The aim of the present study was to clinically and histologically evaluate outcomes of alveolar ridge preservation technique using a collagen membrane associated to bovine-derived bone (MB), and a collagen membrane associated to porcine-derived bone (MP).
Twenty patients were enrolled in the present randomized controlled clinical trial, and underwent single-tooth extraction in the premolar/molar areas. Ten sites were treated with MB, while 10 sites with MP. Vertical and horizontal hard and soft tissue changes were clinically evaluated at the 4-month re-entry surgery for implant placement. At the same time bone core specimens were harvested for the histologic evaluation.
No differences in dimensional vertical and horizontal changes were encountered at the extraction sockets treated with MB and MP. Histomorphometrically, in sites treated with MB the percentage of newly formed bone was 49.08 ± 3.7, and the percentage of connective tissue was 16.37 ± 4.9, while in sites treated with MP, newly formed bone represented 57.13 ± 2.8, and non-mineralized connective tissue 13.65 ± 3.6. In the MB group, the mean percentage of residual graft particles and osteoid tissue was 13.49 ± 2.8, and 21.06 ± 3.8, respectively, while in the MP group, the same percentages were 11.74 ± 4.7, and 17.63 ± 3.8. Histomorphometrically, differences between MB and MP groups were statistically significant (p < 0.05).
Within the limitations connected with the small number of subjects, results showed that extraction sockets treated with MB and MP have similar vertical and horizontal clinical changes. Histologically, sites treated with MP presented at the 4-month evaluation a greater percentage of vital bone formation, and a lower percentage of residual graft particles.
Alveolar bone remodeling, Collagen membrane, Bovine-derived bone, Porcine-derived bone
To present a proof of concept for 3D virtual planning and maxilla repositioning in an experimental set-up with accuracy for the positional differences between the planned and postoperative outcomes less that 0.53 mm.
The spontaneous alveolar bone healing of extraction socket is characterized by a remodeling and reabsorption process that may lead in the first 6 months the loss of about 40% of the height and 60% of the width of the alveolar bone [1-10]. According with the indications of the 2012 Osteology Consensus Report [11], Alveolar Ridge Preservation (ARP) procedure is indicated as a mean of counteracting post-extraction volume loss, and of maintaining a stable ridge volume for optimizing subsequent implant placement. The standard of ARP procedure is the combination of barrier membrane and bone grafts [12-14]. The membrane avoids epithelial down growth into the socket, while grafting materials prevents membrane collapse. Non-absorbable and absorbable membranes associated with graft materials have been used for ARP with similar outcomes [15,16]. To avoid harvesting autograft and eliminating additional surgical procedures, xenografts have been proven as alternative graft materials for ARP. Bovine-derived bone (B) is one of the most widely used xenograft material, and its clinical advantages in ARP are largely supported by the available literature [17]. Porcine-derived bone (P) has been only recently used for bone regeneration. Preliminary data, showing that the P had excellent osteoconductive properties without adverse reactions suggested its use in ARP procedure [18-21]. Therefore, the aim of this randomized controlled study was to compare and evaluate the clinical and histologic outcomes of two techniques: (1) ARP with a collagen membrane and bovine-derived bone, and (2) ARP with a collagen membrane and porcine-derived bone. The two ESP techniques were clinically and histologically compared to each other to determine their respective efficacy.
Twenty patients, requiring extraction of a single premolar or molar tooth, who was interested in receiving a dental implant, were enrolled in the study between October 2016 and January 2017. Criteria for inclusion in the study were:
• Age ≥ 18 years,
• Good general health,
• Adequate restorative space for implant-retained restoration,
• At least 10 mm alveolar bone height without impingement on the maxillary sinus or inferior alveolar canal.
Exclusion criteria were:
• History of systemic diseases that contraindicate oral surgery,
• Long-term non-steroidal anti-inflammatory drug therapy,
• Oral bisphosphonate therapy,
• Pregnancy or lactation,
• Unwillingness to return for the follow-up examinations,
• Cigarette consumption > 10 per day.
All patients were informed about the evidence-based, positive outcome of ESP technique followed by implant placement, and the experimental approach. Each patient signed a free informed consent form after he/she has received detailed information about the study. Treatments were performed according to the principles outlined in the Declaration of Helsinki on experimentation involving human subjects.
Patients were divided into two groups of 10 subjects each:
1) MB group: Extraction sockets grafted with bovine-derived bone (MinerOss X®, BioHorizons, Birmingham, Al, USA), and covered with collagen membrane (Mem-Lok Pliable®, BioHorizons, Birmingham, Al, USA),
2) MP group: Extraction sockets grafted with porcine-derived bone (MinerOss XP®, BioHorizons, Birmingham, Al, USA), and covered with collagen membrane (Mem-Lok Pliable®, BioHorizons, Birmingham, Al, USA).
Mem-Lok Pliable®, used in both groups, is a porcine-derived resorbable collagen-based membrane with an estimated resorption time of 12-14 weeks. Miner-Oss X® is a highly porous anorganic bovine-derived bone mineral matrix with a particle size between 500 and 1000 µm. Miner-Oss XP® is a highly porous an organic porcine-derived bone mineral matrix with a particle size between 250 and 1000µm.
The allocation in MB and MP group was randomly assigned to each patient by computer-generated random number table. The randomization codes were stored in password-protected portable computers and enclosed in sequentially numbered, identical, opaque, sealed envelopes. The envelopes were opened in numerical order after tooth extraction. A third operator -not involved in enrollment or treatment of patients- performed data collection. This research study used a clinical database that included patients who were previously treated either as part of approved research protocols or as part of routine dental care using accepted therapy for each patient's specific clinical needs. As the current research involves an analysis of pre-existing data and current investigators did not have access to identifiable private information, this research did not require a specific approval by an institutional ethics board or committee.
All patients were prescribed prophylactic antibiotic therapy with 2 g of amoxicillin (or clindamycin 600 mg if allergic to penicillin's) 1 h before the extraction procedure and continued postoperatively with 1 g of amoxicillin (or 300 mg clindamycin) three time a day for 5 days. In addition, all patients rinsed for 1 min with chlorhexidine mouthwash 0.2% prior to the surgery (and twice a day for the following 3 weeks). Following local anesthesia with lidocaine with adrenaline 1:50,000, the identified tooth was extracted in a minimally traumatic manner with periotomes and without raising a full thickness flap. If necessary, the tooth was sectioned.
After tooth extraction the following clinical measurements were taken:
1) Vestibular Bone Thickness (VBT), Lingual Bone Thickness (LBT), and Ridge Width (RW). These measurements were obtained using a surgical caliper 2 mm from the crest of the ridge to the nearest 0.5 mm.
2) The Heights of the Vestibular and Lingual Crest (HVC- HLC) were measured by using a periodontal probe to connect the midfacial CEJs of the adjacent teeth, then measuring the vertical distance from that reference line to the crest of bone on the midfacial and midlingual sides.
In both groups, the extraction sockets were grafted up to the buccal and palatal alveolar bone walls and, subsequently, a collagen membrane was gently pushed under the interdental papilla with the use of periotomes. Sutures were used to stabilize the membrane and prevent loss of graft particles. The collagen membrane was left exposed. Postoperative instructions were given and all patients were prescribed ibuprofen 600 mg tablets. Subjects returned approximately 3 months after the extraction for a radiographic examination to evaluate the dimensions of the alveolus prior to implant placement. At the time of implant placement, 16 to 18 weeks post-extraction, minimal buccal and lingual flaps were reflected and ridge width, buccal and lingual heights were measured as previously described. A trephine drill with a 2.0 mm internal diameter was used to take a core biopsy approximately 8 mm in length.
The bone specimens were immediately fixed in 10% buffered formalin and embedded in a glycolmethacrylate resin. After polymerization, specimens were sectioned along their longitudinal and vertical axis to a thickness of 70 microns (plastic Microtome, RM 2265). Slides were stained with trichrome, and examined using an Olympus B51 microscope. The histomorphometry was performed using Bioquant® image analysis software (R&M Biometrics, Nashville, TN, USA) and images were captured with a Q-Imaging camera, 32-0013B-157, RETIGA, Colour 12-bit.11/22/2017
For the pooled data set as well as for each treatment group, the sample distributions of all variables were described univariately by showing means, medians, and standard deviations. For each variable, a Kruskal-Wallis non-parametric one-way analysis of variance was performed, each at a level of 0.05. Each non-parametric ANOVA was followed by two-sided Wilcoxon's rank sum tests for pairwise comparisons including Bonferroni correction. The analysis was performed using R version 2.13.0 (R Development Core Team 2010).
Twenty patients were screened for this study. One of the subjects in each group did not have enough ridge width for a correct implant placement upon re-entry; hence, a core biopsy was not taken and only clinical measurements were made. A total of 18 patients (10 males and 8 females with an average age of 51.5, ranging from 35 to 63) were allocated to the study groups of the trial. Eighteen bone specimens were harvested and 18 dental implants (Laser-Lok® Tapered, BioHorizons, Birmingham, Al, USA) were placed.
In the MB group, a mean RW, HVC, and HLC reduction of 1.25 ± 0.7 mm, 1.18 ± 0.8 mm, and 1.12 ± 0.9 mm respectively occurred. In the MP group, a mean RW, HVC, and HLC reduction of 1.19 ± 0.4 mm, 1.21 ± 0.8 mm, and 1.09 ± 0.6 mm, respectively was found (Table 1). No differences in dimensional vertical and horizontal changes were encountered at the extraction sockets treated with MB and MP. Histomorphometrically, in sites treated with MB the percentage of newly formed bone was 49.08 ± 3.7, and the percentage of connective tissue was 16.37 ± 4.9, while in sites treated with MP, newly formed bone represented 57.13 ± 2.8, and non-mineralized connective tissue 13.65 ± 3.6. In the MB group, the mean percentage of residual graft particles and osteoid tissue was 13.49 ± 2.8, and 21.06 ± 3.8, respectively, while in the MP group, the same percentages were 11.74 ± 4.7, and 17.63 ± 3.8 (Table 2). Histomorphometrically, differences between MB and MP groups were statistically significant (p < 0.05). In Figure 1, Figure 2, Figure 3 and Figure 4 is reported the histological analysis of two sites treated with BM and PM, respectively.
 Figure 1: Histological micrographs of vertical sections of extraction socket, and treated with collagen membrane and porcine derived-bone (Fast Green, 1.25X). View Figure 1
Figure 1: Histological micrographs of vertical sections of extraction socket, and treated with collagen membrane and porcine derived-bone (Fast Green, 1.25X). View Figure 1
 Figure 2: Histological micrographs of vertical sections of extraction socket, and treated with collagen membrane and bovine derived-bone (Fast Green, 1.25X). View Figure 2
Figure 2: Histological micrographs of vertical sections of extraction socket, and treated with collagen membrane and bovine derived-bone (Fast Green, 1.25X). View Figure 2
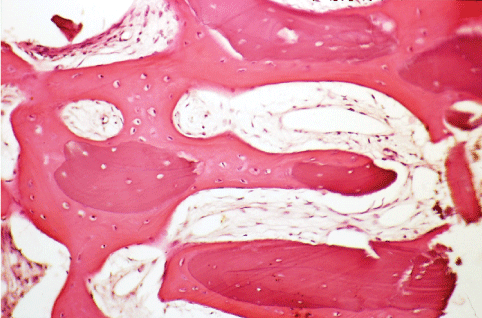 Figure 3: Histological micrographs of vertical sections of extraction socket, and treated with collagen membrane and porcine derived-bone (Ematossin eosin, 50X). View Figure 3
Figure 3: Histological micrographs of vertical sections of extraction socket, and treated with collagen membrane and porcine derived-bone (Ematossin eosin, 50X). View Figure 3
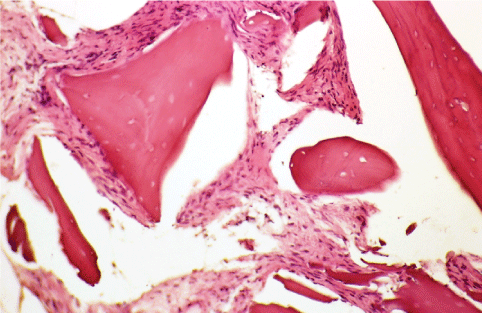 Figure 4: Histological micrographs of vertical sections of extraction socket, and treated with collagene membrane and bovine derived-bone (Ematossin eosin, 50X). View Figure 4
Figure 4: Histological micrographs of vertical sections of extraction socket, and treated with collagene membrane and bovine derived-bone (Ematossin eosin, 50X). View Figure 4
 Table 1: Horizontal and vertical changes at the re-entry surgery. View Table 1
Table 1: Horizontal and vertical changes at the re-entry surgery. View Table 1
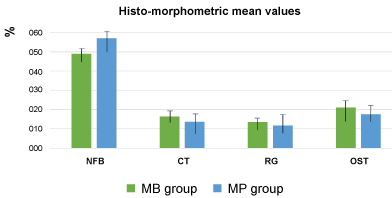 Table 2: Histo-morphometric mean values. View Table 2
Table 2: Histo-morphometric mean values. View Table 2
Direct intra-surgical measurements at re-entry surgery are considered as the most precise method to evaluate the bone volume changes following ARP. Results of the present study showed that both ARP techniques used (resorbable membrane plus bovine-derived bone, and resorbable membrane plus porcine-derived bone) help to counteract bone reabsorption and remodeling of the socket walls, but neither technique was able to entirely compensate for the alveolar ridge reduction. These results agree with previous published data indicating that others factors, such as the extraction socket location, the socket anatomy, and/or the thickness of buccal bone plate, influence the amount of post-extractive alveolar bone remodeling irrespective of the xenograft used [22]. Another factor that may influence the dimensional changes of alveolar ridge is the surgical approach. Some investigations reported that an extractive flapless approach is associated with significantly more horizontal and vertical bone reduction [23,24]. In addition, less RW changes have been observed when primary closure was achieved [23]. Results of the present study showed that the flapless approach associated with secondary soft tissue closure and membrane exposure did not affect the efficacy of ARP technique. These data are in contrast with the over-mentioned findings. Our results could be linked to physical and chemical features of the collagen membrane used in the present study which, in a vitro analysis [25] showed to be characterized by a rate of reabsorption of 12/14 weeks at the intra-oral implantation site, eliciting a low inflammatory and foreign body giant cell response. It is possible hypnotized that the low degree of inflammation and foreign body response may result in enhanced tissue integration and improved wound healing in terms of minimizing scar-like tissue formation. However, since the present study did not include a control group treated with a flap approach and primary closure, further studies are needed to confirm this hypothesis. In addition, the little sample of the present study didn't allow to evaluate the influence of implant site location (mandible or maxilla) on the overall results. Therefore studies with an higher number of patients and extraction sites are needed to confirm our results.
The final goal of any grafting technique should be the achievement of 100% living bone and reactive tissue able to undergo a sustained state of remodeling to maintain the mechanical and the biological function overt time, Moreover, the ideal graft material should be able not only to minimize the ridge remodeling, but also to promote bone formation as fast as possible to shorten the treatment time. Some studies, reporting only a partial resorption of the grafted particles at short time points [26,27] suggested that xenogenic materials may interfere with the earliest stages of socket healing, and arise doubts on the achievement of the osteointegration of implants inserted in augmented sites. Histologic data of the present study indicated that sockets treated with membrane and bovine-derived bone show at the re-entry surgery for implant placement the presence of a more residual graft material, compared to sockets treated with porcine-derived bone. This could indicate a different influence of the bovine- and porcine derived-materials on the alveolar bone healing process. This hypothesis is also supported by the higher percentage of osteoid tissue (bone in maturation phase) founded after 4 months in extraction sockets grafted with bovine-derived bone, compared to that founded in sockets grafted with porcine derived bone. The different histological results between the two groups (BM and PM) might be explained by the different resorption rate of two xenogenic biomaterials. Indeed, the resorption process of xenogenic biomaterials is linked with the pore size, the pore morphology, the pore percentage, the connection between pores and the granulometry.
In conclusion, within the limits of the present study results showed that, the use of bovine and porcine-derived bone associated with a collagen membrane may allow for a preservation of the alveolar ridge volume that is highly desirable for both esthetic and function of the future implant restoration, even thought neither technique was able to entirely compensate for the alveolar ridge reduction. In addition porcine-derived bone, compared to bovine-derived bone, seems to allow for an accelerate alveolar bone healing.