Odontogenic myxoma (OM) is a benign, locally invasive and aggressive, non-metastasizing neoplasm of the jaw bones. This article presents a rare case of odontogenic myxoma occurring in the maxilla of a 42-year-old female patient with review of the literature emphasizing a huge controversy regarding the surgical management.
Odontogenic tumor, Myxoma, Maxilla, Long-term, Follow-up
Odontogenic myxoma (OM) is a benign, locally invasive and aggressive, non-metastasizing neoplasm of the jaw bones.
Myxomas of the head and neck can be identified in two forms (I) Facial bone derived, which had been subdivided in the past into true osteogenic myxoma and odontogenic myxoma and (II) Soft tissue myxoma derived from perioral soft tissue, parotid gland, ear and larynx [1-3].
There has been a great deal of controversy regarding the origin of myxomatous tumors. Virchow, in 1863, coined the term myxoma for a group of tumors that had histologic resemblance to the mucinous substance of the umbilical chord [4,5].
Pathogenesis is disputed. Two hypotheses have been suggested: Tumoral development in residual conjunctive tissue stem cells, or myxomatous degeneration of the fibrous stroma [6].
Odontogenic myxoma of the jaws is a rare benign tumor, incidence rate is approximately 0.07 new cases per million people per year [7,8].
All reports generally agree that the tumour frequently presents in the second or third decades of life [8-10].
Most common clinical characteristics of odontogenic myxoma are swelling of the jaws with only few patients complaining of pain, paraesthesia and ulceration, 28% for pain and 56% for swelling [11].
Several studies indicating that there is a higher rate of tumour incidence in the mandible than the maxilla [12-15], and almost occurring in the premolar and molar region of both jaws and rarely crosses the midline [12,16,17].
Cortical expansion and perforation are common findings; however, maxillary myxomas often extend into the sinus [18]. The adequate management of Odontogenic myxoma (OM) is a challenging problem against the high risk of recurrence.
The aim of this paper is to report a case of odontogenic myxoma that affected the left maxilla focusing especially on surgical management.
A 42-year-old female was referred to our department with a complaint of a growing swelling in the area of the canin and premolar molar in his left maxilla that had been noticed a few weeks before. Upon admission, she noticed a swelling measuring about the size of a peanut in the gingiva of upper premolar tooth. That lesion had been excised by a dentist and the histopathologist conclude to epulis. Although, the tumour had recurred two months later and gradually increased to its present size.
The patient's medical history and review of systems were unremarkable.
On extraoral examination, a solitary swelling was visible on left side of face which was extending anterioposteriorly from nasio-labial fold to corner of mouth. The swelling was round to oval in shape and approximately 2 cm × 3 cm in dimension with ill-defined borders. Overlying skin appears to be normal with no secondary changes. On palpation, the swelling was non-tender, firm to hard in consistency, non-compressible, non-reducible, and non-pulsatile, non-fluctuant and was fixed to underlying structure. There was no cervical mass or lymphadenopathy was found.
On intraoral examination, an overgrowth was visible on maxillary left anterior teeth region extending from distal aspect of 23 to mesial aspect of 26 measuring approximately 3 cm × 4 cm in diameter round to oval in shape.
With well defined borders and lobulated surface there by obliterating the right buccal vestibule. The mucosa overlying the area of the lesion was the same colour and texture as the surrounding mucosa (Figure 1).
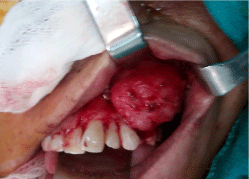 Figure 1: Intraoral image showing an overgrowth on maxillary left anterior teeth region extending from distal aspect of 23 to mesial aspect of 26.
View Figure 1
Figure 1: Intraoral image showing an overgrowth on maxillary left anterior teeth region extending from distal aspect of 23 to mesial aspect of 26.
View Figure 1
On palpation, the overgrowth was nontender, pedunculated, bony hard in consistency, and non-pitting on application of pressure with rolled out margins.
The sensory examination of the infraorbital nerve was normal.
The rest of the patient's clinical head and neck examination and general examination were non-contributory.
Panoramic radiograph revealed a single large expansile radiolucent lesion extending from the left first premolar to second molar and occupied the left maxillary sinus without any trabeculations in the area of bony destruction (Figure 2).
 Figure 2: Panoramic radiographs showing a single large expansile radiolucent lesion without any trabeculations in the area of bony destruction.
View Figure 2
Figure 2: Panoramic radiographs showing a single large expansile radiolucent lesion without any trabeculations in the area of bony destruction.
View Figure 2
Under general anesthesia the tumour was exposed through left intraoral upper vestibular incision from 21 to 27 regions, enucleation of the tumour was performed with curettage of the surrounding bone through.
Macroscopically, the surgical specimen measured approximately 3 × 3.9 × 2.5 cm, on gross examination showed grayish white, glistening smooth, gelatinous mass (Figure 3).
 Figure 3: Gross examination showed grayish white, glistening smooth, gelatinous mass.
View Figure 3
Figure 3: Gross examination showed grayish white, glistening smooth, gelatinous mass.
View Figure 3
Histopathological examination of excised specimen showed tumor mass without encapsulation. It showed spindle and stellate shaped cells in loose, abundantly myxoid connective tissue stroma, closely resembling the mesenchymal portion of a developing tooth.
Some areas showed moderately dense collagen fibers. Few strands of inactive odontogenic epithelium were seen within the connective tissue stroma. Overall histological appearance of the lesion revealed more amount of myxoid stroma in a less fibrous and acellular background (Figure 4).
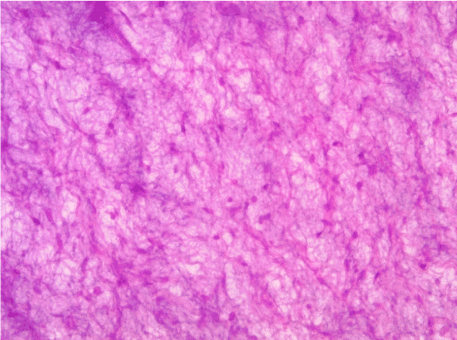 Figure 4: Microphotograph shows mixed area of fibrous tissue and inconspicuous strands of odontogenic epithelium in a myxoid stroma; magnification: x100.
View Figure 4
Figure 4: Microphotograph shows mixed area of fibrous tissue and inconspicuous strands of odontogenic epithelium in a myxoid stroma; magnification: x100.
View Figure 4
Outcome and follow up: Healing was uneventful and follow-up after 4 months showed no recurrence of the lesion. There have been no clinical or radiological signs of recurrence over 02 years follow-up.
The treatment of odontogenic myxoma is primarily surgical, it varies from simple enucleation and curettage to more extensive radical surgery, regarding maxilla it could be partial and total maxillectomies [17,19-21]. Conservative treatments have several advantages over more radical treatments, such as segmental or block resection, with reconstruction surgery. Conservative treatments are substantially less invasive, can be achieved by means of an intraoral surgical approach, preserve function and aesthetics especially in the maxilla because of the proximity of vital structures and the more complex anatomy, in addition offered a shorter hospitalization time [22].
Several investigators have recommended that tumor size should determine whether a radical or more conservative surgical approach should be adopted [9,20].
Boffano, et al. suggested that conservative treatment by enucleation and curettage is recommended when the diameter of an odontogenic myxoma is less than 3 cm, whereas a segmental resection with immediate reconstruction is preferred in patients with larger tumors [23].
Recently, Zanetti, et al. strongly suggested conservative treatment should involve enucleation of the lesion with a wide curettage of normal tissue or a generous amount of apparently uninvolved surrounding tissue, or even peripheral osteotomy, as this has the advantage of preserving vital structures and maintaining oral function [24].
Recurrence is considered to be directly related to the type of therapy, the rate after simple enucleation and curettage has been reported to be as high as 25% [3,19].
Some authors report that the risk of recurrence is greater as the myxoma is not encapsulated and its myxomatous tissue infiltrates the surrounding bony tissue without causing immediate destruction than to the cell proliferative index which was relatively low [22,25,26].
Batsakis accused incomplete removal the reason of recurrence than the intrinsic biological behavior of the tumor [27].
The aggressive nature of OM is well documented in the literature, it can invade critical structures like the orbital floor and the skull base, resulting in a non-resectable situation, especially in cases of recurrence [20]. For the reason, wide resection with 10 to 15 mm margins is generally recommended [28,29].
Yoav LEISER took into consideration that the treatment protocol for OMs resembles that of ameloblastoma, solid type, especially when locally aggressive behaviour is encountered which includes, cortical bone perforation and major tumour expansion and infiltration in a short period of time (weeks to months). A radical primary resection of the tumour with maximal preservation of surrounding anatomic structures is necessary (healthy anatomical borders of 0.5-1 cm beyond the tumours radiographic borders) [30].
It's recommend that further, radical surgery is warranted after recurrence of conservatively-treated odontogenic myxoma, a view that believe some authors in the literature [8,20,31-33].
Reconstruction can begin immediately following the surgical procedure or delayed until an adequate disease free period has past. In our experience, small bony defects (smaller than 5 cm) can be reconstructed using buccal fat pad (maxilla) [34], or using corticocancellous iliac crest bone graft [35]. Larger defects (more than 5 cm) usually require primary prosthetic reconstruction (obturator) [36] followed by a final obturator [10]. Due to the high recurrence rates of these lesions, delayed reconstruction is the treatment of choice, especially in the more locally aggressive tumours. Prosthetic reconstruction in maxillary lesions is highly advisable due to low postoperative morbidity and almost immediate phonetic and masticatory function. Furthermore prosthetic reconstruction by an obturator allows direct visualization of the primary site for recurrence detection [25].
A follow-up period is clearly also necessary. It has been recommended that patients should be followed closely for at least the first 2 years after surgery, which represents the period during which the neoplasm is most likely to recur [9,13].
Rocha, et al. suggested that 5 years of surveillance is needed to confirm that the lesion has healed successful excision, and periodical clinical and radiographic follow-up should be maintained indefinitely [22].
This review of literature shows that all datas are based on cases report, in the other hand each conservative and radical surgery consist of advantages and inconvenients. Thus, to determine the most recommended surgery method is difficult. In future, it is judicious to carry out a metanalysis study which only would offer in future a precise analysis of recurrence and allow a clearly guideline.
Management varies depending on the location and size of the tumour, the age of the patient and individual experience. They range from minimally invasive excisional biopsies to an en bloc resection of a multitude of relevant structures.
The authors declare that they have no conflicts of interest concerning this article.
The authors are grateful for a cytopathologist qamouss wadie who provided a histopatholgical analysis and images.