Spontaneous coronary artery dissection (SCAD) is a rare, though, important cause of acute coronary syndrome that is more commonly seen in young and middle-aged women without risk factors for, or a history of, coronary artery disease. Acute embolization of left ventricular (LV) thrombus is an uncommon but known cause of acute limb ischemia. In this case, we present a rare case of SCAD that led to acute limb ischemia due to embolization of LV thrombus.
Spontaneous coronary artery dissection (SCAD) is a rare, though, important cause of acute coronary syndrome in young and middle-aged women with no or few traditional cardiovascular risk factors. Due to significant underdiagnosis to date, the true incidence and prevalence of SCAD in the general population remains unknown [1]. Its various clinical presentations such as chest discomfort in the context of an acute coronary syndrome (Mostly ST-elevation myocardial infarction), ventricular arrhythmias, cardiogenic shock, and sudden cardiac death, makes it difficult to make an accurate diagnosis [1,2].
Acute limb ischemia is mostly caused by thromboembolism, which will suddenly decrease the limb perfusion, thus, a potential threat to limb viability [3]. Whether from the heart or proximal aorta, the emboli travel through the vascular system and tends to obstruct at bifurcations, where the lumen of the arteries is narrowed.
SCAD is a very rare cause of LV thrombus, but still should be taken in consideration. In this case, we present a rare case of SCAD that led to acute limb ischemia.
A 53-year-old female with a past medical history of hypertension, hyperlipidemia, deep venous thrombosis (DVT) on anticoagulation therapy, COPD, and nicotine dependence presented with acute right lower extremity (RLE) pain. The patient mentioned that she had an acute chest pain one week prior to presentation, but the pain lasted for few hours so she didn't seek a medical care for.
In duplex ultrasound, monophasic low resistance waveform was revealed in the right external iliac and femoral arteries, suggesting the presence of RLE inflow disease (Figure 1).
 Figure 1: Duplex ultrasound showing the presence of monophasic low resistance waveform in the right external iliac and femoral arteries.
View Figure 1
Figure 1: Duplex ultrasound showing the presence of monophasic low resistance waveform in the right external iliac and femoral arteries.
View Figure 1
Echocardiogram demonstrated severe hypokinetic apex with protruding thrombus (Figure 2) and a moderate decrease in left ventricular ejection fraction. Left heart catheterization was performed, which demonstrated long, multiple radiolucent lumens lesion of the proximal to mid left anterior descending coronary artery (LAD) consistent with spontaneous coronary artery dissection (SCAD) type1 (Figure 3). Abdominal and bi-iliofemoral angiogram showed subtotaled occlusion of the right common iliac artery with well-formed thrombus (Figure 4).
 Figure 2: 2D Echocardiogram showing the presence of severe hypokinetic apex with protruding thrombus.
View Figure 2
Figure 2: 2D Echocardiogram showing the presence of severe hypokinetic apex with protruding thrombus.
View Figure 2
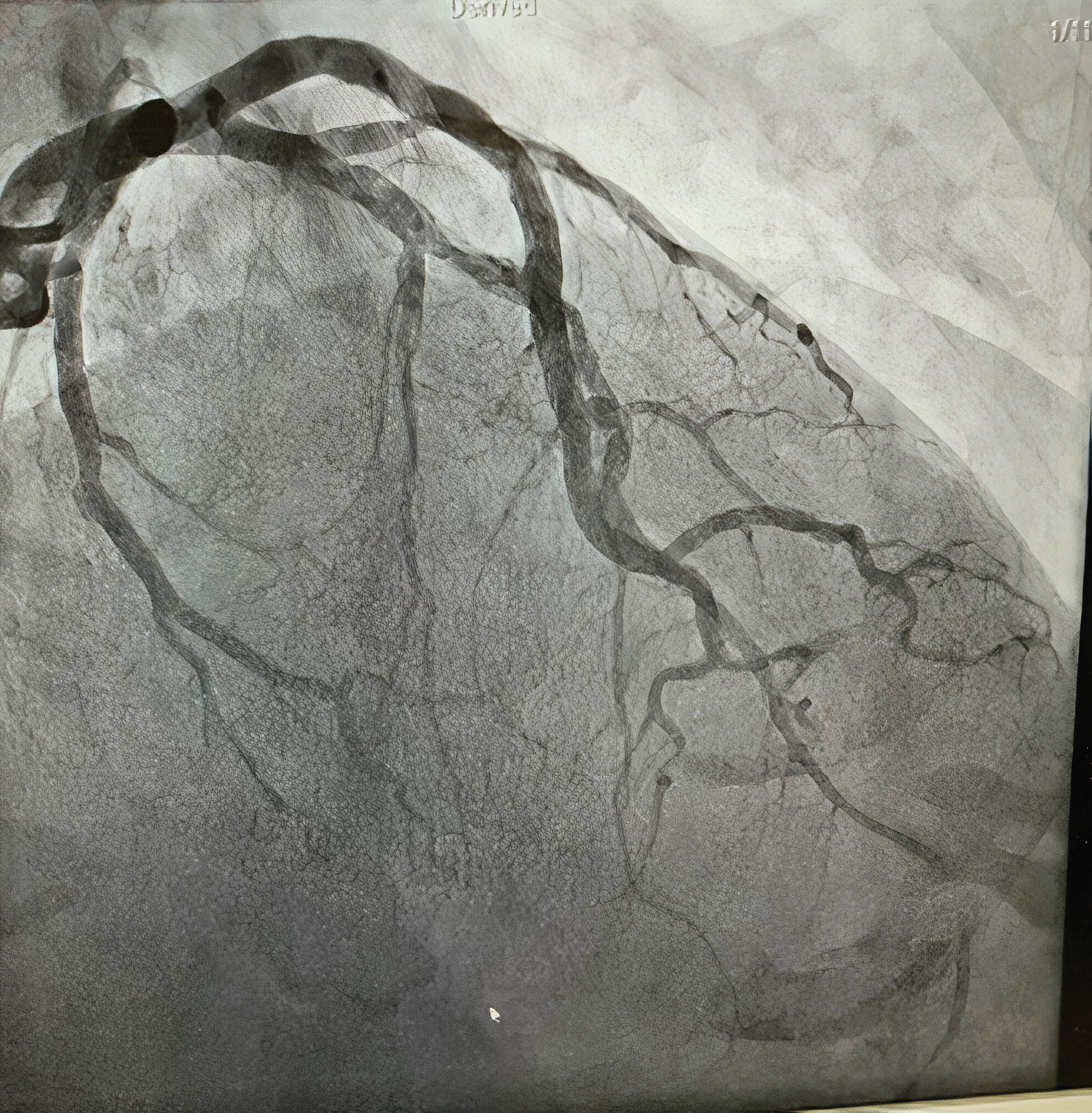 Figure 3: Coronary angiogram showing long, multiple radiolucent lumens lesion of the proximal to mid left anterior descending coronary artery (LAD).
View Figure 3
Figure 3: Coronary angiogram showing long, multiple radiolucent lumens lesion of the proximal to mid left anterior descending coronary artery (LAD).
View Figure 3
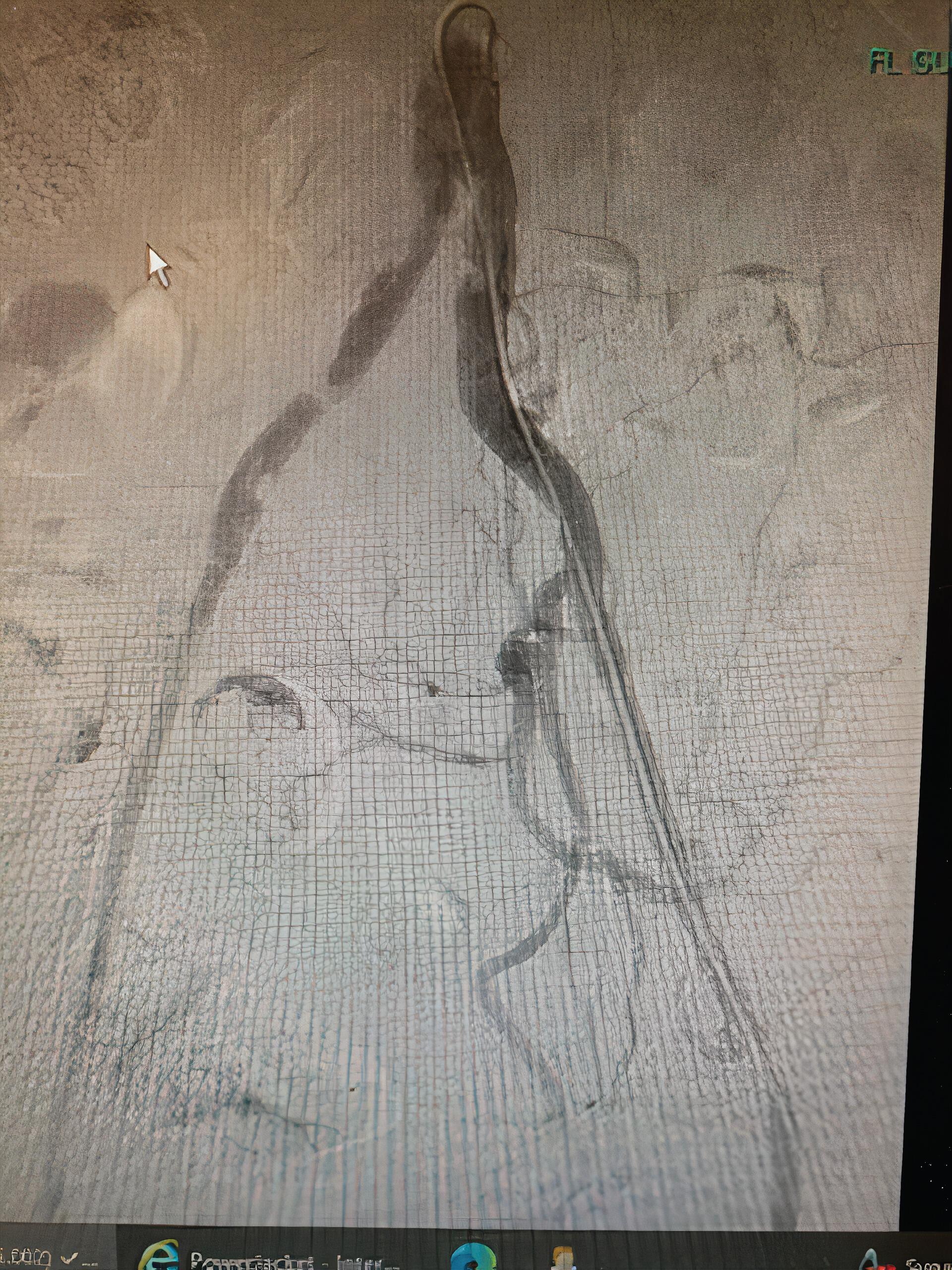 Figure 4: Abdominal and bi-iliofemoral angiogram showing subtotal occlusion of the right common iliac artery with well-formed thrombus.
View Figure 4
Figure 4: Abdominal and bi-iliofemoral angiogram showing subtotal occlusion of the right common iliac artery with well-formed thrombus.
View Figure 4
The cause of SCAD is usually multifactorial, but there are certain conditions or factors that have a strong association with SCAD [4,5]. For instance, fibromuscular dysplasia is the most frequently associated condition with SCAD, with prevalence of 25-86% reported in recent cohort studies [4]. Also, there is a strong association between SCAD and pregnancy, due to pregnancy hormonal variations that affect the composition of the vessel [6]. Other factors include multiparity (>/= 4 births), inherited arteriopathy and connective tissue disorders, exogenous hormones, and systemic inflammatory diseases [4].
Due to its similar presentation with atherosclerotic coronary artery disease (from mild chest pains to sudden cardiac death) [5], it is difficult to suspect the diagnosis of SCAD during acute coronary syndrome, thus, the true incidence and prevalence of SCAD in the general population remains unknown [1].
The primary role in the pathogenesis of SCAD is either due to a tear in the intima or spontaneous rupture of vasa vasorum which can lead to the formation of an intramural hematoma (IMH) within or between any of three layers (intima, media, or adventitia) of the coronary artery wall [1]. This hematoma can compress the true lumen, resulting in myocardial ischemia and infarction. In contrast to propagation in atherosclerotic vessels which is limited due to the presence of medial atrophy and scarring, SCAD most commonly affects normal vessels or vessels with weakened arterial wall, [7,8] resulting in extensive anterograde and retrograde propagation of dissections [1].
Thromboembolism from a cardiac source is mostly caused by atrial fibrillation [9], and rarely by left ventricular thrombus. The most common cause of left ventricular thrombus is dilated cardiomyopathy followed by apical hypokinesis post-myocardial infarction. The embolus tends to obstruct at bifurcations, where the lumen of the arteries is narrowed. In the lower extremity, this occurs most frequently at the common femoral artery and popliteal artery, whereas in the upper extremity, this mostly occurs at the origin of the profunda brachialis or brachial artery bifurcation [3]. Obstruction will cause a sudden limb ischemia that can result in amputation, unless appropriate treatment is given.
To classify SCAD, Saw, et al. proposed a classification system to better characterize the lesions [10]: Type 1 dissection: Pathognomonic contrast dye staining of arterial wall with multiple radiolucent lumen, with or without the presence of dye hang-up or slow contrast clearing (Figure 5).
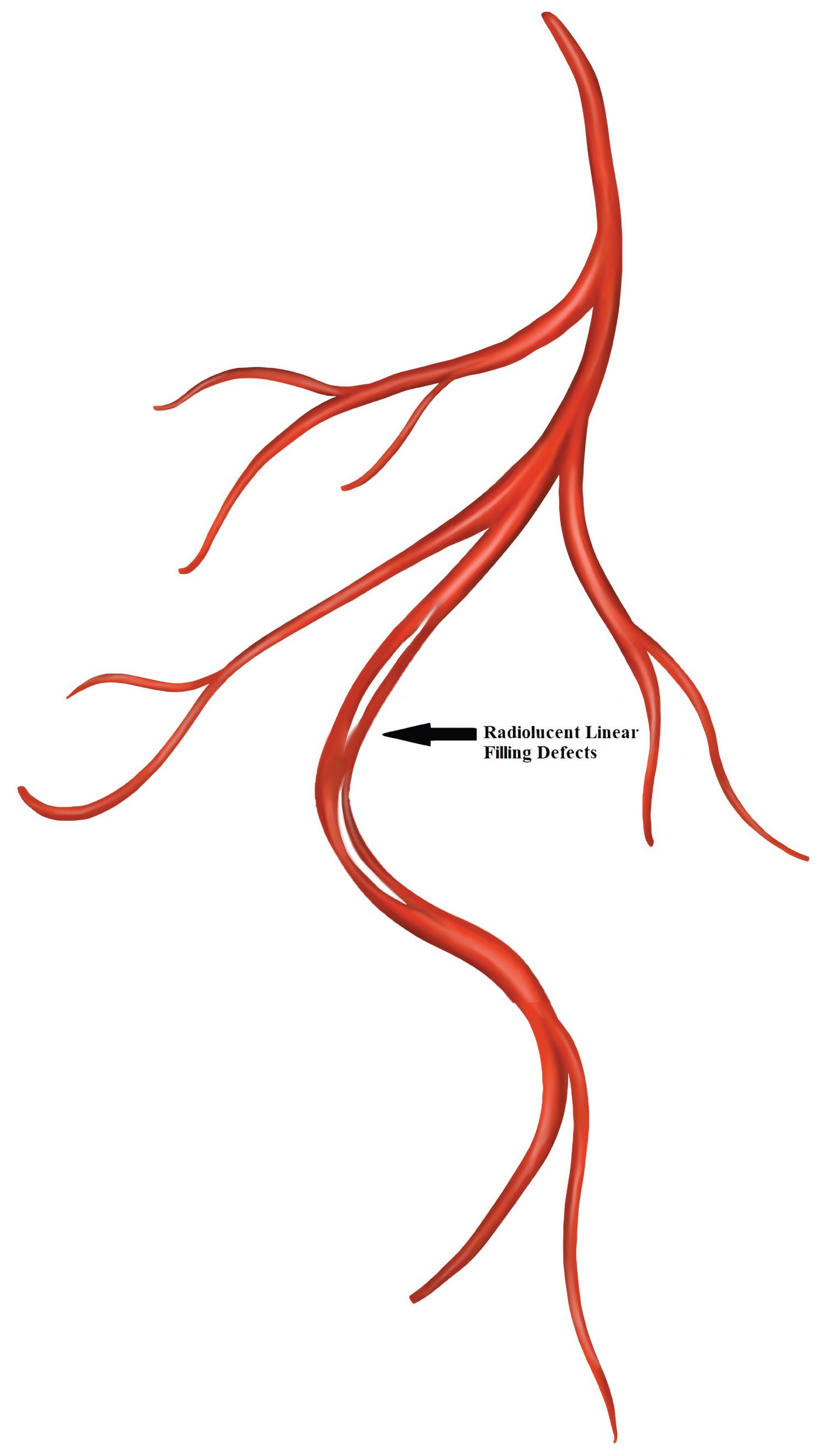 Figure 5: Spontaneous coronary artery dissection Type 1.
View Figure 5
Figure 5: Spontaneous coronary artery dissection Type 1.
View Figure 5
Type 2 dissection: Diffuse long and smooth stenosis of varying length and severity, with type 2A variant having diffuse narrowing bordered by normal artery segments proximally and distally to IMH, and type-2B having diffuse narrowing extending to the apical tip of the artery [11] (Figure 6).
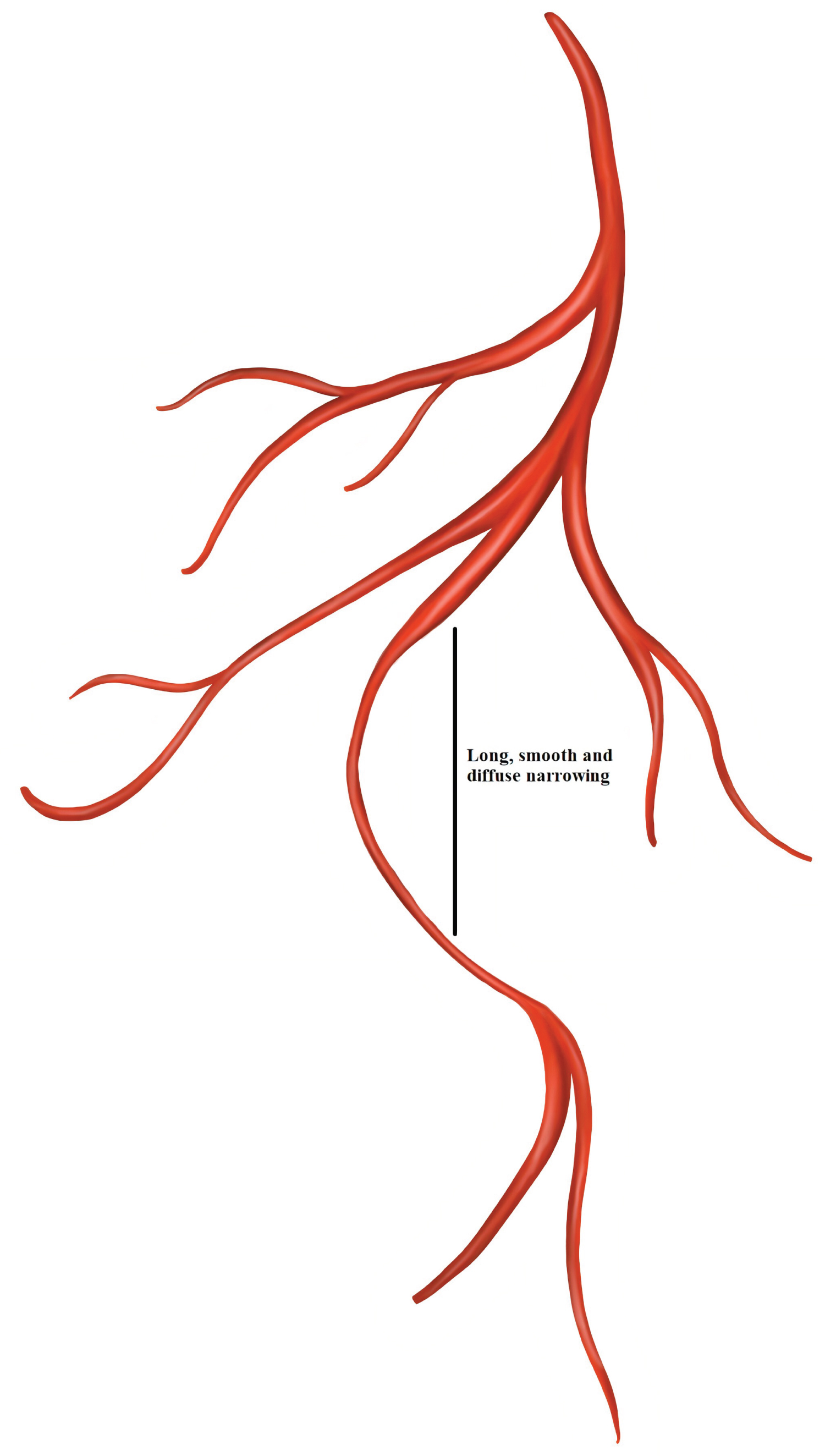 Figure 6: Spontaneous coronary artery dissection Type 2.
View Figure 6
Figure 6: Spontaneous coronary artery dissection Type 2.
View Figure 6
Type 3 dissection: Mimics atherosclerosis with focal or tubular stenosis and requiring optical coherence tomography (OCT) or intravascular ultrasound (IVUS) to diagnose IMH or double-lumen [11] (Figure 7).
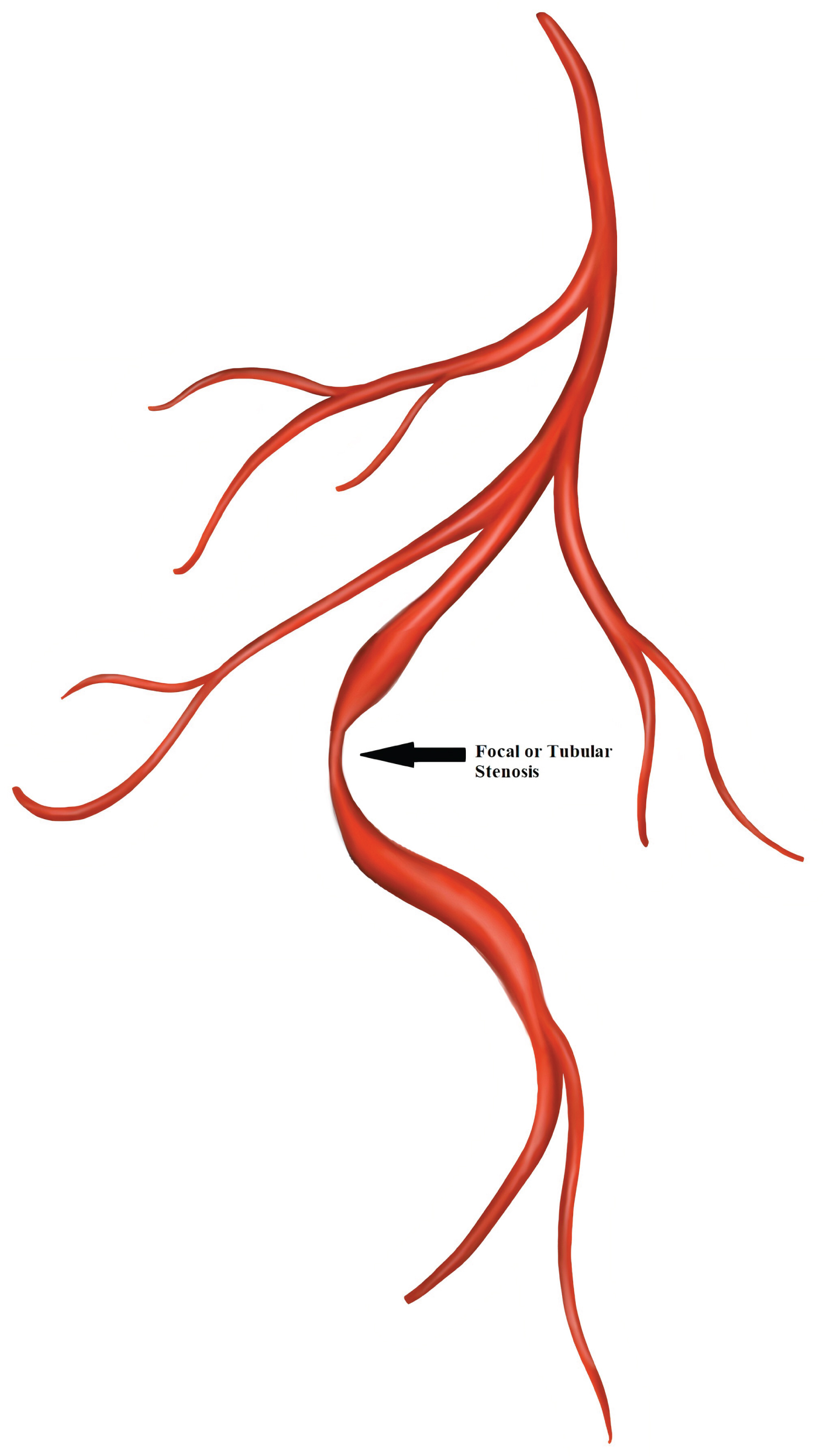 Figure 7: Spontaneous coronary artery dissection Type 3.
View Figure 7
Figure 7: Spontaneous coronary artery dissection Type 3.
View Figure 7
In the diagnosis of SCAD, coronary angiography is considered as the first-line diagnostic tool, and may be supplemented with intracoronary imaging if the angiographic diagnosis is unclear [1]. Current recommendations regarding SCAD management are based on expert opinions and observational series [1]. However, conservative therapy including aspirin and beta-blockers, is typically recommended since majority of SCAD cases will heal spontaneously after almost 35 days. Percutaneous coronary intervention (PCI) has an increased risk of in-stent restenosis and stent thrombosis after placement of long stents, and possible malapposed coronary stents after resorption of the IMH [1]. Thus, PCI should be reserved for high-risk patients with hemodynamic instability or ongoing ischemia. CABG has a high rate of graft failure, but still should be considered in patients with left main dissection or unsuitable anatomy for PCI [1]. Due to high recurrence rate of SCAD, prolonged inpatient monitoring and close follow-up is important [1].
Jun, et al. treated a 35-year-old man with no known prior medical history presented with right leg pain with stable vital signs [12]. The patient showed the symptoms of acute limb ischemia without chest symptoms during a hospital visit. Further investigations like CT angiography, electrocardiogram, and transthoracic echocardiogram made the diagnosis of SCAD and left ventricular thrombi that led to acute ileofemoral artery thromboembolism.
To the best of our knowledge, this is the second reported case highlighting the importance of considering acute limb ischemia as an unusual presentation of SCAD [12].
SCAD as a rare cause of acute coronary syndrome has been shown to be associated with various presentations. Certain presentations like acute limb ischemia are considered to be very rare. Our case highlights the need to consider acute limb ischemia as a presentation for SCAD.