The full blood count (FBC) analyzers, Sysmex XE-5000 and Unicel DxH 800, are equipped to perform routine and extended parameters tests; thus, this study considered extended red blood cell (eRBC) parameters to distinguish iron-deficient erythropoiesis related disorders.
Malaysian female subjects comprising three main ethnic groups (Malay, Chinese and Indian) were included. Three groups of findings were distinguished based on FBC, morphology, and iron status of the subjects: normal, latent iron deficiency (LID) and iron deficiency anemia (IDA). Spearman's correlation test was performed to assess the concordance of FBC parameters in both analyzers. Test of normality and mean comparison tests were carried out to determine statistically significant parameters (p < 0.05) in discriminating iron-deficient erythropoiesis. Finally, receiver operating characteristic (ROC) curves were generated to determine area under the curve, sensitivity, and specificity of the parameters.
Malaysian female subjects of the Indian ethnic group were observed to have higher prevalence of IDA (55.8%) and LID (42.7%) compared to other ethnic groups. Both analyzers were confirmed to have good correlation between RBC-Y in Sysmex XE-5000 and MAF in Unicel DxH 800 (r = 0.918). The best parameter to distinguish LID from IDA is MAF (AUC: 0.954; 95% CI: 0.927-0.980; Cut-off: 9.25; Sensitivity: 89.02%; Specificity: 88.46%) while the best parameter to distinguish IDA from LID is %MicroR (AUC: 0.915; 95% CI: 0.874-0.956; Cut-off: 0.655%; Sensitivity: 85.57%; Specificity: 86.58%).
This study conclusively reported the utility of eRBC parameters from Sysmex XE-5000 and Unicel DxH 800 to distinguish LID from IDA. These eRBC parameters could potentially form more robust diagnosis of subjects suspected with iron-deficient erythropoiesis related disorders.
Anemia, Full blood count, Latent iron deficiency, Microcytes, Red cell parameters
FBC analysis is an essential staple in performing hematological diagnostic tests ever since its advent decades ago. Having aided diagnostics laboratories in elaborating hematological findings, more recent generations of FBC analyzers are equipped with novel state-of-the-art laser technology which enabled them to distinguish various cell types better, faster, and more accurately [1]. New generation FBC analyzers such as Sysmex XE-5000 and Unicel DxH 800 have been amply utilized in hematological research [2-5] as they provide extended or advanced research parameters on top of the existing routine parameters present in earlier generations of FBC analyzers. Despite having their own versions of extended or advanced research parameters, such as Ret-He (reticulocyte hemoglobin equivalent) and %Hypo-He (hypochromic red blood cells) in Sysmex XE-5000, aside from low hemoglobin density (LHD), red cell size factor (RSF), and microcytic anemia factor (MAF) in Unicel DxH 800, these two analyzers have been proven to bear good correlation based on literature [6]. Sysmex XE-5000 and Unicel DxH 800 analyzers have not only been used to generate preliminary values to formulate reference intervals of healthy individuals [7,8], but also for different groups of diseases, including anemia [9], thalassemia [9], infections [10], and even onco-hematological cases [5]. Clinically, however, routine hematological findings may only vary slightly in diseases which are closely related to each other, and thus, to circumvent this, our study explored the possibility of utilizing eRBC parameters in effectively distinguishing two clinically relevant iron deficiency-related disorders namely latent iron deficiency (LID) and iron deficiency anemia (IDA).
LID has been observed to be a preceding stage prior to the development of IDA, whereby such progression may be halted by catching the condition early and providing subjects suffering from LID with proper iron supplements [11-13], thus, avoiding the derogatory risks of IDA. If left untreated,IDA has been associated with risks of delivering premature babies with low birth weights in pregnant subjects [14]. Healthy subjects have also been shown to develop other medical complications arising from IDA, some of which are common place, such as lethargy and chest pains [15], while others may be life-threatening, such as stroke [16-18]. Despite having been studied in many cases with pregnant female and not normal female subjects, there exist only a handful of parameters which involve serum ferritin coupled with hemoglobin (Hb) levels as a standard to distinguish LID from IDA. Within the multi-ethnic population of Malaysia alone, female patient data of a previous nation-wide study (n = 907) reported over 12% of both premenopausal and postmenopausal female population suffer from anemia, in which IDA was the anemia subset suffered by 8.7% and 3% of total female population who were premenopausal and postmenopausal respectively [7].
By considering the eRBC parameters obtainable from the two FBC analyzers, our current study, thus, aims to evaluate the potential utility and reliability of these parameters in distinguishing iron-deficient erythropoiesis related disorders within Malaysian female population. This study would then give a clearer diagnostic insight to better stratify and classify the iron-deficient erythropoiesis related disorders in order to improve treatment plan and patient outcome.
All clinical aspects of this study have been approved by the Medical Research Ethics Committee of the Ministry of Health of Malaysia. This study was also registered under the National Medical Research Register (Research ID 10-277-5480 and ID 17-2708-38327). Aforementioned patient consents were ensured to be received prior to data collection of respective subjects.
First phase of the study was performed by retrospective analysis on data collected during Hematological Reference Intervals in Malaysian Adults (Research ID 10-277-5480) from September 2011 - December 2012 to identify IDA and LID cases. Our research group gathered data from consenting subjects situated at Klang Valley, the states of Selangor, Negeri Sembilan, Perak, and Pahang. Patient data collection was carried out in a 16-month period throughout September 2011 to December 2012. Second phase of prospective study was conducted on eRBC parameters in LID and IDA cases referred to Hospital Ampang, Malaysia in year 2017-2018 (Research ID 17-2708-38327).
Upon receiving patient consent, blood samples were collected in two separate tubes, the first being three ml in volume within a K2 EDTA tube and the second being five ml in a plain tube. Serum was separated and subsequently stored at -80 ℃ for biochemical analysis of serum ferritin and soluble transferrin receptor.
Blood sample analysis was carried out as described in a previous study [7]. The automated hematological analyzer Sysmex XE-5000 (Sysmex, Kobe, Japan) and Unicel DxH 800 (Beckman Coulter, Miami, Florida, USA) utilized to perform FBC within 6 hours of patient sample collection which is in concordance with the International Council for Standardization in Hematology (ICSH) guideline. Blood smears were also generated for all patient samples by the use of SP1000i automated slide maker (Sysmex, Kobe, Japan). Following this, supravital brilliant cresyl blue (BCB) stains (Merck Millipore, Darmstadt, Germany) were generated to detect H inclusions while the capillary electrophoresis instrument, Capillarys 2 (Sebia, France) was utilized to perform hemoglobin analysis. All analyzers involved within this study had been maintained and stringently calibrated based on manufacturer's instructions.
The biochemical parameters highlighted within this study were quantified by the use of assays for serum ferritin (Modular E170, Roche, Switzerland) and soluble transferrin receptor (Cobas Integra 400, Roche, Switzerland). Our laboratory is enrolled in the Royal College of Pathologists of Australasia (RCPA) proficiency programs which covered full blood count, hemoglobin, and serum ferritin analysis. Quality control (QC) had been performed internally to determine precision of assays used while external QC determined accuracy of said assays.
Subjects were distinguished as normal, LID and IDA based on the FBC parameters, morphologic examinations, Hb analysis and iron studies based on our previously established reference ranges in Malaysian adults [7]. Elevated soluble transferrin receptor (above 5 mg/L) was useful in identification of LID based on our previously published study in agreement with Leonard, et al. [7,19].
Other inclusion criteria for this study include female Malaysian citizens of 18 years of age or older who are non-pregnant. Subjects who had histories of blood malignancies, renal disease, immune thrombocytopenia, and thalassemia were excluded from this study. Subjects of the three major ethnic groups of Malaysia (i.e. Malay, Chinese, and Indian) were considered from this study.
All statistical analyses in this study were carried out were carried out on the IBM SPSS Statistics 22 software (SPSS, Chicago, USA). Initially, the demographics of subjects involved within this study were analyzed. Secondly, Spearman's correlation analysis was also carried out to determine the correlation between values generated by Sysmex XE-5000 and Unicel DxH 800. Shapiro-Wilk test of normality was carried out for each of the parameters to determine their distribution. This was followed by one way analysis of variance (ANOVA) with a test of homogeneity of variance was carried out to assess differences in red cell and biochemical parameters from normal, IDA, and LID subjects. The eRBC parameters were tested in both FBC analyzers [reticulocyte hemoglobin (Ret-He), reticulocyte-Y (RET-Y), red blood cell-Y (RBC-Y), percentages of microcytes (%MicroR), percentages of macrocytes (%MacroR), immature reticulocyte fraction (IRF), high-fluorescence reticulocytes (HFR), medium-fluorescence reticulocytes (MFR), low-fluorescence reticulocytes (LFR), %Hyper-He, %Hypo-He on Sysmex XE-5000 and low hemoglobin density (LHD), microcytic anemia factor (MAF) and red blood cell size factor (RSF) on Unicel DxH 800. Welch test was also performed to confirm parameters which did not meet the homogeneity of variance. Tukey post-hoc test was carried out for parameters which were found to fulfil the homogeneity of variance were, while Games-Howell post-hoc test was carried out for parameters which did not fulfil the homogeneity of variance. Differences which yield p-values of < 0.05 would be considered as a candidate to generate ROC curves for further analysis.
Similar to the statistical analyses conducted, ROC curves were generated via IBM SPSS Statistics 22 software (SPSS, Chicago, USA). Having been demonstrated previously, the guideline to determine the diagnostic power of each parameter was based on the values of their generated area under the curve (AUC), sensitivity, and specificity. For this study, parameters which have > 0.8 AUC was considered to have strong diagnostic utility in distinguishing IDA from LID or vice versa. Cut-off points were also determined while maintaining good sensitivity and specificity values.
A total of 907 (307 Malay, 325 Chinese, 275 Indian) normal subjects and 186 subjects diagnosed with iron-deficient disorders (104 IDA, 82 LID) were considered for this study. A total of 186 patient data were considered for this LID and IDA study. Of these subjects, 104 were diagnosed with IDA while 82 were diagnosed with LID.Within Indian females, IDA (n = 58) and LID (n = 35) were more prevalent (IDA: 55.8%; LID: 42.7%) when compared to the remaining Malay (IDA: 26.9%; LID: 30.5%) and Chinese (IDA: 17.3%; LID: 26.8%) females. We did not find sufficient evidence to elucidate differences between three Asian ethnic groups in literatures; hence we combined all three Malaysian major ethnic groups for subsequent analysis.
Next, parameter of groups studied were tabulated in Table 1 alongside normal range parameters previously established by our research group [7]. Notable differences could be observed between the tabulated groups. As a prime example, IDA subjects have universally lower Hb, HCT, MCV, MCH, MCHC, Ret-He, %Hyper-He, %MacroR, RET-Y, RBC-Y, RSF, and MAF, while possessing higher IRF, HFR, %Hypo-He, sTfR, and LHD, upon comparison with normal and LID subjects. Similarly, LID subjects, despite having similar parameter values to normal subjects, also displayed notable differences, such as lower %Hypo-He and %MicroR upon comparison with normal and IDA subjects. It should also be noted that standard deviation values of the parameter ferritin are alarmingly high for all groups. The seemingly dispersed ferritin data may be attributed to the pre- or post-menopausal status of female subjects. Despite being markedly reduced in IDA and LID groups in comparison with the normal group, differences between the two disease groups were not clinically significant. IDA and LID patient data analyzed also yielded ferritin values of < 13.0 μg/l, which is not suitable for it to be a good distinguishing candidate.
Table 1: Red cell and biochemical parameter data of female IDA and LID subjects compared to normal female subjects. View Table 1
Shapiro-Wilk's test for normality displayed abnormal distributions for the parameters MCHC, RDW-SD, RDW-CV, RET, RET#, IRF, LFR, HFR, RPI, %Hypo-He, %Hyper-He, %MicroR, sTfR, ferritin, and LHD within IDA group. Abnormal distributions were also observed in the parameters RET, RET#, IRF, LFR, MFR, HFR, RPI, HYPORBC, HYPERRBC, MICROCYTES, MACROCYTES, sTfR, ferritin, LHD, and MAF within LID group. The ANOVA and post-hoc tests performed yielded significant differences in some parameters between all study groups as indicated in Table 1. All parameters showed significant differences between IDA and LID patient data are indicated with p < 0.05.
Spearman's correlation coefficient analysis was performed between Sysmex XE-5000 and Unicel DxH 800 to assess the concordance of eRBC parameters in order to identify parameters that have similar meaning clinically as the manufacturers have different designation and algorithms for these parameters. Correlation between the two FBC analyzers is shown in Figure 1, whereby RBC-Y is a Sysmex XE-5000 parameter while MAF is a Unicel DxH 800 parameter. Other parameters which show good correlation between the two analyzers include Ret-He with MAF (r = 0.906), %Hypo-He with LHD (r = 0.768), %Hypo-He with MAF (r = -0.894), %MicroR with MAF (r = -0.898), and RET-Y with MAF (r = 0.906).
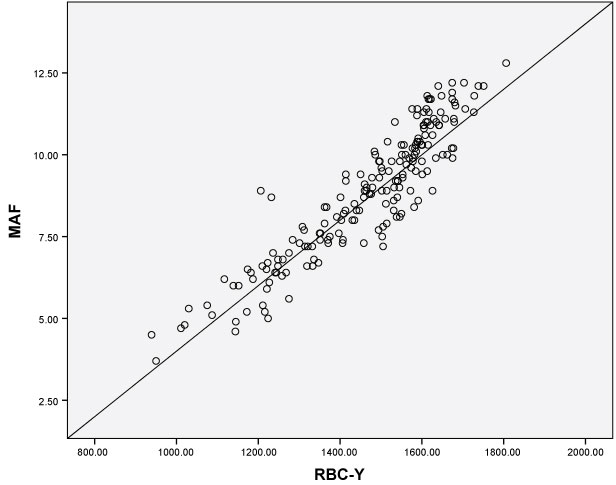 Figure 1: Spearman's correlation revealed an excellent correlation (r = 0.918, p < 0.01) between RBC-Y (Sysmex XE-5000) and MAF (Unicel DxH 800) parameters.
View Figure 1
Figure 1: Spearman's correlation revealed an excellent correlation (r = 0.918, p < 0.01) between RBC-Y (Sysmex XE-5000) and MAF (Unicel DxH 800) parameters.
View Figure 1
ROC curves were generated for only shortlisted eRBC parameters which harbored statistical differences (p < 0.05) and were deemed clinically significant. Within this result section, subsequently, only ROC curves which yielded AUC > 0.8 were displayed to emphasize on high diagnostic power of the parameters. Figure 2 and Figure 3 display the ROC curves constructed to visualize the AUC of each of the tested parameter to distinguish LID from normal subjects and LID from IDA cases respectively. Table 2 was generated to summarize sensitivity and specificity findings for all considered parameters.
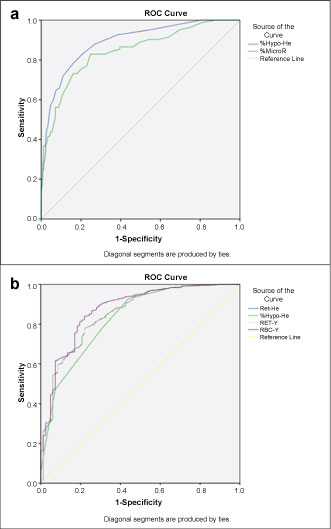 Figure 2: ROC curves of shortlisted red cell parameters to distinguish. a) LID from normal subjects; b) Normal subjects from LID.
View Figure 2
Figure 2: ROC curves of shortlisted red cell parameters to distinguish. a) LID from normal subjects; b) Normal subjects from LID.
View Figure 2
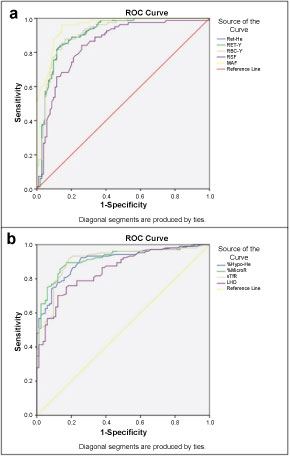 Figure 3: ROC curves of shortlisted red cell parameters to distinguish. a) LID from IDA; b) IDA from LID. Several parameters yielded excellent AUC, such as MAF to distinguish LID from IDA (AUC: 0.954; 95% CI: 0.927-0.980; Cut-off: > 9.25; Sensitivity: 89.02%; Specificity: 88.46%) and %MicroR to distinguish IDA from LID (AUC: 0.915; 95% CI: 0.874-0.956; Cut-off: > 0.655%; Sensitivity: 85.57%; Specificity: 86.58%).
View Figure 3
Figure 3: ROC curves of shortlisted red cell parameters to distinguish. a) LID from IDA; b) IDA from LID. Several parameters yielded excellent AUC, such as MAF to distinguish LID from IDA (AUC: 0.954; 95% CI: 0.927-0.980; Cut-off: > 9.25; Sensitivity: 89.02%; Specificity: 88.46%) and %MicroR to distinguish IDA from LID (AUC: 0.915; 95% CI: 0.874-0.956; Cut-off: > 0.655%; Sensitivity: 85.57%; Specificity: 86.58%).
View Figure 3
Table 2: ROC data elaboration for each red cell parameter to distinguish LID from normal subjects and IDA cases. View Table 2
Several parameters yielded excellent AUC, such as MAF to distinguish LID from IDA (AUC: 0.954; 95% CI: 0.927-0.980; Cut-off: > 9.25; Sensitivity: 89.02%; Specificity: 88.46%) and %MicroR to distinguish IDA from LID (AUC: 0.915; 95% CI: 0.874-0.956; Cut-off: > 0.655%; Sensitivity: 85.57%; Specificity: 86.58%).
Current diagnosis of LID and IDA are carried out by measuring serum ferritin and Hb levels by the means of FBC [20]. The most distinctive pattern between LID and IDA is that LID occurs as an onset of the disease progression with reduced serum ferritin, increased soluble transferrin receptor with normal Hb level [19,21]. IDA subjects, on the other hand, present with both reduced serum ferritin and Hb levels [21]. The availability of only a few routine variables to properly distinguish normal from LID and LID from IDA (e.g. Hb) became the rationale of our current study which proposed the utilization of eRBC parameters available in Sysmex XE-5000 and Unicel DxH 800 to provide differential diagnosis for LID and IDA. Detection of onset of iron depletion at early stage with the utilization of eRBC parameters will be a faster and cheaper surrogate method in the diagnosis of LID which left untreated, can progress to IDA.
Within the patient data being considered for this study, it was observed that there is higher prevalence of both IDA (55.8%) and LID (42.7%) in the Indian ethnic group as compared to the Malay and Chinese ethnic groups. This finding mirrors the trend of another population-wide study conducted in India by Alvarez-Uria, et al. which reported a whopping yet consistent 50% prevalence of anemia in female subjects upon onset of menarche [22]. With regards to the FBC analyzers,Spearman's correlation analysis of Sysmex XE-5000 and Unicel DxH 800performances depicted in Figure 1 showed that both FBC analyzers have high degree of correlation (r > 0.8) with emphasis on the eRBC parameters focused in this study. Our correlation results are in concordance with correlation analyses performed in other studies [2,6].
Prior to generating ROC curves of eRBC parameters, some of these parameters were excluded due to the clinically insignificant nature of their involvement in LID and IDA development. Parameters that were excluded within this step were IRF, LFR, MFR, HFR, %Hyper-He, and %MacroR. The biochemical parameter ferritin also displayed values which were affected by the menopausal status of female subjects [7] and thus, it is not a suitable parameter to distinguish LID from IDA in a Malaysian female population as a collective regardless of their menopausal statuses. A final total of five Sysmex XE-5000 and three Unicel DxH eRBC parameters and soluble transferrin receptor were reported to hold excellent diagnostic utility within the scope of this study. MAF, which is a Unicel DxH 800 parameter yielded the best AUC to distinguish LID from IDA above the cut-off point of 9.25 (AUC: 0.954; 95% CI: 0.927-0.980; Sensitivity: 89.02%; Specificity: 88.46%). In a similar study conducted by Dopsaj, et al. MAF was shown to effectively distinguish iron-deficient athletes from healthy subjects [23], which support our findings. As a Sysmex XE-5000 parameter, %MicroR also generated the best AUC to distinguish IDA from LID above the cut-off point of 0.655% (AUC: 0.915; 95% CI: 0.874-0.956; Sensitivity: 85.57%; Specificity: 86.58%). Despite being classified based on red cell sizes into macrocytic, normocytic, and microcytic anemia, the diagnostic utility of microcytes within this study strongly indicate that most of the iron deficiency anemia considered within female subjects are microcytic anemia, which is often associated with iron deficiency [24], which further explained our findings. This study also establishes other effective red cell parameters to distinguish LID from IDA or vice versa as shown in Table 2. The biochemical parameter, serum transferrin receptor (sTfR) also generated promising AUC value in distinguishing IDA from LID above the cut-off point of 5.45 mg/l (AUC: 0.909; 95% CI: 0.865-0.953; Sensitivity: 84.61%; Specificity: 85.36%).
The utility of eRBC parameters is not only limited to distinguishing iron-deficiency related disorders, as shown in a similar study by Torino, et al. [25] which reported %Hypo-He and %MicroR as a parameter of interest in distinguishing anemia of chronic disease (ACD) from IDA Other research groups have also considered the utilization of various red cell parameters in characterizing hematological diseases, such as non-sickling hemoglobinopathy [26], chronic nephropathies [27], and even mortality in subjects undergoing hemodialysis [28]. In the future, simple algorithms may even be formulated by arithmetically incorporating more than one routine or eRBC parameters to achieve better distinction between 2 conditions of interest with the aid of ROC curves.
The advent of newer generations of FBC analyzers has unveiled possibilities to utilize such technology in providing a surrogate first-line indication of iron deficiency-related disorders, especially in facilities which lack resources or access to gold standard diagnostic methods. Our study considered the data of 186 Malaysian female subjects suffering from LID and IDA collectively, whereby no clinically significant differences were observed in different ethnic groups. By employing single eRBC parameters obtainable from Sysmex XE-5000 and Unicel DxH 800, our study was able to isolate several parameters by statistical mean comparison and ROC curve analysis which were effective in distinguishing the IDA from LID or vice versa. Fulfilling our study objective, we report the functional utility of the eRBC parameters Ret-He, RET-Y, RBC-Y, RSF, MAF, %Hypo-He, %MicroR, LHD, and the biochemical parameter sTfR in distinguishing LID from IDA or vice versa. These parameters are extremely useful in aiding the physicians to recognize iron depletion in apparently healthy women in order to initiate further investigation and timely treatment before the onset of IDA. Our findings need to be verified in a larger cohort in other subsets of population on risk of developing IDA.