Acute leukemia, the commonest cancer in children, accounts for approximately 30% of all childhood malignancies approximately 75% of which are Acute Lymphoblastic Leukemia (ALL) affects children of any age, commoner in ages 1-4 years, boys more than girls [1]. The kidneys are sites of extra medullary leukemic involvement usually seen late in the course of the disease [2].
ALL with renal involvement may mimic Wilms's tumor due to overlap of their clinical and radiological findings. These include malaise, anemia, easy bruising, uncontrolled clotting, pedal edema, ballot able kidneys, deranged renal function tests, signs of immunosuppression and common imaging features of enlarged kidneys bilaterally in both diseases [3].
We report the case of a 3½-year-old male child with ALL whose initial working clinical diagnosis was a Bilateral Wilms's Tumor. However, plain chest radiographic findings of a superior mediastinal mass, the ultrasound and CT findings of bilaterally enlarged kidneys, a rarity in Wilms's, prompted further evaluation for proliferative hematologic disorder. Subsequent hematologic investigations led to the definitive diagnosis of ALL.
Patient responded well to treatment with steroids and chemotherapeutic agents and had sustained clinical and radiological improvement.
Acute lymphoblastic leukemia, Wilms's tumour
Leukemia is a group of malignant diseases of the hematopoietic system characterized by the uncontrolled overproduction of either immature (acute leukemia) or terminally differentiated leukocytes (chronic leukemia). Acute leukemia, the commonest cancer in children, accounts for approximately 30% of all childhood malignancies. Approximately 75% of these tumors are found before the age of 5 years [1].
Although ALL is among the most curable cancers, the University of Maryland Medical Center reports that there are approximately 1,400 deaths each year in the United States due to ALL. The cure rate being higher in children than in adults [4]. According to St. Jude Children's Research Hospital, boys are more affected than girls and it is also commoner in whites than in blacks. About 98-99% of children experience complete remission within six weeks of treatment and 90% are leukemia-free for at least ten years, when they are considered to be cured [5].
Wilms' tumor is the most common cause of pediatric renal mass, accounting for over 85% of cases and for 6% of all childhood cancers. It typically occurs in early childhood (1-11 years) with peak incidence between ages 3 and 4, which is similar to that of ALL [6].
Children with Acute Lymphoblastic Leukemia (ALL) often present with signs and symptoms that reflect bone marrow infiltration and/or extramedullary disease. When leukemic blasts infiltrate the bone marrow, patients present with signs of bone marrow failure, including anemia, thrombocytopenia, and neutropenia. Other presenting signs and symptoms of pediatric ALL include the bone pains, arthritis, fevers (low or high), fatigue, lymphadenopathy and hepatosplenomegaly. Extramedullary masses may precipitate signs in the abdomen which may include vomiting, lethargy, head/neck; CNS involvement such as headache, nuchal rigidity and mediastinal mass results in respiratory distress/stridor.
Majority of the presenting signs and symptoms listed above may well point to the diagnosis of a Wilms' tumor, especially in this index case where patient's presenting complaint and clinical signs favoured the diagnosis of Bilateral Wilms' tumor.
Radiologic investigations of chest radiography, USS and CT Abdomen revealed bilateral enlarged kidneys with no mass lesion, hepatosplenomegaly, prevertebral mass lymphadenopathy and anterior superior mediastinal mass prompted the suspicion of lymphoproliferative disease. Hematologic investigation was therefore conclusively employed in the definitive diagnosis of ALL.
It is therefore pertinent for the clinician to have a high level of suspicion, to save the time lost to misdiagnosis and reduce the high rate of mortality accrued to an otherwise highly treatable ALL as compared to Wilms' tumor whose general clinical outcome is poor.
A 3 years and 6-month-old male pre-school child presented with history of abdominal distension and bilateral leg swelling of one month duration. Abdominal distension was sudden in onset, painless and progressive. Three days later, he developed bilateral pedal swelling. There was no facial swelling, reduction in urine output, hematuria or frothiness. The appetite was good but there was remarkable weight loss while the bowel habit remained essentially normal. There was associated occasional non-productive cough with difficulty in breathing of two weeks duration and no hemoptysis. Family history of similar illness was not given.
Physical examination revealed a chronically ill-looking male child. He was febrile (37.7 °C), pale, anicteric, well hydrated, no pedal edema but with significant weight loss. Chest auscultation showed dyspneic patient with coarse crepitation/pleural rub on the left lower lung zone with reduced air entry ipsilaterally. There was tachycardia with pulse rate of 132 beats per minute and normal heart sounds (S1 and S2).
The abdomen was grossly distended with bilateral flank fullness, bilateral firm ballotable kidneys (L >> R) with irregular surface. A working diagnosis of bilateral renal masses possibly Wilms' tumour with pulmonary metastasis was made.
• Chest Radiograph (Figure 1) revealed mediastinal widening with a wedge-shaped opacity in the anterior mediastinum that effaced the cardiac silhouette indicative of anterior mediastinal mass.
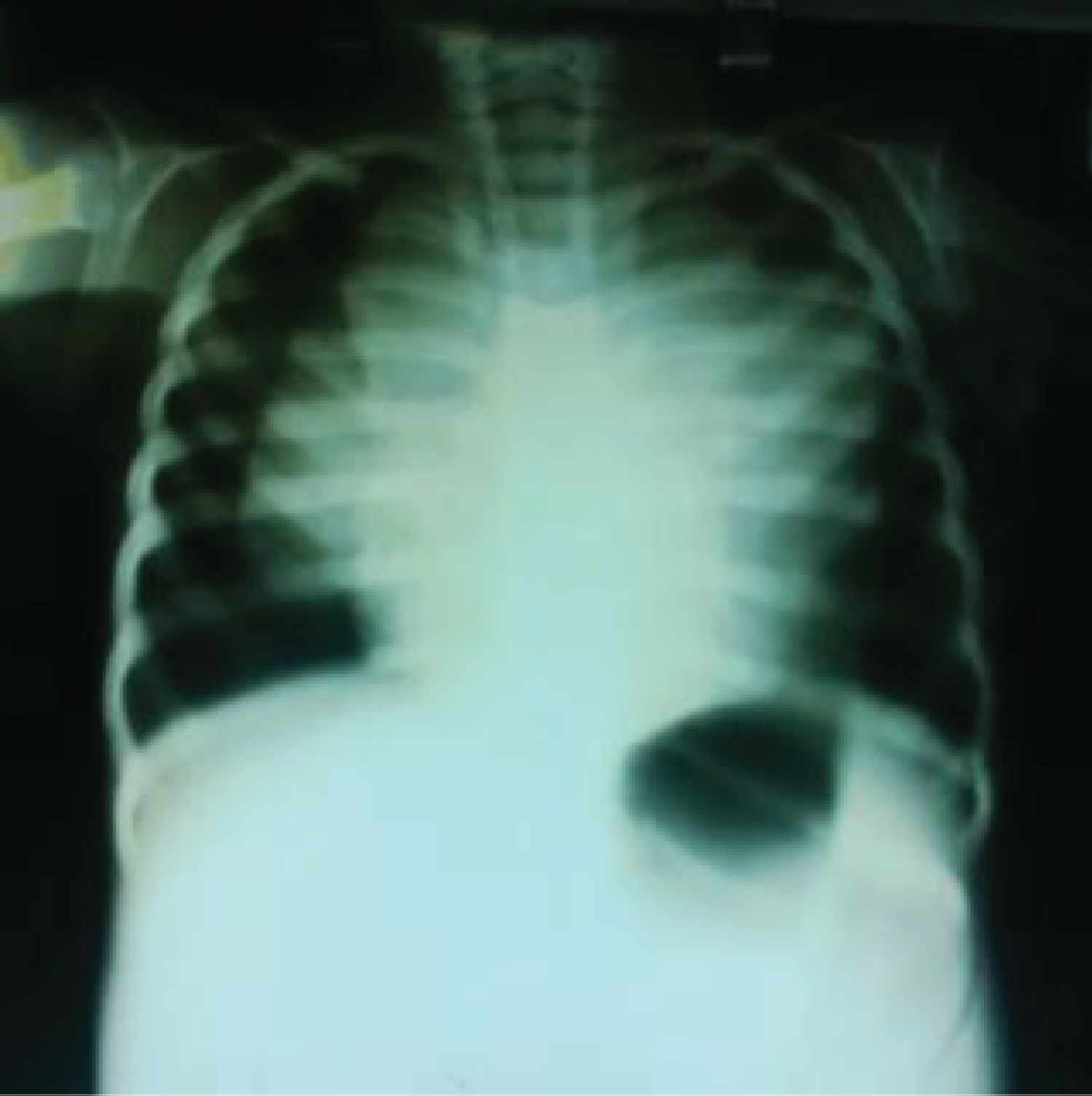 Figure 1: Shows anterior mediastinal mass, collapse of the right middle lung zone and collapse consolidation of the superior and inferior lingular segments of the left lung. There is associated bilateral pneumothorax.
View Figure 1
Figure 1: Shows anterior mediastinal mass, collapse of the right middle lung zone and collapse consolidation of the superior and inferior lingular segments of the left lung. There is associated bilateral pneumothorax.
View Figure 1
• Collapse of the right and middle lung lobes with pneumothorax in the right hemithorax was also noted.
• There was also collapse consolidation of the superior and inferior lingular segments of the left lung and ipsilateral peripheral pneumothorax.
Figure 1 shows anterior mediastinal mass, collapse of the right middle lung zone and collapse consolidation of the superior and inferior lingular segments of the left lung. There is associated bilateral pneumothorax.
• Lobulated large anterior mediastinal mass of soft tissue density almost encapsulating the heart anteriorly (Figure 2) with normal cardiac chambers suggestive of lymphadenopathy.
 Figure 2: Axial chest CT of the lower thoracic segment showing anterior mediastinal mass.
View Figure 2
Figure 2: Axial chest CT of the lower thoracic segment showing anterior mediastinal mass.
View Figure 2
• Hepatosplenomegaly of homogenous density devoid of focal lesions noted suggestive of lymphoproliferative disorder.
• Homogenously enlarged bilateral kidneys with good corticomedullary differentiation, preserved pelvicalyceal system and excretory function, No focal mass in pre/post contrast images. These ruled out Wilms' tumor (Figure 3).
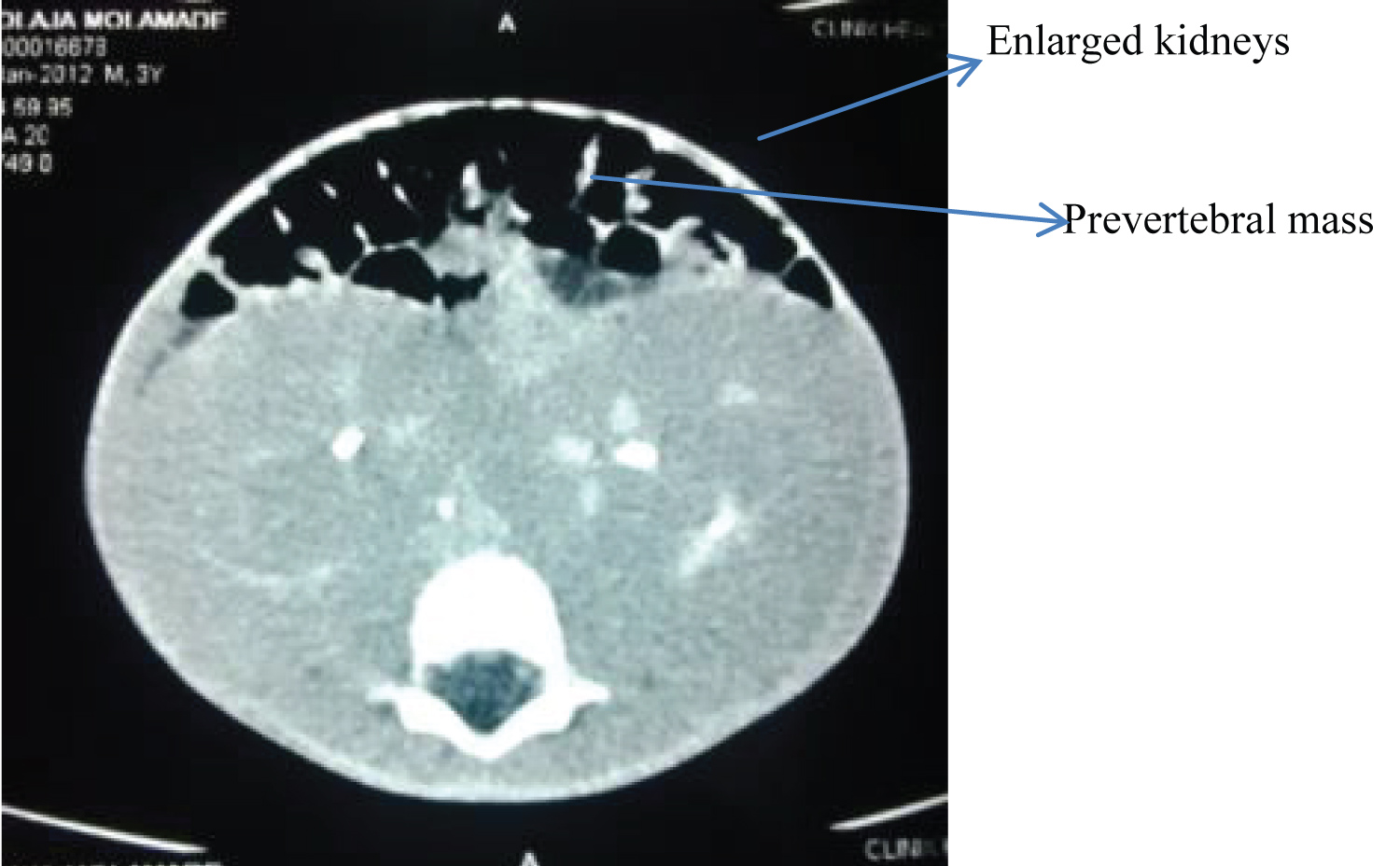 Figure 3: Post contrast axial abdominal CT showing bilateral enlarged functioning kidneys.
View Figure 3
Figure 3: Post contrast axial abdominal CT showing bilateral enlarged functioning kidneys.
View Figure 3
• A prevertebral mass of mixed density displacing the aorta anteriorly indicative of lymphadenopathy (Figure 4).
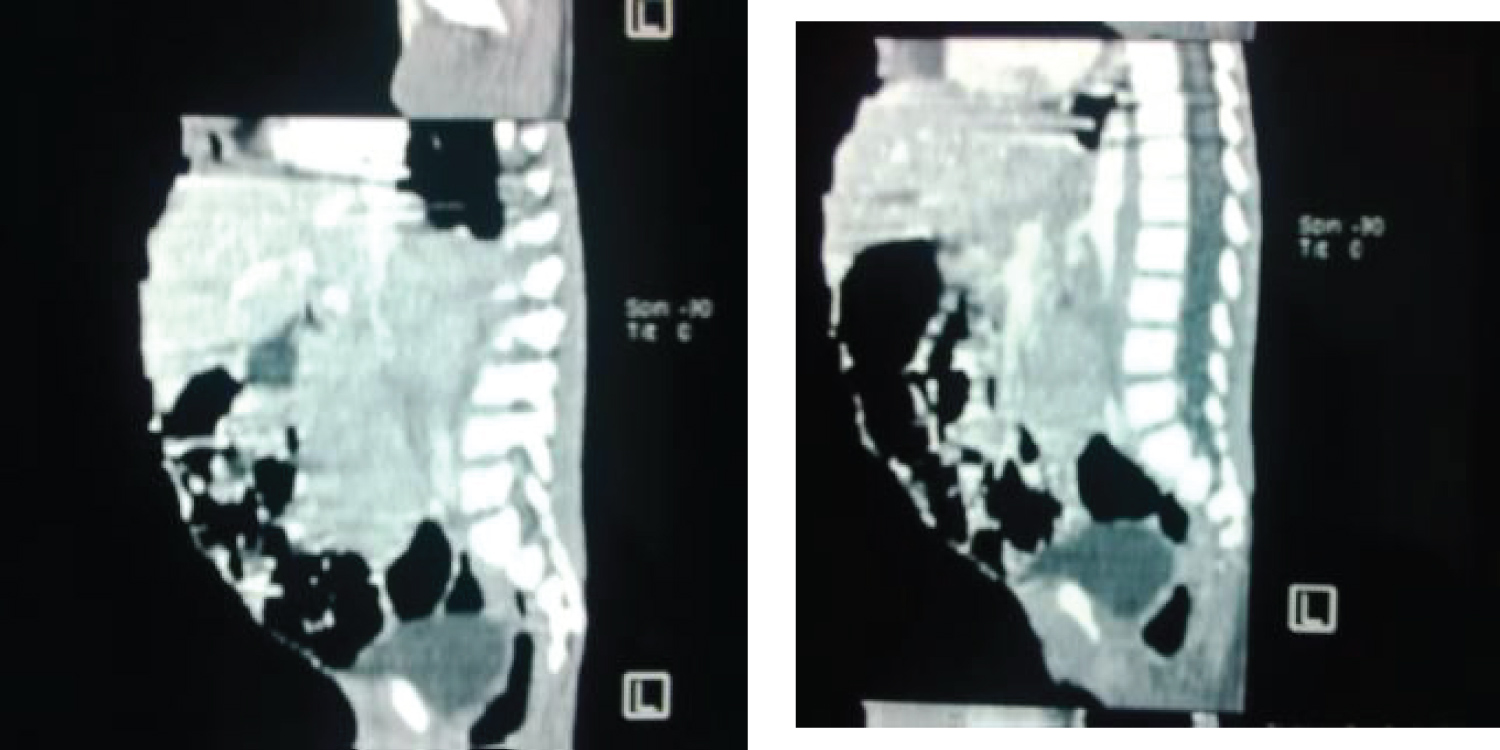 Figure 4: Reformatted sagittal view of CT abdomen showing a prevertebral mass.
View Figure 4
Figure 4: Reformatted sagittal view of CT abdomen showing a prevertebral mass.
View Figure 4
The electrolytes, urea and creatinine showed Urea 8.5 mmol/L (1.7-9.1 mmol/L), Sodium 144 mmol/L (135-145 mmol/L), Creatinine 1.4 μmol/L (53-115 μmol/L) and Uric acid-19.2 μmol/L (150-480 μmol/L). The hematology results revealed full blood count: Packed cell volume 22%, White blood cell count 12.3 × 103/L (4.0-11.0 × 103/L) and differential white cell count of 90% lymphoid cells and 05% neutrophils. Platelet count 52 × 109/L (150-400 × 109/L).
Peripheral Blood film showed anisopoikilocytosis with hypochromia among red cells. Moderate leukocytosis with predominance of homogenous small-sized lymphoid cells with scanty cytoplasm and open chromatin pattern. Platelets appeared reduced. The definitive diagnosis of acute lymphoblastic leukemia (L1 subtype) was made. However, immunohistochemistry could not be done due to financial constraints.
Chest intubation was done to relieve the pneumothorax with post intubation Chest radiographic evaluation. Patient was also optimized and subsequently commenced on the chemotherapy using intravenous Vincristine and oral Prednisolone as Pre-Induction stage while a combination of Methotrexate, Doxorubicin, L-Asparaginase and Prednisolone were used as the Induction Courses. The patient made significant recovery and the follow-up chest radiograph was devoid of any mediastinal masses or lesion.
Renal involvement could either be diffuse involvement or focal accumulation of leukemic cells, the latter being rare [7,8]. In the index patient a diffuse involvement was noted.
The Computed tomographic scan finding in the index patient was bilateral enlarged kidneys demonstrating symmetrical homogeneity with good corticomedullary differentiation. This is contrary to what obtains in Wilms, being often unilateral large heterogeneous soft tissue density renal masses, displacing adjacent structures with areas of calcification, fat density with patchy enhancement allowing for better delineation [6,9,10]. However, no focal renal contour concavity, parenchyma cupping called Claw sign, mass lesion common features in Wilms tumour were seen in the kidneys of this index case.
Both bilateral multiple and solitary unilateral and bilateral low attenuation masses, unilateral and bilateral nephromegaly presentation in various patterns had been described in a study of renal leukemia involvement in 12 patients [10]. The bilateral nephromegaly noted in this index case could be attributed to leukemic infiltration, simple hypertrophy or hyperplasia. This report shows the enlargement on CT as gross hypertrophy.
Renal leukemic infiltration occurs in the decreasing order of acute lymphoblastic leukemia and acute myeloblastic leukemia especially the M4 and M5 variants [2]. It does not commonly cause renal dysfunction. The few cases of renal failure being secondary to diffuse bilateral infiltration was reportedly noted with chronic lymphocytic leukemia and acute myeloblastic leukaemia [11-15]. Our patient had a preservation of renal function as evidenced in the electrolyte, urea and creatinine results as well as the abdominal CT scan.
Enlarged kidneys in acute lymphoblastic leukemia are considered an unfavorable prognostic sign [16]. Renal impairment could be from leukemic infiltrates, hyperuricemia, septicemia or haemorrhage [7]. Leukemic cells infiltrate the interstitial tissue and renal sinus with replacement of the tubules [7]. Since the nephrons are unaffected early in the course of the disease, the renal function usually remains preserved as observed in the index patient [2]. Nephromegaly without leukemic infiltration is said to be associated with hepatosplenomegaly [2]. No focal intrahepatic lesion was seen.
Patients with renal leukemic involvement present with extramedullary leukemic involvement of the mediastinum, pericardium, spleen, liver, pancreas, lymph nodes, spinal canal and the brain as found in the index patient [2]. Metastasis to the lungs is found in 85% of cases but this was not found in the index case probably because of early diagnosis.
An isolated cardiac involvement of B-cell acute lymphoblastic leukemia has also been noted with enhanced computed tomography. This is demonstrated as an infiltrative cardiac mass involving the right and left ventricular myocardium and pericardial recess [17]. The cardiac mass was dramatically smaller at after 2 weeks following the initiation of chemotherapy in the index patient.
Patients with acute lymphoblastic leukemia who have mediastinal masses at the time of diagnosis are said to have a poor prognosis [18]. However, other studies have established that when such is controlled for age, sex, white cell count, extent of lymphadenopathy, the presence of a mediastinal mass did not predict prognosis [19]. Others opined that if complete resolution of mediastinal tumor is not achieved, residual mediastinal tumor is of no prognostic relevance [20]. The index case at diagnosis had mediastinal mass that responded well to management and achieved complete resolution of the mediastinal mass on follow up chest radiographic imaging.
It is therefore expedient for attending physicians to have high index of suspicion in similar patient presentation. Such patients must be subjected to comprehensive radiologic imaging and hematologic investigations if accurate diagnosis is to be made early and treatment commenced on time. Undue morbidity and mortality are therefore prevented.
AYAO and AAP wrote the abstract and introduction, IOJ, AOF, OAO and EEE wrote the case report. AAP, IOJ, AOF evaluated the patient radiologically. OAT carried out the laboratory evaluation. AYAO, OAO, EEE worked on the discussion. All authors agreed on the radiological and hematologic findings, read the manuscript and approved it for publication.