Seventy-one patients (71) with chronic kidney disease (CKD) (21 men and 18 females non-treated with hemodialysis and 19 men, respectively 13 females treated with regularly repeated hemodialysis) are investigated. The mean clearance of creatinin in investigated group were 0.17 mL/sec (10.2 mL/min). The average age of non-dialyzed group were 40.3 years, and in the dialyzed patients -38.6 years.
The transfixant iliac bone biopsy is commit in two patient's group (non-demineralized sections; Goldner coloration). The electronic pencil with "on line" computer connection ("Apple") is utilized for measurement of trabecular bon mass and the volume of howship lacunes (HL). The number and volume of HL is correlated with the blood concentration of c-iPTH and CT. The mean value of the proportion: "volume of the HL/trabecular bone mass "(%) in non-dialyzed patients is 0.01%, and in dialyzed group = 0.008% (the difference is statistically non significant). The correlation between the immunoreactive "C" peptide of parathormon (c-iPTH) and HL's volume in non-dialyzed patient's group is non-significant too (r = NS), but in the dialyzed subjects, it is negative (r = -0.31).
However, the relationship between the plasma concentration of calcitonin (CT) and the volume of HL is reciprocal in two groups of investigated patients (r1 = -0.51; r2 = -0.10).
Chronic kidney disease, Calcitonin, Parathyroid hormone, Bone disease, Bone biopsy
Chronic kidney disease (CKD) exerts nosocomial damage of the skeletal and muscle system in CKD patients (the so called uremic/renal osteodystrophy = ROD). The elucidation of the pathogenesis of renal metabolic bone disease (ROD) enables medicamentos, dialysis and/or surgical interventions, thus providing profilaxis and better treatment of this severe complication of chronic uremia [1].
The aim of the study is to correlate the relationship of the "skeletal" proteohormones (parathormon = PTH, calcitonin = CT) and their influence on the skeletal "how-ship" destruction (Howship Lacunes = HL) in a given critical moment when bone biopsy and blood chemistry for determining serum concentration of c-iPTH and CT are done in patients treated in the Department of nephrology (Faculty of Medicine, University "St. Cyrill and Methodius, Skopje, Macedonia).
39 CKD patients not treated with dialysis (21 males and 18 females, mean age 40.3, range 18-60 years) and 32 hemodialysis patients (19 males and 13 females, mean time of dialysis 22.3 months, range 4-108 months, mean age 38.6, range 21-55 years) have been investigated. Both groups being age and sex matched and had creatinin clearance below 0.17 mL/sec (10.2 mL/min) with residual urine output less than 0.006 mL/sec (500 mL/24 h).
Patients non-treated with hemodialysis had not been previously taking "specific" therapy (inorganic phosphate chelators, calcium salts, supranormal doses of vitamin D or its active metabolites). Hemodialysis patients (3 x 4 h/weekly) had been regularly treated with aluminium hydroxide (2.4-4.8 g/day) and calcium (Ca-Sandoz, 1 g/day and/or CaCO3 up to 6 g/day).
The selected hemodialysis patients were not taking any other medications (such as vitamin D3). Blood for estimating the serum concentration of c-iPTH and CT has been taken at 8 o'clock in the morning (5 mL). RIA method was used with commercial kits (PTH-RIA-100/EO 182-1, IRE and Byk-Mallinckrodt, Belgium).
Immediately thereafter, transfixant iliac bone biopsy has been performed by a modified Bordier's needle (inner diameter = 5 mm). The obtained bone cylinders, after fixation with 96% ethanol, are impregnated in 1% methyl methacrylate to preserve the nascent ossal structure during the tissue cutting (with Jung-K mycrotom). The bone cuts (thickness between 4-5 μm), coloured following Goldner, are utilized for numbering and measuring of the volume of the sc "howship" lacunas (HL).The measurements of the mentioned histomorphologic parameters are done on optical microscope (Zeiss, N0 473356-990, objective: 40x, transmissional increase 1.25, ocular planimeter with microscopic scale-10x) with total enlargement of 500x. The surface of Zeiss-1 integrator is defined by ocular micrometer (calibration is done using Zeiss's ethalone 5 + 100/100). The numbering of HL/mm2 is pursuit with rectilinear movement of the integrator over the all surface of the ossal cylinder in the visual field.
The absolute volume of the trabecular bone mass (TBM) and HL has been automatically measured by electronic pencil with "on line" computer connection ("Apple"). The results of the volume of HL were correlated with the total volume of TBM from each patient separately in both groups. The volume of TBM and HL is calculated by the Deless equation (absolute area of TBM and HL multiplied by correction factor: 4/3.14.. = 1.273..). Finally, the mean volume of HL was estimated in both groups and the significance of their differences was correlated.
Each separate value for plasma immunoreactivity of c-PTH and CT was linearly correlated with the total volume of HL (a sum of volumes of all the present HL in a given sample). The result of the number and volume of HL are presented as a mean of the means values of each patient separately.
The results are presented in the tables.
The Table 1 presents the mean serum concentration for c-iPTH and CT in two investigated groups of patients, the mutual interrelationship (CT ↔ c-iPTH; coefficient of correlation (r)) and the significance of statistical difference for the obtained values in two patient's series.
Table 1: The mean serum values (X ± SEM, SEM = Standard Error Mean) for c-iPTH and CT in non/hemodyalized patients. View Table 1
Analysing Table 2, one could note that the absolute number of HL in non-dialysis patients is significantly higher in females than males (the difference in means is statistically significant, Z = 2.02 SEM). The ratio is reverse in dialysis patients, but statistically not significant (Z = 1.00 SEM). Correlating means of the same variable in non-dialysis and dialysis patients (the whole group, females and males), one coud note significant difference only in the mean absolute number of HL in females (Z = 2.97 SEM).
Table 2: The mean values (X ± SEM) for absolute number of HL, number of HL/μm3, volume of HL (μm3) and percent of HL from total, absolute trabecular bone mass (non dialysis vs. dialysis subjects). View Table 2
The number of HL per volume unit of TBM (HL/μm3) shows a similar distribution as the previous variable. In the group of non-dialysis patients, females have higher number of HL/μm3 in the TBM (Z = 4.80 SEM) than males. Non-dialysis females have also higher number of HL/μm3 in TBM, than females on maintenance hemodialysis (Z = 3.14 SEM). The rest of the groups and subgroups don't show significant difference in the means.
The volume of HL in dialysis patients is significantly smaller than in the group of non-dialysis patients (Z = 2.07 SEM). The same relationship is in the female group (Z = 2.16 SEM) whereas for the male group there is no significant difference in the volume of HL. The ratio of the same variable in the non-dialysis vs. dialysis males and females is statistically insignificant (Z = NS).
The difference in proportions (Zp1-p2) of participating volume of HL in TBM in non-dialysis and dialysis group of patients, though significant at first sight (1.25 × bigger proportions in non-dialysis patients) is statistically insignificant.
Table 3 shows significant positive correlation (r = 0.65) between c-iPTH and the number of HL/μm3 trabecular bone in non-dialysis patients. In dialysis patients the relationship between the c-iPTH serum level and distribution of HL is paradoxically negative (r = -0.68). CT did not significantly correlared with this micromorphometrical indicator in both groups of patients (dialyzed and non-dialyzed).
Table 3: Correlation of c-iPTH and CT with the number (HL/μm3) and volume of HL (in μm3). View Table 3
The mean volume of HL does not show significant relationship with c-iPTH in non-dialysis patients. Whereas dialysis patients show negative, moderately significant correlation (r = -0.31). Higher plasma concentrations of calcitonin are associated with lower volume of HL (r = -0.51) in non-dialysis uremic patients, whereas for dialysis patients there is practically non significant influence of calcitonin on the volume of HL (r = -0.10).
Previous date from literature show that CT ant PTH are physiological antagonists regarding the plasma regulation of divalent ions, particularly calcium and magnesium [2].
The antagonism is specially emphasized in the process of skeletal remodeling. PTH stimulates the growth and activity of both osteoblasts and osteoclasts (multinuclearity, increasing the number of cytoplasmatic podocytes..), whereas CT decreases the number and activity of osteoclasts (thus promoting hypocalcemic action) [3]. HLs are a form of local bone destruction that appears by action of active, polynuclear osteoclasts. Their magnitude and number are proportional to the activity of pro-destructive factors (particularly PTH), and inversely proportional to appositional factors of the skeleton (depositing osteoid and its mineralization) (Figure 1).
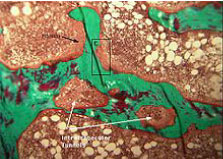 Figure 1: The fibrocystic osteitis with HL lacunae.
View Figure 1
Figure 1: The fibrocystic osteitis with HL lacunae.
View Figure 1
As in forming of an HL one or more osteoclasts may be implicated, the final effect of their acting results in a forming of a bigger (or smaller) number of HL, with bigger or smaller volume.
The volume of HL (margin trabecular, intra-trabecular) is a true expression of bone resorption and represents the fibrocystic osteitis [4].
Through bone biopsy and estimating plasma levels of c-iPTH and CT are performed simultaneously, thus having one moment of the natural evolution of renal osteodystrophy/metabolic osteopathy (non-dialysis patients) and it's developing during dialysis therapy, we don't have any insight in the processes of skeletal remodeling before and after the freezing. Thus, having in mind the theoretical assumptions, efforts are made to correlate the levels of c-iPTH and CT with the extent of lacunar HL-resorption along the margin and inner space of the trabecular bone in clinical conditions. We noted moderately high positive correlation (r = 0.65) between the serum PTH level and the number of HL per unit volume of TBM (in μm3) in non-dialysis patients. In patients on maintenance hemodialysis, the correlation is totally adverse (r = -0.68). This indicates reduced or absent destructive effect of PTH on bones, at least when regarding forming of HL-areas. This result implicates better skeleton reactivity to the procalcemic action of PTH in conditions of maintenance dialysis (more active periosteocytic osteolysis vs. HL formation).
The level of CT insignificantly correlates to the number of HL/μm3 trabecular bone in both groups of patients.
The volume of HL (in μm3) did not show significant correlation with PTH level in patients not treated with maintenance hemodialysis and CT level in dialysis patients, whereas the dialysis patients showed paradoxically inverse moderate correlation (r = -0.31) for the relation: PTH ↔ volume of HL, and negative (expected) moderately intense association for the relationship: Volume of HL ↔ CT-level (r = -0.51) in non-dialysis patients.
From the above mentioned correlations it can be concluded that PTH and CT do not exert the expected effects in dialysis patients (don't exert progressive increase/decrease in the number and volume of HL).
In non-dialysis patients PTH would force forming of HL, but would not influence their increase. It seems as it exert primary osteoagression ("primum movens"), but allows other factors to influence the further growth of HL (increased bone resorption out of control).
CT inversely, does not affect the number of HL per unit trabecular bone volume, but decreases their growth (decreases the activity of osteoclasts; makes conversion of polynuclear in oligo-nuclear osteoclast cells) [5].
Finally, in dialysis patients there isn't any difference in the skeletal responding to these hormones in relation to sex, whereas in the group of non-dialysis patients, females have greater degree of bone dynamics (the representation of HL/μm3, absolute number of HL and volume of HL-are significantly higher in females compared to males).
Frequency and size of HL in trabecular bone are pathological processes featuring fibrous osteitis. In patients non-treated with maintenance dialysis (natural evolution of the renal metabolic bone disease) the number of HL in trabecular bone is positively correlated with PTH (r = 0.65), but there is an inverse relationship between CT and volume of HL (r = -0.51). The process of multicentre bone resorption is probably directly related to PTH activity, and the extension of lacunar osteoresorption is well controlled by calcitonin.
In dialysis patients, the negative correlation between HL/μm3 and plasma level of c-iPTH (r = -0.61) implicates primarily non-lacunar (periosteocytic osteolysis) bone resorption, and CT more moderate inversely correlates (r = 0.31) with the size of HL.
In both groups of patients according to our results, a beneficial therapeutic effect would be expected upon application of exogenous calcitonin (reduction of the HL volume with the better therapeutic response in non-dialysis patients group).