Objective: Post-tonsillectomy pain is a common concern. This study aimed to assess the effectiveness of peritonsillar infiltration of tramadol during tonsillectomy in terms of analgesia requirement, pain scores and time to commencement of liquid diet, as there is heterogeneity in the randomized controlled trials conducted.
Data sources: Pubmed, Embase, Medline, Cochrane Database.
Review methods: All randomized controlled trials comparing peritonsillar infiltration of tramadol with normal saline were included in the study. Odds ratio (OR) and 95% confidence interval (95% CI) were calculated for dichotomous data, whilst continuous data was analysed using standardized mean difference (SMD). The primary outcome was time to first analgesic request; secondary outcomes were pain scores (CHEOPS) at 1 hr, 4 hr and 12 hours postoperatively, post-operative nausea and vomiting (PONV), and time to commencement of liquid diet. All statistical analyses were performed using Review Manager 5 (RevMan; Version 5.1). Further analysis of the results through trial sequential analysis and meta-regression was done to improve the validity of the outcomes of interest.
Results: Twelve published between 2001-2022, including patients aged 3-48 years were identified and analysed. Time to request first analgesia [SMD 1.75; 95% CI 0.70-2.81; p < 0.001]. The calculated number needed to treat for rescue analgesia was 2.94 (1/risk difference = 1/0.34). Pain scores (CHEOPS) at 1 hr, [SMD -2.40; 95% CI -3.23 – -0.58; p < 0.00001], 2 hours [SMD -1.59; 95% CI -2.97 – -0.21; p = 0.02], 4 hrs [SMD -1.64; 95% CI -2.71 – -0.56; p = 0.003] and 24 hours [SMD -0.70; 95% CI -1.27 – -0.13; p = 0.02] favored the experimental group as did time to liquid diet [SMD -0.87, 95% CI -1.70 – -0.04, p = 0.04]. Publication bias was suggested with Egger's test (p = 0.02), but this was not supported by the Duval and Tweedie fill and trim analysis nor meta regression based on the amount of Tramadol injected (p = 0.81). Trial sequential analysis supported using Tramadol infiltration over saline for tonsillectomy.
Conclusion: Intraoperative peritonsillar infiltration of tramadol yielded significantly greater control of post-tonsillectomy pain across all measured outcomes when compared to patients who received normal saline. This included a prolonged time to first request of analgesia, a lower CHEOPS and earlier commencement of a liquid diet post-tonsillectomy.
Tonsillectomy, or surgical removal of the tonsils, has been described as early as the first century CE [1] and remains a commonly performed surgical procedure. It is generally a safe procedure; however, there are several accepted complications and side effects. Post-operative pain is a common side effect of tonsillectomy and is experienced by almost all patients. Post-operative pain is not only relevant to patient satisfaction and comfort, but may also delay a return to normal oral intake leading to dehydration and increased risk of post-operative haemorrhage [2].
The exquisite sensitivity of the tonsillar fossae represents a significant challenge in achieving adequate control of post-tonsillectomy pain [3]. Intraoperative application of local anaesthetics, either by peritonsillar infiltration or by soaking swabs and placing them in the tonsillar fossae, is a widely used technique to try and improve post-tonsillectomy pain. This is a method of 'pre-emptive' analgesia which is employed to reduce the sensitisation of peripheral and central pain pathways that occurs in response to tissue injury and inflammation [4]. In addition to traditional local anaesthetic agents, tramadol is now increasingly used as a peritonsillar infiltrate to improve post-tonsillectomy pain. Although tramadol is primarily used as a centrally acting opioid agonist, its local anaesthetic activity renders it an opportune avenue for post-tonsillectomy analgesia. But its efficacy remains inconclusive.
There are currently several randomized control trials, which look at the impact of peritonsillar tramadol infiltration on post-tonsillectomy pain.
This is the first systematic review and meta-analysis, which aims to look at analgesic efficacy. Specifically, we aimed to assess the effectiveness of peritonsillar infiltration of tramadol during tonsillectomy in terms of analgesia requirement, pain scores and time to commencement of liquid diet.
A systematic review and meta-analysis of all published data pertaining to the use of peritonsillar tramadol infiltration for post-tonsillectomy pain.
Electronic searches of MEDLINE, EMBASE, Pubmed and the Cochrane Database were performed, with no limits placed on date or language. The search strategy is summarized in Appendix 1, and a summary of the search process is shown in Appendix 2. Reference lists from identified studies were also screened for eligibility.
All abstracts were assessed for eligibility against the inclusion criteria. We included all randomised control trials comparing peritonsillar infiltration of tramadol with normal saline. Study populations consisting of both adults and children were included. Studies were excluded if combination treatments (such as peritonsillar tramadol plus IV tramadol) were used rather than peritonsillar tramadol alone, along with studies that did not use peritonsillar saline as the control group. Two separate authors performed initial screening of the abstracts, during which duplicates were identified and removed. Reference lists from selected studies were also assessed against the eligibility criteria (Appendix 3).
Data was collected (Appendix 4) and entered into a standardized spreadsheet for analysis. Initial data extraction was performed by a single author and this was repeated by a second author for quality assurance. Odds ratio (OR) and 95% confidence interval (95% CI) were calculated for dichotomous data, whilst continuous data was analysed using standardized mean difference (SMD). Primary outcome was time to first analgesic request; secondary outcomes were pain scores (CHEOPS) at 1 hr, 4 hr and 12 hrs postoperatively, post-operative nausea and vomiting (PONV), and time to commencement of liquid diet. All statistical analyses were for these outcomes performed using Review Manager 5 (RevMan; Version 5.1).
Meta regression was performed to identify the cause of heterogeneity. To evaluate the risk of publication bias in relation to all outcomes, a funnel plot was drawn and visually examined. Egger's regression coefficient was calculated for comparison. On the y-axis, the standard error of the mean difference of the outcome of interest (measure of trial size) was plotted as a function, on the x-axis, of the mean difference of the outcome. Our results were verified with the performance of Duval and Tweedie's trim and fill test, in which the smaller studies producing funnel plot asymmetry are removed and the omitted trials and their missing counterparts are replaced, and Egger's linear regression test using Comprehensive Meta-Analysis (Version 3.3, Biostat, 2014, New Jersey, USA). Funnel plot with precision modeling were performed as well as cumulative analysis based on Tramadol dosage.
Trial sequential analysis was performed with TSA viewer (Version 0.9.5.10 Beta, Copenhagen Trial Unit, 2016, Copenhagen, Denmark). The Sidik Jonkman random effects model, that is less likely to underestimate the heterogeneity between trials, was chosen to calculate the Z-statistic, which is equal to the meta-analysed intervention effect divided by its standard error.
The strength of the available evidence can be considered by determining the required information size for a conclusive and reliable meta-analysis. It can be derived from the risk of type-1 and type-2 statistical errors, which we set at 5%, and 20% and 10% respectively, resulting in a power of 80% and 90%. In order to control for the risk of type 1 error, the Lan and DeMets alpha-spending function was used to adjust the threshold for statistical significance to account for the elevated risk of random error before the meta-analysis has surpassed its required information size. In order to control for the risk of type-2 error, an extension of the Lan and DeMets alpha-spending function was used to adjust the threshold for non-inferiority or non-superiority, or no difference, representing what is referred to as futility boundaries before the meta-analysis has surpassed its required information size. We also penalized the Z-values by the strength of the available evidence and number of statistical tests, called the law of the iterated logarithms. This law is utilized to adjust the inflation of Type I errors due to repeated significance testing.
Searches identified twelve randomized controlled trials published between 2001-2022, comprising a cumulative total of 312 patients receiving peritonsillar tramadol (age 3-48). Study characteristics are summarized in Appendix 5.
Seven studies (408 patients) reported on time to first analgesia request [5-9]. SMD 1.75; 95%CI 0.70, 2.81; P = 0.001 (Figure 1) demonstrating a statistically significant, thus a favourable result in the peritonsillar tramadol group when compared with placebo. From this the number needed to treat was calculated as 2.94.
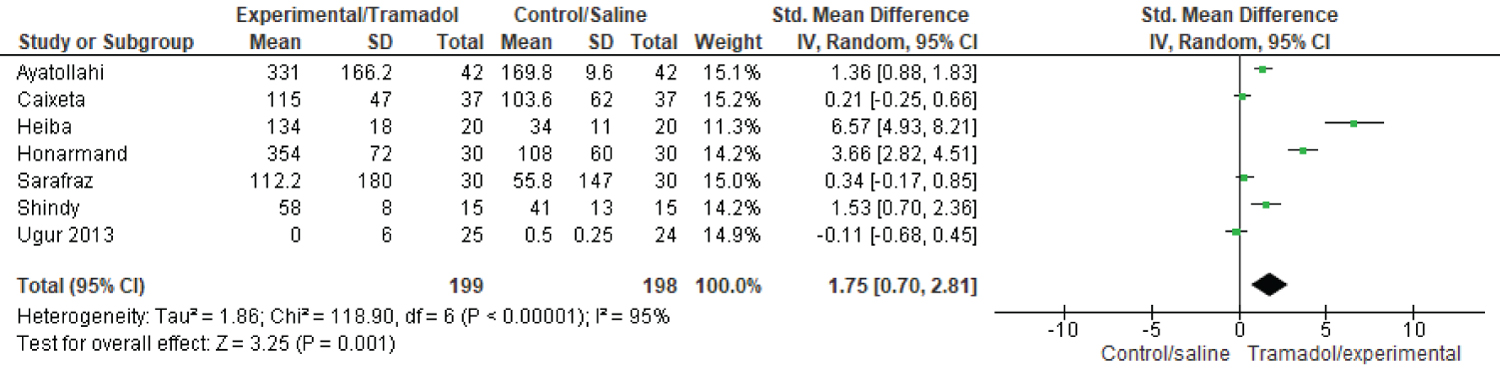 Figure 1: Time to first analgesia request in tramadol and control groups.
View Figure 1
Figure 1: Time to first analgesia request in tramadol and control groups.
View Figure 1
Pain outcomes were assessed via CHEOPS scores in four of the included studies (234 patients) [5,7,9]. CHEOPS at 1 hour [SMD -2.40; 95%CI -3.23, -1.58; P < 0.001], 2 hours [SMD -1.59; 95%CI -2.97, -0.21; P = 0.02], 4 hours [SMD -1.64; 95% CI -2.71, -0.56; P = 0.003] and 24 hours [SMD -0.70; 95% CI -1.27, -0.13; P = 0.02] all favoured the tramadol group compared with placebo (Figure 2).
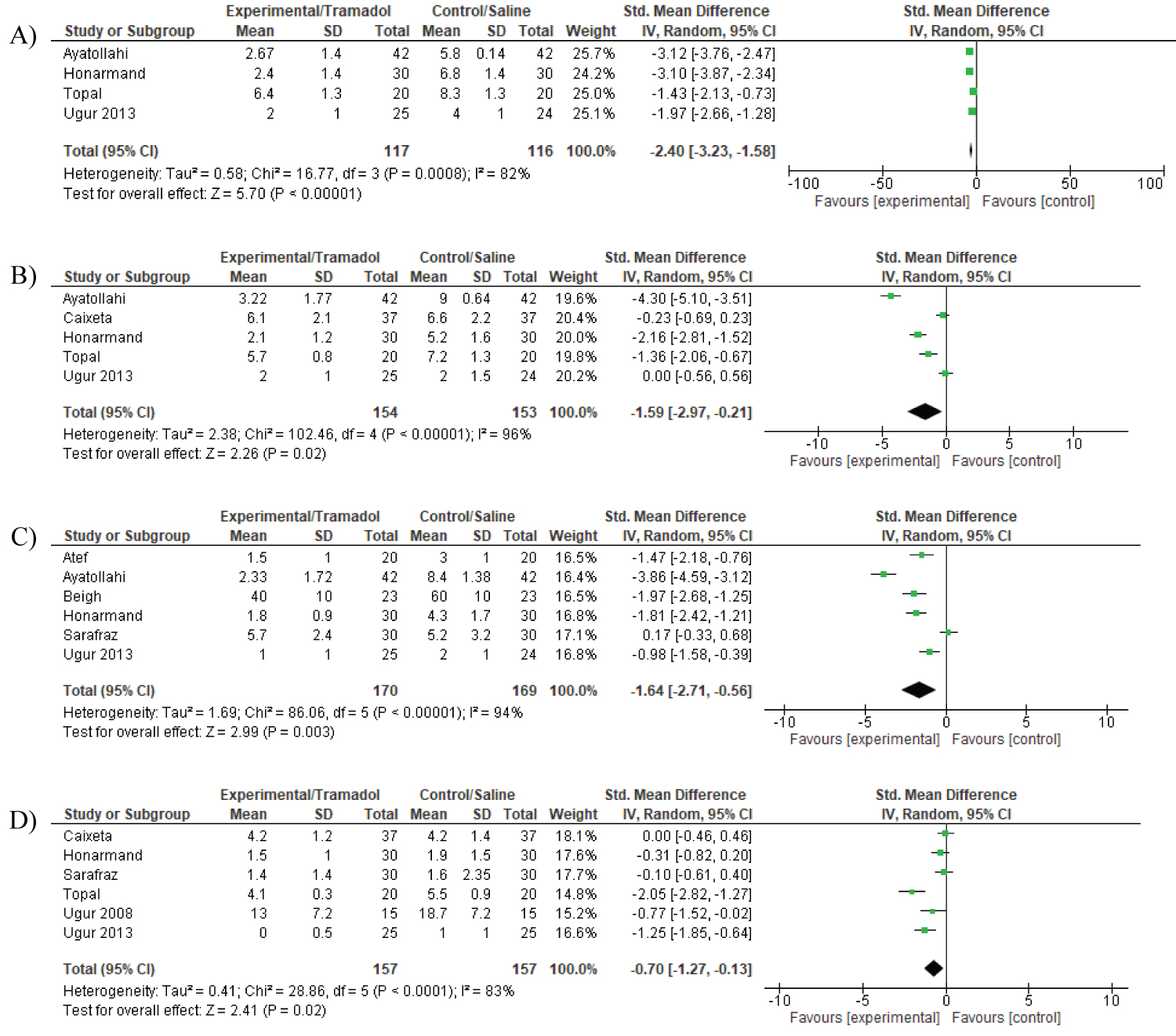 Figure 2: Pain scores (CHEOPS) at A) 1; B) 2; C) 4, and; D) 24 hours post tonsillectomy.
View Figure 2
Figure 2: Pain scores (CHEOPS) at A) 1; B) 2; C) 4, and; D) 24 hours post tonsillectomy.
View Figure 2
Four studies reported on the occurrence of post-operative nausea and vomiting [5,7,10,11] with no difference observed between groups (OR 1.02; 95%CI 0.42, 2.49; P = 0.96 ) (Appendix 6).
Six studies reported on time to commencing liquid diet (Figure 3) which significantly favored the tramadol group when compared to placebo [SMD -0.87; 95% CI -1.70, -0.04; P = 0.04].
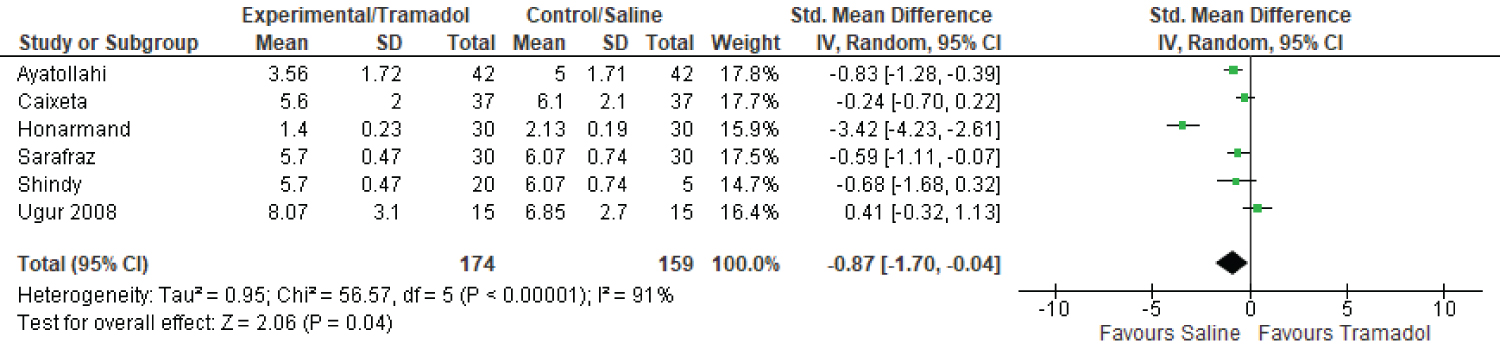 Figure 3: Time to commencing a liquid diet for tramadol and control groups.
View Figure 3
Figure 3: Time to commencing a liquid diet for tramadol and control groups.
View Figure 3
Four studies used different variations of visual-analogue scores (VAS) [6,8,11,12] however, due to the heterogeneity of this data, a meta-analysis was not possible. Descriptive analysis of these studies demonstrated a trend towards less pain among the tramadol group with varying degrees of statistical significance. Appendix 7 summarises these four studies, the type of VAS used and a summary of their outcomes.
In the TSA the required information size of 314 patients were reached for 80% and 394 patients for 90% power, and the Z-line crossed the trial sequential monitoring boundary and the futility boundaries suggesting firm evidence of using Tramadol infiltration over saline for tonsillectomy. Performing the law of the iterated logarithm, the green line was parallel to the z curve thereby demonstrating no differences either for 80% or 90% power (Appendix 8).
Egger's test suggested publication bias (P = 0.02;) but the Duval and Tweedie fill and trim did not show any publication bias neither did a visual inspection of the precision modelling of the study. Meta regression based on the amount of Tramadol injected did not have any overall impact either (P = 0.81). Cumulative analysis demonstrated for Tramadol low to high dose no difference until 60% of the relative random weight was reached following which there significant difference was observed. For the trim and fill effect there was only one trial imputed to the right of the mean difference to impart symmetry- observed values being of point estimate 1.80 (0.71, 2.88) and for the adjusted value being 2.18 (0.88, 3.48).
There are a number of factors which determine post-operative pain, including patient factors (e.g. an individual's response to pain and pain tolerance), operative factors such as the technique used and the use of intraoperative local or systemic analgesia, and post-operative care including the type, dose and frequency of analgesia used.
Pre-excisional 'pre-emptive' analgesic infiltration has been used for a long time in an attempt to improve post-operative pain outcomes. It was first described by Allen in 1953 who infiltrated efocaine (1% prilocaine, 1% prilocaine hydrochloride and 5% butyl aminobenzoate) into the tonsillar fossa post tonsillectomy or adenotonsillectomy [13].
A myriad of drugs have been proposed, to improve post-operative pain, through a variety of routes including IV, IM, direct infiltration and a combination of the three. In this meta-analysis we looked at all articles comparing tramadol infiltration with saline, which we labeled as our placebo.
Tramadol is a synthetic opioid of the aminocyclohexanol group which demonstrates central opioid agonism with less respiratory depression compared to morphine [14].
Post-operative surgical pain is caused primarily by trauma at the nerve endings. Through this process various chemical mediators namely serotonin, bradykinin, substance P and histamine are released which in turn activate noci-receptors. This means that pre-incisional anaesthetic infiltration could suppress the release of these mediators [15,16]. The use of such an analgesic, therefore, may provide better pain control in the immediate post-operative period and lead to fewer complications by causing less post-operative nausea, vomiting and delayed eating.
So far, to the best of our knowledge, no other reviews have conducted a meta-analysis looking at a comparison between tramadol and saline infiltration. In all the papers included in our meta-analysis a comparable cold-steel surgical technique was used thus ensuring as similar comparison as possible.
Our results demonstrated that tramadol infiltration increases time to first post-operative analgesia request (P = 0.001), and lowers post-operative pain scores (CHEOPS) at 1, 2, 4 and 24 hours (P < 0.001, P = 0.02, P = 0.003, and P = 0.02, respectively). Pain was further assessed with VAS, but due to the heterogeneity of the data it was analysed using descriptive statistics. Shindy, et al. showed that there were significantly lower pain scores (VAS) when comparing tramadol with saline but that there was also a significantly lower pain score when comparing tramadol and tramadol with bupivacaine [8]. These studies add weight to the argument that tramadol provides both immediate and a lasting reduction in post-operative pain.
Though achieving better pain control is an important outcome, other studies have investigated ways to try and improve upon this. Honarmand, et al. have shown that the addition of IV ketamine, to direct infiltration of tramadol, can prolong the analgesic affect up to 24 hours [7]. However, Ayatollahi, et al. demonstrated that ketamine causes hallucinations and did not have the same efficacy as tramadol in reducing post-operative pain [5].
Individually many studies have hypothesised that improved pain control should lead to a shorter time until commencement of liquid diet or a reduction in post-operative nausea and vomiting and have shown that this can be significant. This was also shown in our study, where tramadol reduces the time to commence a liquid diet (P = 0.04), which can be attributed to the less pain experienced by the tramadol group. A reduction in post-operative nausea and vomiting is also reflected in our data supporting the above.
When looking at new regimes to improve patient care it is important to monitor their outcomes and document any complications that may occur. The use of local anaesthetic has been theorised to increase the rate of post-operative haemorrhage but this has not been shown in any of the studies that we have used where there were no post-operative complications relating to tramadol infiltration.
Meta regression demonstrated that the heterogeneity in the amount of tramadol had little impact on the outcomes of the study, with P = 0.81, therefore raising the assumption that the heterogeneity observed before performing the meta regression could be due to the nature of the trials themselves rather than the difference between studies. Cumulative analysis indicates that doses of tramadol below 60% of body weight did not provide a statistically significant impact on the outcomes measured.
When comparing the results of this meta-analysis to the previous comparator paper [17,18], meta regression and TSA helped to further clarify heterogeneity and sample size. Although Tsaousi looked at similar outcomes we had a much more defined intervention and control groups compared to theirs. We also calculated the NNT for rescue analgesia required postoperatively and found that time to start of liquid diet was comparatively significant in this study.
There are limitations to this study. The number of studies comparing peritonsillar tramadol and saline are few along with the studies that mention the different outcomes looked at. There is significant heterogeneity amongst the studies but meta-regression performed did not reveal any obvious cause for it thereby raising the assumption that the heterogeneity observed could be due to the nature of the trials within themselves rather than between. Moreover, the number of patients included in the primary outcome were few, however following a trial sequential analysis (TSA) we found the z curve was significantly more than required information size for both 80% and 90% power calculation.
However, when TSA was performed for CHEOPS at the chosen time intervals, the z curve was significantly more than the required information size (RIS) for 80% power at all time intervals except at 2 hours where the RIS was not reached implying that we needed more studies to make a firm conclusion at 2 hour interval period for CHEOPS score. The z curve of TSA for starting oral intake was significant and the RIS was reached at 80% power.
In conclusion, 'pre-emptive' tramadol infiltration can be used safely to improve pain control up to 24 hours post-operatively, prolong time to first request of analgesia and reduce the time to commence a liquid diet post-tonsillectomy.
The authors have no conflict of interest to declare.