Malaria is a major public health problem in the world, particularly in the developing countries where the disease is difficult to manage due to multi-drug resistant parasites. The study aimed to evaluate the antiplasmodial activity of Azadirachta indica in the treatment of malaria disease.
Fresh leaves of Azadirachta indica were air-dried at room temperature for six weeks and pulverized into powder using electric blender. Two hundred and fifty grams (250g) of the crushed powder was weighed with weighing balance and soaked in 1.2 liters of 96% ethanol for 72 hours. The ethanolic extract was obtained from the powder using rotary evaporator. Thirty-five albino mice housed under standard conditions were used for the in-vivo experiment. Ninety-six (96) hours (4 days) curative test in mice model was done to check the effect of the plant extract on the P. berghei in Swiss albino mice.
The result showed that the antiplasmodial activity of the A. indica varied significantly among the graded dosages (P ˂ 0.05). However, the highest antiplasmodial activity of the plant (100%) was observed at 600 mg/kg of the plant extract and 10 mg/kg of chloroquine. At these graded dosages all the erythrocytic stages of P. berghei were completely eliminated by the extract in the infected mice at day 2 of the plant extract and day 3 of the chloroquine treatment. Similarly, the percentage curative of the extract and the control showed that 100% curative was obtained in mice treated with 600 mg/kg of the ethanolic plant extract of A. indica for 24 hours and 10 mg/kg of the standard drug (chloroquine) for 48 hours. However, the least percentage curative (31.20%) was observed in infected mice treated with 800 mg/kg.
The results further showed that the parasitemia resulted in weight loss in all the infected mice except those treated with 800 mg/kg of A. indica and 10 mg/kg of chloroquine at 72 hours and 48 hours respectively. The findings of this research show that the ethanolic extract of A. indica has antiplasmodial property against P. berghei, hence, it can be used as herbal treatment for malaria infection.
Azadirachta indica, P. berghei, Parasitemia, Antiplasmodial, Albino mice
Malaria is an infection whose vector is the infected female Anopheles mosquito which spreads this disease to humans and other animals. Plasmodium protozoan is specifically the parasite that causes malaria [1]. Studies indicate that Plasmodium falciparum is responsible for the highest mortality rate in malaria cases whereas Plasmodium ovale, Plasmodium malariae and Plasmodium vivax, which are other types of the protozoa generally cause a milder form of malaria [2,3]. Another species (P. knowlesi and P. berghei), have been reported to rarely cause malaria disease in rodents but rarely in the human populace [1,4]. P. berghei and P. falciparum are having similarities in many biochemistry and genetic relationships [5] for this reason P. berghei has been a very useful model system in studying P. falciparum Infection with this protozoan is caused by the bite of mosquito of the genus Anopheles.
The clinical symptoms of human malaria caused by P. falciparum, P. ovale, P. vivax and P. malariae are similar to that of P. knowlesi infection in rodents. These clinical symptoms include fever, headache, anemia, splenomegaly, loss of appetite and loss of weight. However, the prevalence of P. falciparum malaria is higher in tropical climate especially in Africa countries. It was reported by WHO [6] that approximately 90% malaria morbidity and mortality occurred in African countries. This implies that the African continent is the most endemic continent in the world.
Herbal medicine is still the mainstay of about 75-80% of the world population, mainly in the developing countries for primary health care [5]. This is primarily because of the general belief that herbal drugs are without any side effects besides being cheap and locally available [7]. According to the World Health Organization (WHO), the use of herbal remedies throughout the world exceeds that of the conventional drugs by two to three times. The use of plants for healing purposes predates human history and forms the origin of most modern medicine. Large number of plant species has been identified as anti-malaria medicinal plant, the first antimalarial drug used in the occident was extracted from the bark of the Cinchona (Rubiaceae) species, the alkaloid quinine which is still largely used. Infusion of the plant bark has been used to treat human malaria [8] and several years later quinine was isolated [9] and thus becoming the oldest and the most important antimalarial drug.
Azadirachta indica is used as traditional herbal medicine and the extracts contain various antimalarial substances which makes it difficult for the parasite to develop resistance against a single component. The leaf extract of A. indica has been prescribed orally for the treatment of malaria by Indian ayurvedic practitioners for centuries [10]. The leaf extract contains sterols, limonoids, flavonoids, glycosides and coumarins [11]. The leaf extract of A. indica has schizontocidal and gametocidal effects [12]. Thus, the combination of two gametocidal agents e.g. artesunic acid and neem extract would be expected to further decrease transmission of malaria Plasmodium. Therefore, this study aimed to evaluate the efficacy of Azadirachta indica in the treatment of Plasmodium berghei using as Swiss albino mice as test model.
Fresh leaves of Azadirachta indica free of insects were collected from the environment of Federal University of Technology Akure, Ondo State, Nigeria. The leaves were identified by a plant taxonomist in the department of Crop, Soil and Production (CSP) of Federal University of Technology Akure.
The leaves were thoroughly washed and air dried at room temperature (28 ℃ ± 2 ℃). Subsequently, the dried leaves were crushed into coarse powder using electric blender. Two hundred and fifty grams (250g) of the crushed powder were weighed on weighing balance and soaked in 1.2 liters of 96% ethanol for 72 hours. The preparation was stirred for 1 hour at every 24 hours. It was then decanted and filtered with muslin cloth. The extracts obtained was concentrated using a rotary evaporator. The crude ethanolic extract was then concentrated in a vacuum oven to remove all traces of solvent. The extract was refrigerated at 4 ℃ prior to use.
Thirty five (35) mice with average weight range of 18-23g were purchased from the Institute of Advanced Medical Research and Training (IAMRAT), College of Medicine, University of Ibadan. The animals were enclosed in iron cages in the animal house of IAMRAT and were allowed to acclimatize for 4 days at room temperature (28 ℃ ± 2 ℃) for 12 hours dark/night periodicity. The caged animals were fed with commercial chow (purchased from Cap Feeds Ibadan, Nigeria) and water ad libitum for four days.
The malaria parasite (Plasmodium berghei (NK65) used for this research was obtained from the Department of Parasitology, Institute of Advanced Medical Research and Training (IAMRAT), College of Medicine, University of Ibadan, Nigeria. A Swiss albino (donor) mouse intraperitoneally injected with P. berghei and in which the parasite has attained a 40 -50% parasitemia serve as the donor. The mouse was sacrificed and 0.1 ml of acid citrate dextrose (ACD) was drawn into the syringe before blood was withdrawn from the heart of the donor mouse by cardiac puncture. The blood was then diluted with isotonic saline to make inoculum for infecting the experimental mice. The experimental mice were inoculated with 0.2 ml of diluted parasitized red blood cells specimen (containing about 1 × 107 parasitized cell).
The curative activity (Rane's test) of the plant extracts against established sensitive Plasmodium berghei NK65 infection in mice was determined using the method described by Iyiola, et al. [13]. Thirty- five mice (weighing 18-23g) were divided randomly into seven groups, each group containing five mice each. P. berghei was injected intraperitoneally into 25 mice in group one-five on day one. One sixty-eight hours later (day seven), the mice in group one-three were administered through oral cannula with the 400, 600 and 800 mg/kg of the plant extracts respectively as described in Table 1 while the mice in group four (standard control group) were administered with 0.2 ml 0f 10 mg/kg chloroquine phosphate solution also on day seven. Mice in groups six and seven were not infected, meanwhile, group six (plant extract control group) were administered with 800 mg/kg of the plant extract on day seven. Meanwhile no treatment was administered to the mice in group seven (positive control). Monitoring of the parasitemia began 24 hours after parasite inoculation.
Table 1: Experimental plan for control and test animals. View Table 1
Blood were collected from each of the experimental mice through a tail cut from which both thick and thin smear were prepared. The smear was allowed to dry and the thin smear was fixed with methanol, both thick and thin films were then stained with 10% Giemsa stain for 15 minutes. The slides were observed under oil immersion objective lens of the compound microscope at magnification of × 100. Each slide was observed and both the parasitized red blood cells (RBCs) and unparasitized RBCs were recorded. The data obtained were used to determine percentage parasitemia using the method described by Hilou, et al. [14] percentage parasitemia and percentage curative were calculated using the formula below:
The results obtained were analyzed for statistical significance using one-way ANOVA. The mean and the level of significance for the differences between the means of the data obtained were computed using Duncan's New Multiple Range Test (DNMRT) at P ˂ 0.05. All statistical analyses were done using Statistical Package for Social Sciences (SPSS) version 20.0.
The results presented in Table 2 showed significant difference in the antiplasmodial activities of the plant extract at graded dosages of 400, 600 and 800 mg/kg body weight and the controls at 10 mg/kg (standard control) and negative control group (infected untreated). The results showed that highest percentage parasitemia (30.34%) was observed in mice treated with 400 mg/kg for 4 consecutive days while no parasitemia (0%) was observed in mice treated with 600 mg/kg of A. indica extract for two days and 10 mg/kg of chloroquine for three days respectively. However, relapse of the P. berghei infection was noted on day three in mice treated with 600 mg/kg of the plant extract. The percentage parasitemia in the mice increased from 0% in day two to 3.44% in day three. It was further observed that only 10 mg/kg of chloroquine was able to eliminate all the erythrocytic stages of P. berghei in the infected mice without relapse. In addition, the percentage parasitemia of the mice infected but untreated increased significantly from day one to five.
Table 2: Percentage parasitemia of the mice in each treatment group. View Table 2
The results presented in Table 3 showed the effect of the parasite on the weight of the mice. It was observed that there was weight loss all through the periods of treatment except in mice treated with 800 mg/kg of the plant extract for 72 hours. However, the highest percentage weight loss (16.28%) was observed in mice treated with 400 mg/kg of A. indica while no percentage weight loss (0%) was observed in mice treated with 800 mg/kg for 72 hours respectively. The result further showed that weight loss among the groups was significant (P < 0.05). However, the percentage parasitemia does not correlate with weight loss (P > 0.05). This is because the mice treated with 400 mg/kg A. indica extract for 72 hour, showing highest percentage parasitemia (30.34%) only recorded 4.9% percentage weight loss while mice treated with the same dose of the extract for 48 hours showing 1.26% percentage parasitemia were observed to have highest percentage weight loss (16.26%). However, mice with similar parasitemia (1.22%) but treated with 800 mg/kg for 72 hours showed no percentage weight loss.
Table 3: Average Percentage Weight Loss in Each Treatment Group. View Table 3
Figure 1 showed the percentage curative/suppression of the administered treatment. For 400 mg/kg, no suppression was recorded for all the post-treatment days, hence the blue bar did not occur in the chart. For 600 mg/kg, there was 100% elimination at 24 hours. For 800 mg/kg, 31.20% suppression was recorded at 24 hours and 54.14% suppression recorded at 72 hours. For chloroquine, 93.18% suppression was recorded at 24 hours while there was 100% elimination was noted in subsequent post-treatment days.
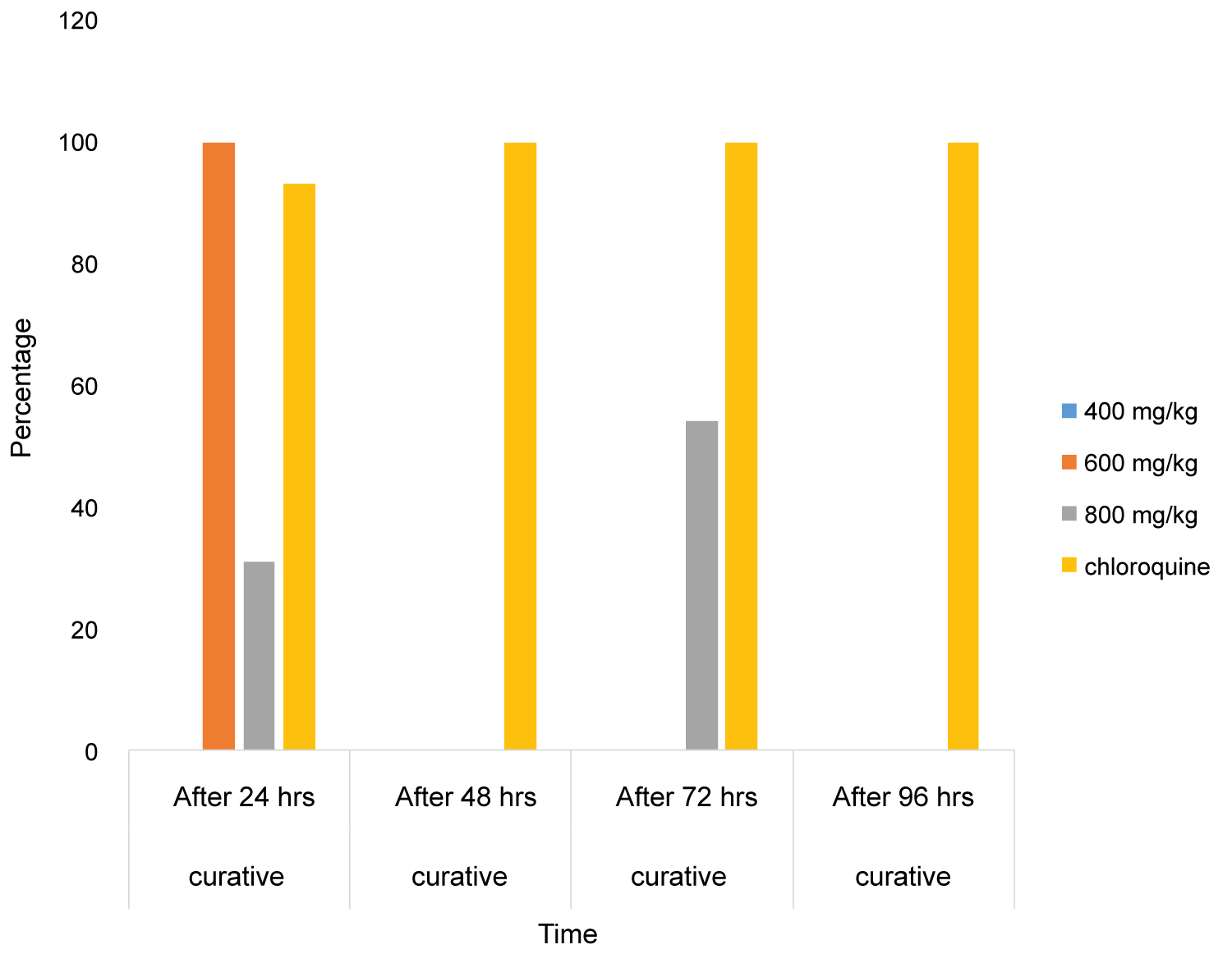 Figure 1: Percentage curative of A. indica at 24 hours interval.
View Figure 1
Figure 1: Percentage curative of A. indica at 24 hours interval.
View Figure 1
Plants are very rich source of antimalarial drugs in use today. This study evaluated the antiplasmodial activity of A. indica in respect to parasitemia and weight of the mice used. The results showed that parasitemia (parasitic load) was completely eliminated at 600 mg/kg of A. indica and 10 mg/kg of chloroquine at day two and day three respectively. This result showed that the plant extract (A. indica) could only achieve 100% curative at high dosage of 600 mg/kg instead of lower dosage. It also means that if lower doses (100-400 mg/kg) of the plant extract were considered for this research, the effectiveness of the plant extract will be highly insignificant. The result also showed that A. indica achieved 100% curative at 24 hours post-treatment while chloroquine the standard drug used achieved 100% at 48 hours post treatment.
However, recrudescence of the disease was observed at all dosages of the plant extract except the standard control (mice treated with chloroquine) and mice treated with 600 mg/kg of the A. indica extract. Relapse was noted in mice treated with 600 mg/kg because all the parasites (P. berghei) were completely eliminated from the mice blood on day two (48 hours) post-treatment, however, merozoites which were likely to emerge from the liver cells later invaded the red blood cell on day three (72 hours) post-treatment to re-establish infection [15]. The recrudescence was as a consequence of submicroscopic parasitemia and delayed schizogony [15]. The parasite become insurgent and increase in the infected treated mice while relapse occurs as a result of new invasion of the red blood cells by the liver hypnozoites after the initial erythrocytic stages of the parasite have been completely eliminated [15]. Recrudescence was also noted in infected mice treated with 400 mg/kg at day 4 and 800 mg/kg at day five. Meanwhile, when the percentage parasitemia of the infected and treated mice were compared with the negative control (infected untreated mice). It was observed that the percentage parasitemia steadily increased from day one to five. This might be that the mice innate immunity might not be strong enough to suppress the parasite. The parasite suppression obtained from A. indica extract agrees with the findings of Momoh, et al. [16] who observed significant suppression of parasitemia of 47.80%, 50.96% and 52.30% in infected mice treated with 200, 400 and 800 mg/kg of methanolic leaf extract of A. indica. The findings of [17,18]. Also concurred with the present study. The authors reported that the methanolic extract of A. indica is effective in the treatment of malaria. The antiplasmodial activity of A. indica has been attributed to the presence of high antioxidant in the extract [19].
It was further observed from this research that most of the infected mice lost weight when the initial weight before treatment was compared with weight after treatment. The weight loss was noted in all the treatment dosages except 800 mg/kg for 72 hours where the initial weights of the mice were maintained. This result is in variance with the findings of [20] who reported that active anti-malarial agent has been expected to protect weight loss associated with malaria infection. The discrepancy might be due to imbalance of the protective effect of the extract and the cumulative pathophysiologic changes associated with the malaria infection.
The percentage curative result obtained from this study further validated the antimalarial activity of the ethanolic extract of A. indica. The results showed that only 10 mg/kg chloroquine and 600 mg/kg of A. indica achieved 100 percenatge curative while the curatives of the other dosages were less than 100%. This shows that the optimal antiplasmodial activity of A. indica was achieved at 600 mg/kg for 24 hours.
The in-vivo test showed that Azadirachta indica showed significant parasite suppression in the infected mice. Chloroquine the standard drug was noted to completely eliminate the parasite (P. berghei) at 10 mg/kg body weight compared to the plant extract which the percentage curative varies with doses. The optimal antiplasmodial activity of A. indica extract was noted at 600 mg/kg. 800 mg/kg of the plant extract could not afford 100% suppression, however minimal percentage parasitemia was maintained in the mice treated with the dose. The high dose of the plant extract might have produced recrudescence in some of the parasites, hence the drug might no longer be effective on them. This suggests that A. indica should be administered at 600 mg/kg to avoid recrudescence in parasite. However, further research such as toxicological and hematological tests should be carried out on A. indica to determine its safety to consumers. It was further observed that infection with P. berghei produced significant weight loss in mice.
The authors are grateful to Mrs. Thomas of the Institute of Advanced Medical Research and Training (IAMRAT) University College Hospital, University of Ibadan for providing technical support during the laboratory work. The technical support provided by Mrs. E.T. Ojo during the plant extraction using Soxhlet machine is also appreciated.
The authors have no competing interests.
The authors received no funding for this research.
The ethic and consent to use mice for this research was waived by the Institute of Advanced Medical Research and Training, College of Medicine, University of Ibadan. However, the research was conducted in the Institute laboratory where the methodology was closely monitored by the Institute.