is alive and well. Information on the father was not able to be discerned. The rest of the family history was unremarkable. The patient was also referred for genetic screening for BAM syndrome.
A full physical exam was conducted on the patient. The anterior fontanelle was open and no bruits were appreciated. The patient had wide set eyes, downward palpebral fissures, hypertelorism, and microphthalmia. Bilateral tympanic membranes were pearly and gray. The patient had a high arched soft palate. The nose was absent. The neck was supple with a patent tracheostomy tube. A 2/6 systolic murmur at the left sternal border was appreciated as well as a 2/6 murmur which radiated to the back. Bilateral rhonchi were appreciated. The patient was tachycardic and tachypneic. The abdomen was soft and nondistended. The patient had subcostal retractions and a patent gastrostomy tube. He had normal strength and tone and was moving all extremities well. There was no right testicle appreciated; however, a left one was appreciated. Lastly, the patient also had microphallus.
A venous blood gas was taken and proved to be unremarkable. The patient's oxygen saturation was 83%. Polymerase chain reactions (PCRs) were conducted for Influenza A and B as well as Respiratory Syncytial Virus. All three were negative. Chest X-ray was taken and revealed bibasilar opacification which was read as viral pneumonia versus reactive airway disease by the radiologist. The tracheostomy tube was suctioned as well as administration of 1.25 mg of albuterol to the patient who responded well. He had a major clinical improvement with a decreased respiratory rate and decreased subcostal retractions. After being stabilized, the patient was transferred to a secondary care center (Figure 1).
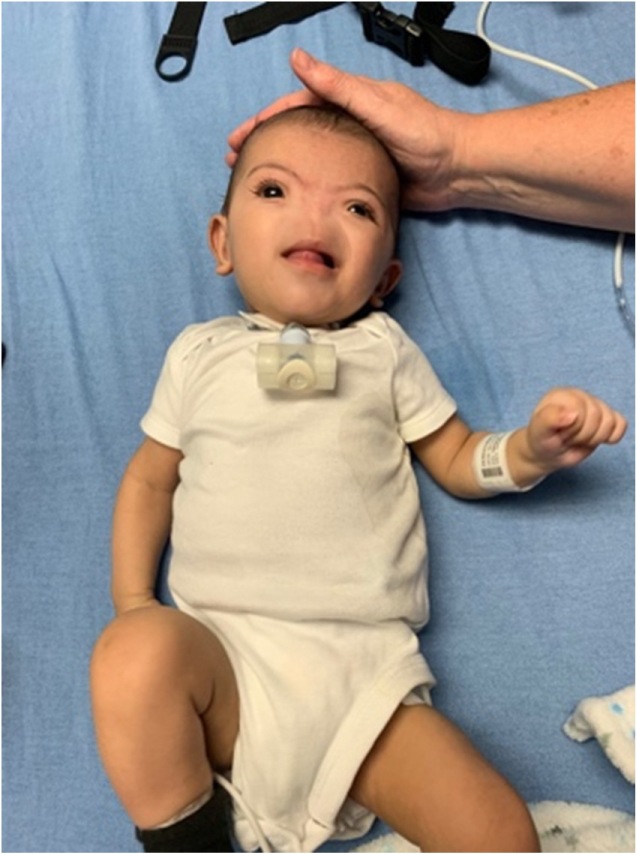 Figure 1: 10-week-old infant with Bosma Arhinia Microphthalmia Syndrome.
View Figure 1
Figure 1: 10-week-old infant with Bosma Arhinia Microphthalmia Syndrome.
View Figure 1
Surgical considerations are typically case dependent and depending on the severity can be quite extensive. It has been noted that a tracheostomy tube is common because of the difficulty with respirations while in early life, especially while feeding. It has also been common practice for individuals with BAM syndrome to get extensive facial surgery to repair whatever structural deformities there might be (cleft palate, choanal atresia). With the advancement of technology in recent years, there has been improvement in the nasal prosthesis that can be made to make them more realistic and appealing. Currently, BAM syndrome has more symptomatic management rather than a defined treatment [3]. It should be noted that in our particular case, the patients current cardiac deformities are being followed by a cardiologist as they might require surgical intervention as well [4].
Bosma arhinia microphthalmia (BAM) syndrome was originally described in 1981 with the identification of 2 unrelated males presenting with severe hypoplasia of facial structures, deficient gustatory and olfactory sensations, and hypogonadotropic hypogonadism with cryptorchidism [5]. Variations of diagnostic nomenclature include "Gifford-Bosma syndrome" and "Ruprecht Majewski syndrome" in acknowledgement of the doctors of the affected patients. According to the National Organization for Rare Disorders (NORDs) Database, less than 100 patients worldwide have been diagnosed with BAM syndrome in the past 100 years. The three defining features of BAM syndrome include: 1) Arhinia, 2) Ocular malformations, and 3) Sexual maturation dysfunction [3].
Our patient presented with unique symptomatology of cardiac abnormalities not otherwise described in literature relating to BAM syndrome. Anatomically, he had a patent ductus arteriosus and questionable aortic coarctation, a patent foramen ovale, and some pulmonary and tricuspid valve insufficiency. Clinically, the patient is currently not receiving treatment related to these cardiac defects. However, in the future there are numerous complications that will most likely need to be addressed including cyanosis, Eisenmenger syndrome, clubbing, polycythemia, and heart failure due to left to right shunting and right ventricular overload. These cardiac abnormalities are notable to mention and transcribe in literature to further understand if they are a part of the etiology of BAM syndrome, if they are a subset within the classification of BAM syndrome, or if they are outliers not representative of the majority of BAM syndrome patients.
There are a variety of theories as to the etiology of BAM syndrome, with mutations of the SMCHD1 gene, PAX6 genes, and de novo reciprocal translocation of t (3;12) (q13.2; p11.2) being the top three most likely causes. One key disease allowing for the observance of SMCHD1 gene function is the association to facioscapulohumeral muscular dystrophy type 2 (FSHD2) involving mutations of the same SMCHD1 gene [6]. The leading theory of SCHDM1 gene mutation resulting in BAM syndrome is due to the strong association with FSMD2 involving mutations of the same gene [7]. Missense mutations of SMCHD1 have been documented in a wide spectrum of phenotypic associations including craniofacial dysfunction, reproductive abnormalities and muscular dystrophy, suggesting the SMCHD1 gene is not a single entity responsible for diseases such as BAM and FSMD2, but rather is part of a multifactorial and pleiotropic transmission pathway. Additionally, there is still controversy in the identification of SMCHD1 mutations having gain-of-function or suppressing functions due to the varying representation of clinical syndromes described in the limited population of patients with BAM syndrome [8,9].
Selected cases of patients have demonstrated unique genetic variations including PAX6 gene mutations and de novo reciprocal translocation of t (3;12) (q13.2; p11.2), that present with criteria inclusive of BAM syndrome [10-13]. This leaves a vast arena to continue accumulating genetic databases for patients presenting with congenital arhinia and microphthalmia.
This aberrant discrimination of genetic causes and clinical presentation suggests the necessity to categorize BAM syndrome with a delineated set of diagnostic criteria to differentiate potential subcategories of this syndrome based on either, etiology, clinical presentation abnormalities, or a combination of both factors. Currently the suggested criteria for BAM syndrome diagnosis include congenital arhinia, hypoplastic maxilla, hypogonadotropic hypogonadism, and normal cognition in males. As stated, further documentation of cases must be accumulated to evaluate and take into account females with arhinia and microphthalmia, and deviations from typical phenotypic presentations including respiratory distress syndromes and cardiac abnormalities. Additionally, ethnic considerations must be taken into account when determining the increased risk for developing BAM syndrome as well as future treatment protocols. In patient cases such as ours who present with outlier-like presentations, they must be determined where they fall into the spectrum of BAM syndrome. At this time, surgeries that are usually required for patients rely ultimately on the presentation and severity of the deformities associated with their syndrome. This will ultimately help current and future patients diagnosed with BAM syndrome to have an understanding of their diagnosis as well as expected treatments and future complications.
None.
None.