In this rare case we describe a 56-year-old woman who presented with an acute pulmonary embolism (PE) complicated by right heart strain indicating the need for catheter-directed mechanical thrombectomy. Pre-operative two dimensional (2D) transthoracic echocardiogram (TTE) showed no evidence of intracardiac thrombi. Subsequently, post-operative 2D TTE indicated large biatrial thrombi with no evidence of PFO, or other intracardiac shunt. Here, we describe this rare and unique presentation and discuss the possible causes of such finding to help prompt diagnosis and guide management.
Thrombus, Intracardiac, Biatrial, Echocardiogram
Intracardiac thrombi typically develop in the setting of multiple underlying disorders. Right atrial (RA) thrombi are occasionally found with pulmonary embolism (PE), or under the setting of atrial fibrillation (AF), but are rarely found in sinus rhythm (SR) [1,2]. Left atrial (LA) thrombi are most commonly found in the setting of AF, and sometimes with severe mitral valve disease, and like RA thrombi are almost never found in a patient in SR [3]. To take this one step further, the development of biatrial thrombi in the setting of SR, is an even rarer phenomenon. There are only a handful of case reports that describe the finding of biatrial thrombi in the setting of SR [1,2,4,5]. In the cases presented by Oe, et al. [2] and Arslan, et al. [4], the patients had preexisting conditions of heart failure (HF) and restrictive cardiomyopathy, respectively. Other documented cases are reported in patients with a patent foramen ovale (PFO), here the biatrial thrombus is known as "thrombus in transit" [1,5]. To our knowledge, the presentation of biatrial thrombi in SR without the presence of preexisting conditions described above has not been well documented [1,2,4,5]. We herein report a case of a 56-year-old woman who presents with acute pulmonary embolism (PE) and incidentally found biatrial thrombi after catheter-guided partial thrombectomy of PE.
A 56-year-old woman presented to our hospital with sudden onset chest pain and dyspnea. The day prior, she was discharged with close follow up after presenting with left lower extremity pain, stable vital signs, and a negative venous ultrasound duplex for deep vein thrombosis (DVT) of the left lower extremity. The patient was reported to have a near syncopal episode and was hypoxic on room air. On transport, the patient was loaded with aspirin and sublingual nitroglycerin with no improvement of symptoms. The patient's past medical history is significant for morbid obesity (BMI 43), hypertension, type II diabetes mellitus, asthma, and hyperlipidemia. Family history was significant for a sister with lower extremity deep venous thrombosis (DVT) in adulthood, and a son with lower extremity DVT in college. On physical exam the blood pressure was 102/61 mmHg (189/94 mmHg the day prior), and her heart rate was regular at 115 beats/min. The patient was tachypneic with a rate of 24 breaths/min with increased work of breathing saturating 96% with non-rebreather mask. Her lungs were clear to auscultation and no other physical exam findings were present. EKG showed sinus tachycardia with nonspecific ST-T waves changes, and troponin peaked at 0.063 ng/mL. Chest X-ray was unremarkable. In the emergency department, the patient was started on a continuous heparin infusion. A pulmonary embolism protocol computed tomography angiography showed multiple bilateral pulmonary emboli (Figure 1a). The patient was admitted to the intensive care unit and underwent a transthoracic echocardiogram (TTE) which showed a severely dilated right ventricle (Figure 1b) with associated right ventricular systolic dysfunction. Left ventricular ejection fraction (EF) was preserved at 55%. No intracardiac thrombi were noted. The significant right heart strain prompted mechanic partial thrombectomy of the right lobar pulmonary embolism. Post-procedural TTE showed an echodense 0.9 × 1.5 cm mass in the left atrium that prolapsed into the ventricle during diastole, and an echodense 1.4 × 1.7 cm mass in the right atrium (Figure 1c and Figure 1d). A transesophageal echocardiogram (TEE) with bubble study was planned given the presence of biatrial thrombi and the indication to rule out a PFO. Unfortunately, the hospital course was complicated by acute thrombocytopenia with progressively enlarging bilateral intracardiac thrombi. Platelet count values on admission to the hospital were 286 × 109/L. Six days after admission the platelet count dropped by > 50% to 136 × 109/L, and the following day reached nadir at 62 × 109/L. Subsequently, heparin was discontinued, and argatroban was started. Platelet-factor-4 (PF4) antibody test and serotonin release assay were ordered to rule out heparin induced thrombocytopenia (HIT) type 2. They both came back negative in initial and repeat study. The rapid drop in platelet count was a contraindication to TEE, as a result, TTE with bubble study was performed. No evidence of PFO, or any other intracardiac shunt was noted. Cardiac magnetic resonance imaging was done for further evaluation of progressing bilateral atrial thrombi, and to again look for a possible interatrial shunt. Again, there was no evidence of PFO. The patient ultimately recovered with no major complications and was discharged on lifelong anticoagulation therapy. A workup for systemic hypercoagulability was performed in the outpatient setting.
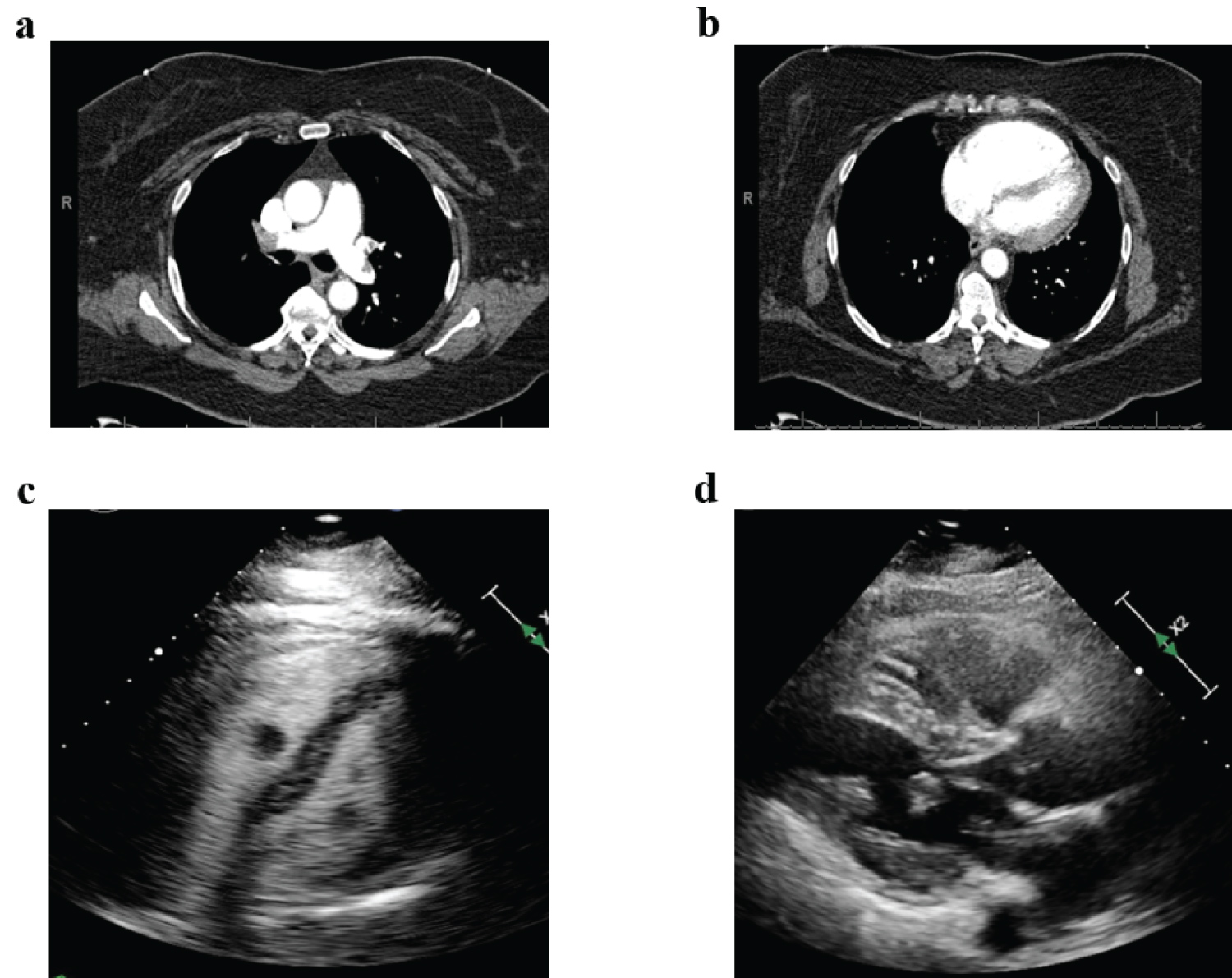 Figure 1: (a) Pulmonary embolism visualized in the right and left main pulmonary arteries bilaterally; (b) Bowing of the intraventricular septum with a right ventricular dilatation, suggestive of right heart strain; (c) Biatrial thrombus prolapsing to ventricles during diastole; (d) Left atrial thrombus prolapsing to left ventricle during diastole.
View Figure 1
Figure 1: (a) Pulmonary embolism visualized in the right and left main pulmonary arteries bilaterally; (b) Bowing of the intraventricular septum with a right ventricular dilatation, suggestive of right heart strain; (c) Biatrial thrombus prolapsing to ventricles during diastole; (d) Left atrial thrombus prolapsing to left ventricle during diastole.
View Figure 1
Work up for inherited thrombophilia was performed given the clinical dilemma described above. Hypercoagulable studies demonstrated Factor V Leiden wild type and Prothrombin 20210 wild type. Studies for Antithrombin III Deficiency and Lupus Anticoagulant Panel (including Antiphospholipid and Lupus Anticoagulant Antibodies) were negative. Protein C and S deficiency testing were done as would not be valuable in the setting of long-term warfarin use. Follow up 2D TTE showed no evidence of intracardiac thrombi, showing complete resolution of previous findings.
Atrial thrombi are most commonly detected in association with atrial arrythmias, with AF being the most common type [3,6]. When a patient presents with an atrial thrombus, while in SR, the etiology of this finding must be further investigated. While the presence of the RA thrombi in the setting of submassive PE (defined as hemodynamic stability with right heart strain), is an occasional finding (although more common in massive PE defined as hemodynamic instability plus right heart strain), it is the concomitant LA thrombus that has created this clinical dilemma [1].
A shunt between the venous and arterial system, or RA to LA, known as paradoxical emobli, is a possibility for the patient's findings. The most common trans-atrial shunt is a PFO, which is found in about 25% of patients [1]. Other less common causes of shunts are extracardiac arteriovenous connections, like a patent ductus arteriosus or pulmonary arteriovenous fistula [7]. While a thorough workup for PFO or other atrial septal defect (ASD) showed no evidence of trans-atrial septal flow, it is possible that the thrombi are occluding any previously patent orifice.
When a PFO is not present, other less common causes of LA thrombus in SR needs to be investigated. According to Agmon, et al. [3], there are three high-risk features for LA thrombus in patients with SR; significant left ventricular dysfunction (EF < 30%), aortic or mitral valve disease, or a previous history of AF. These specific cardiac abnormalities were not present in our patient.
To add to the mystery, it was not until after PE intervention, did the biatrial thrombi present. While it was possible the developing thrombi were missed on initial 2D TTE, the etiology of LA thrombus was still not explained. However, detection of intracardiac thrombi with acute PE, does complicate management, as there is increased risk for systemic embolization of LA thrombus when undergoing catheter-based mechanical thrombectomy [8]. In our patient, who was on continuous parenteral anticoagulant therapy since diagnosis of PE, the FlowTriever20 was used, which is a catheter-based mechanical thrombectomy device. Although no complications were noted during the procedure, it is possible that a trans-atrial shunt was created, or PE retrieval resulted in distal embolization.
With the assumption that a PFO or other ASD, was in fact not present, and the findings of this case were not a complication of the PE catheter-directed thrombectomy, the investigation needed to explore other explanations such as hypercoagulability disorders. The first plausible cause would be cancer related hypercoagulability of both the arterial and venous system. The patient reported that she was up-to-date on cancer screening. Primary work-up for major malignancies were negative. Given her age, risk factors, and overall clinical picture an occult malignancy presenting with PE and intracardiac thrombi could not be absolutely ruled out. The patient was admitted to our hospital in July 2020 in the middle of COVID-19 surge, when we had about 500 COVID-19 admitted patients in our hospital limiting our thorough diagnostic work-up. Moreover, no malignancy-related signs and symptom noted later during one-year follow-up.
The next possible explanation, although already worked up during the hospital course, was HIT type 2. Intracardiac thrombi presenting with HIT is a rare phenomenon reported in a few cases [9]. In a case report by Ahmad, et al. [9], the group describes a patient with HIT type 2 who developed multiple intracardiac thrombi. Similar to our case, the thrombi were present in both the RA and LA. The study reported that the increased RA pressures forced the thrombus to protrude through a closed interatrial septal orifice. While it seems like a plausible explanation for our case, a negative work up argues against it. The ELISA test for PF4-heparin antibodies and serotonin release assay were both negative, and the chances of our mysterious findings being a result of a false negative test on two studies was almost zero.
Lastly, the need to work up other hypercoagulable causes, such as congenital thrombophilias was important given the positive family history. Although the patient denied any prior history of thromboembolism, she reported a sister and son with unprovoked DVTs at a young age. Her workup so far has been negative for the most commonly inherited thrombophilia disorders, Factor V Leiden mutation and Prothrombin G20210A mutation [10]. These mutations although commonly present with unprovoked DVT and PE, rarely present with intracardiac thrombi [10]. When they do it is typically right sided intracardiac thrombi [10]. Antiphospholipid Syndrome (APS) has been reported to consist of both arterial and venous thrombosis. Although intracardiac thrombi are rare with APS, thrombus formation has been described in every cardiac chamber [11]. While all studies (e.g., Lupus Anticoagulant Panel) were so far negative, determining the exact inherited thrombophilia mutation could contribute to solving this clinical mystery. However, hypercoagulable workup did not change clinical management. The presented case will most likely require lifelong anticoagulation therapy in the setting of life-threatening thromboembolism with significant clot burden.
In conclusion, herein, we described a rare case of acute PE complicated by biatrial thrombi without known predisposing factors for left-sided intracardiac thrombi development.
The patient has given permission and informed consent for the publication of this case report.
The presented data in this case report are available upon reasonable request.
The authors have no financial relationships to disclose.
No external funding was utilized to support this study.
All authors have read and approved the manuscript and have taken care.