Helicobacter Pylori affects many individuals in developing countries. Inflammation caused by helicobacter Pylori differs depending on the virulence factors, density and host tissue response of the bacteria. This study is intended to investigate the relation between density of helicobacter Pylori colonization in gastric mucosa in biopsy specimens and gastric mucosal inflammation severity.
Our study included 685 patients who applied to the hospital with dyspeptic complaints and had gastroscopy between the dates of January the 1st 2017 and January the 1st 2018. Histopathologic results of endoscopic biopsy specimens of these patients were retrospectively screened. Inflammatory severity and helicobacter Pylori intensity were assessed by Sydney scoring.
68.6% of the patients were infected with Helicobacter Pylori. As the intensity of inflammation increased, the intensity of Helicobacter Pylori increased as well. The relation between the intensity of Helicobacter Pylori and the severity of inflammation was statistically significant (p < 0.001).
Helicobacter Pylori infection increases gastric inflammation.
Helicobacter Pylori, Gastritis, Endoscopic biopsy
Helicobacter Pylori (H. pylori/Hp) is a microaerophilic gram negative bacilli initially detected in the antral gastric mucosa by Warren and Marshall [1]. Today, around half of the world's population is infected with Helicobacter Pylori [2]. Helicobacter Pylori is most commonly found in the antrum. Bacilli is placed deep into the mucosa gelatin covering the gastric mucosa or between the mucus layer and the gastric epithelium. H. Pylori causes mucosal inflammation with proinflammatory factors that it released and it also spoils mucus layer with released enzymes. The inflammation caused by helicobacter Pylori depends on the virulence factors, the density and the host tissue of the bacterium [3]. In various studies, H. pylori has been shown to be associated with gastritis, peptic ulcer, gastric carcinoma and Mucosal Associated Lymphoid Tissue (MALT) lymphoma [4-6].
Gastritis is an important pathological condition that causes gastric atrophy and cancer characterized by infiltration of inflammatory cells, especially lymphocytes and plasma cells, as well as neutrophils in the lamina propria. Gastritis does not have a specific finding that can be detected by endoscopic imaging [7,8]. However, histopathologic examination of the specimens taken from the tissue by endoscopy is accepted as the gold standard in the diagnosis of gastritis. The Sydney system is a good scoring system for histopathological evaluation and classification of gastritis. Sydney scoring is a classification based on topographic, morphological and etiological criteria of gastritis. It was accepted by consensus of gastroenterologist and pathologists at the World Congress of Gastroenterology in Sydney in 1990. The most important feature of this system is that the changes in the gastric mucosa are graded as five major histologic features (chronic inflammation, neutrophil activity, glandular atrophy, intestinal metaplasia and H. pylori density) [9]. This classification facilitates the clinical, endoscopic and morphological association of gastritis. It has been reported that there is a good correlation between polymorphonuclear cell infiltration and H. pylori infection histologically with the updated Sydney system [10].
This study is intended to investigate the relation between the intensity of Helicobacter Pylori colonization and the severity of gastric mucosal inflammation in biopsy specimens taken from the gastric mucosa.
This cross-sectional analytical study was carried out in Aksaray at Training and Research hospital, Internal Diseases and Gastroenterology outpatient clinics between the dates of January the 1st 2017 and January the 1st, 2018 on 685 patients who were admitted with dissipative complaints and underwent upper gastrointestinal endoscopy. The study data were obtained by retrospectively examining the histopathological results of endoscopic biopsy materials of patients.
The histopathological diagnosis and classification of gastritis in our study was assessed by Sydney scoring. In our study, inflammation grade was evaluated as as mild (< 1/3), moderate (1/3-2/3), and severe (> 2/3) according to the infiltration of neutrophils in lamina propria or surface epithelium.
The existence of a Helicobacter Pylori was investigated by May Grünwald Giemsa stain in mucosa samples obtained from antrum and its density was evaluated (Table 1).
Table 1: The evaluation of Helicobacter Pylori density. View Table 1
In evaluation of the obtained data, SPSS (Statistical Package for Social Sciences) for Windows 20.0 program was used for the statistical analysis. Descriptive statistics for continuous variables were given in terms of mean and standard deviation, and descriptive statistics for categorical data were given in frequency and percentage. The Chi-Square test was used to compare the data in the categorical structure. Correlation between parameters was done by Pearson correlation analysis. Correlation coefficient (r) was evaluated as weak between 0.00-0.24, moderate between 0.25-0.49, strong between 0.50-0.74, very strong between 0.75-1.00. The results were evaluated in a confidence interval of 95% and in a significance level of p < 0.05.
In our study, 65.8% (n = 451) of 685 patients were female and 34.2% (n = 234) were male. Mean age of all patients was 47.51 ± 15.61 year (47.79 ± 15.79 in women and 46.99 ± 15.28 in men. 68.6% (n = 470) of the patients were infected with H. Pylori. Histopathologic results of the patients were presented on the table (Table 2).
Table 2: The frequency of H. pylori and other histopathological findings. View Table 2
No statistically significant relationship was found between H. pylori presence and metaplasia status in our study (χ2 = 2.416, p = 0.120). There was a significant relationship between the severity of inflammation and the intensity of H. pylori in our study. As the intensity of H. pylori increased, the intensity of gastric inflammation increased as well (Table 3). The frequency and intensity of H. pylori in chronic active gastritis was statistically significantly higher than that of chronic gastritis (Figure 1). As the severity of inflammation increased, H. pylori frequency also increased (Figure 2).
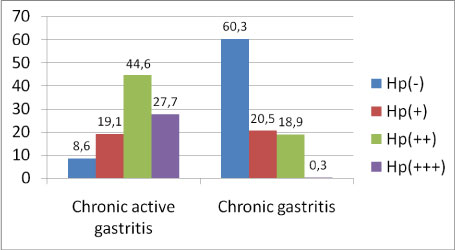 Figure 1: The degree of H. pylori intensity in the chronic gastritis and chronic active gastritis (χ2 = 2.092, p < 0.001).
View Figure 1
Figure 1: The degree of H. pylori intensity in the chronic gastritis and chronic active gastritis (χ2 = 2.092, p < 0.001).
View Figure 1
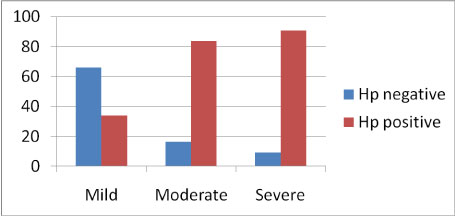 Figure 2: The relation of H. pylori frequency with degree of inflammation (χ2 = 2.010, p < 0.001).
View Figure 2
Figure 2: The relation of H. pylori frequency with degree of inflammation (χ2 = 2.010, p < 0.001).
View Figure 2
Table 3: The relation of H.pylori intensity with degree of inflammation (x2 = 3.940, p < 0.001). View Table 3
The prevalence of H. pylori is quite high throughout the world in developed and also in developing countries like our country. H. pylori positivity was 68% in our study. In a study by Konakcı, et al. in our country, H. pylori positivity was reported to be 50% [11]. In a study performed in Romania by Olar, et al. H. pylori prevalence was found to be 63% [12]. In a study in Japan, Nomura, et al. found out H. pylori positivity as 62% [13].
H. pylori plays a major role in the pathogenesis of gastric diseases (gastritis, ulcer, cancer) due to the fact that it causes mucosal destruction. However, it is not known exactly how the mucosal damage develops with H. pylori. It is thought that proteases released by H. pylori and gastric glycoprotein breaks down mucus structure and as a result, acidic gastric juice passes through this mucous membrane and causes mucosal damage [14]. Cell damage results from the direct toxic effect of ammonia, which is released into the gastric lumen by bacterial urease activity. It is also known that the various cytokines and chemotactic factors secreted by bacteria are also direct cytopathic and inflammatory initiating effects.
While there was no significant relationship between H. pylori and inflammation grade in various studies [15,16], there was a significant correlation in some other studies [17,18]. There was a statistically significant relation between the intensity of H. pylori and the severity of inflammation in our study. As the intensity of H. pylori increased, the severity of inflammation increased as well. In a study by Yakoob, et al. a significant relationship was found between H. pylori colonization intensity and chronic gastritis activity [19]. As in our study, Basir, et al. also found a significant relationship between H. pylori colonization intensity and chronic gastritis activity [20]. In a study performed by means of Histopathological examination of endoscopic biopsy specimens of 461 patients, Türkay, et al. reported that as the intensity of H. pylori increased, the intensity of inflammation increased, too [21]. In a study conducted by Alagöz, et al. a significant correlation was observed between H. pylori severity and lymphoplasmacytic cell infiltration and inflammation activation [22]. In contrast, in a study of 272 gastric biopsy specimens by Ardakani, et al. no significant relationship was found between the density of H. pylori and the severity of chronic gastritis activity [3]. Again, Choudhary, et al. found no significant relation between H. pylori density and chronic gastritis activity [23].
As a result, studies have shown a relationship between the intensity of H. pylori and the severity of gastritis. Histopathological examination and classification of gastritis are important for accurate diagnosis of H. pylori infection affecting upper gastrointestinal system by various complications. Proper treatment of H. pylori infection can prevent gastritis and its complications.