The balance of neutrophilic and lymphocytic populations in the body is sensitive to neuroendocrine changes present in acute and chronic emotional stress, influencing a range of behaviors intrinsically related to real or potential environmental stressors. In this sense, the Neutrophil/Lymphocyte Ratio (NLR) is a simple and low-cost tool, derived from the analysis of the blood count, capable of showing changes in several health parameters, since it is related to the pro-inflammatory status of the organism. In this article, through a narrative review of the literature, the applicability of NLR in behavioral studies and its possible clinical uses are presented, both at the diagnostic, prognostic and treatment levels. The physiology of NLR and its relationship with stress, as well as topics such as mental health, obesity, eating behavior, smoking and physical activity are mentioned, NLR appears to be an accurate, useful and promising biomarker for use in monitoring and preventing diseases in health, with an emphasis on mental health. Its study becomes very important for the determination of reference values and understanding of its working mechanisms, which still demands further investigation.
Blood count, Mental health, Psychobiology, Neuroimmunomodulation
The human Immune System is composed of several cell populations that, in their different functions, make up an arsenal of mechanisms active in acute and chronic inflammatory processes [1,2]. Such inflammatory processes are particularly present in behavioral changes, especially those resulting from emotional stress [3-5].
Two of the cell populations most affected by stress are neutrophils and lymphocytes [6]. Both respond to increased serum glucocorticoid concentrations, especially cortisol [7,8]. Therefore, the examination of the ratio between circulating Neutrophils and Lymphocytes (RNL), performed through the evaluation of the leukogram, can provide important information on behavioral changes, especially those associated with the inflammatory process.
Increased NLR has been associated with acute episodes of mania in patients with Bipolar Mood Disorder [9,10], being a possible marker of suicidal behavior in such psychopathology [10,11]. Its relationship has been further investigated for psychotic episodes in schizophrenic patients [12,13], in addition to a number of other pathophysiological conditions relevant to mental health, such as Alzheimer's Disease [14], Major Depressive Disorder [15], and Attention Deficit Hyperactivity Disorder [16]. Such conditions have the similarity of being, to a greater or lesser degree, associated with dysregulation in the Hypothalamus-Pituitary-Adrenal (HPA) axis and with neuroinflammation contexts [17,18].
In addition to psychopathology, NLR has been explored in behavioral contexts associated with obesity [19,20], eating behavior and physical exercise [7,21,22]. High abdominal fat and dietary restriction have been associated with elevations in NLR [19,20]. High-intensity and long-term physical exercise has also been responsible for modulating the populations of circulating neutrophils and lymphocytes [7,21,22]. Other conditions, such as those involving lifestyle, such as smoking [19,23,24] and social stress [25,26] has also drawn the attention of scholars on the subject.
Despite the varied themes of study, there is still a lack of concrete subsidies about the physiology of NLR and the motivations for which behavioral changes affect it. Thus, the present paper is a literature review in which topics related to NLR and its applicability in behavioral studies will be addressed. In this way, we intend to expand the understanding and evaluate the possibility of using this marker in the practice of studies in psychobiology and possibly in clinical behavior.
During the month of December 2021, two independent researchers performed a search on the PubMed platform using descriptors such as "Behavior', "Food behavior and obesity", "Stress", "Psychopathology", "Suicidal behavior" and "Physical exercise", plus of the term "Neutrophil/Lymphocyte Ratio". Articles from 1980-2022 were considered, without limitation of language, audience or type of study.
In all, 167 articles were selected, and those that were closely related to the topic were selected, that is, those that addressed the Neutrophil/Lymphocyte Ratio as a marker of behavioral states. A total of 32 articles were included in the review.
The number of articles selected by descriptor was: Food behavior and obesity: 9.4%; Stress: 31.2%; Psychopathology: 43.7%; Suicidal behavior: 6.2%; Physical exercise: 9.4%. The data are presented in Figure 1 and Figure 2.
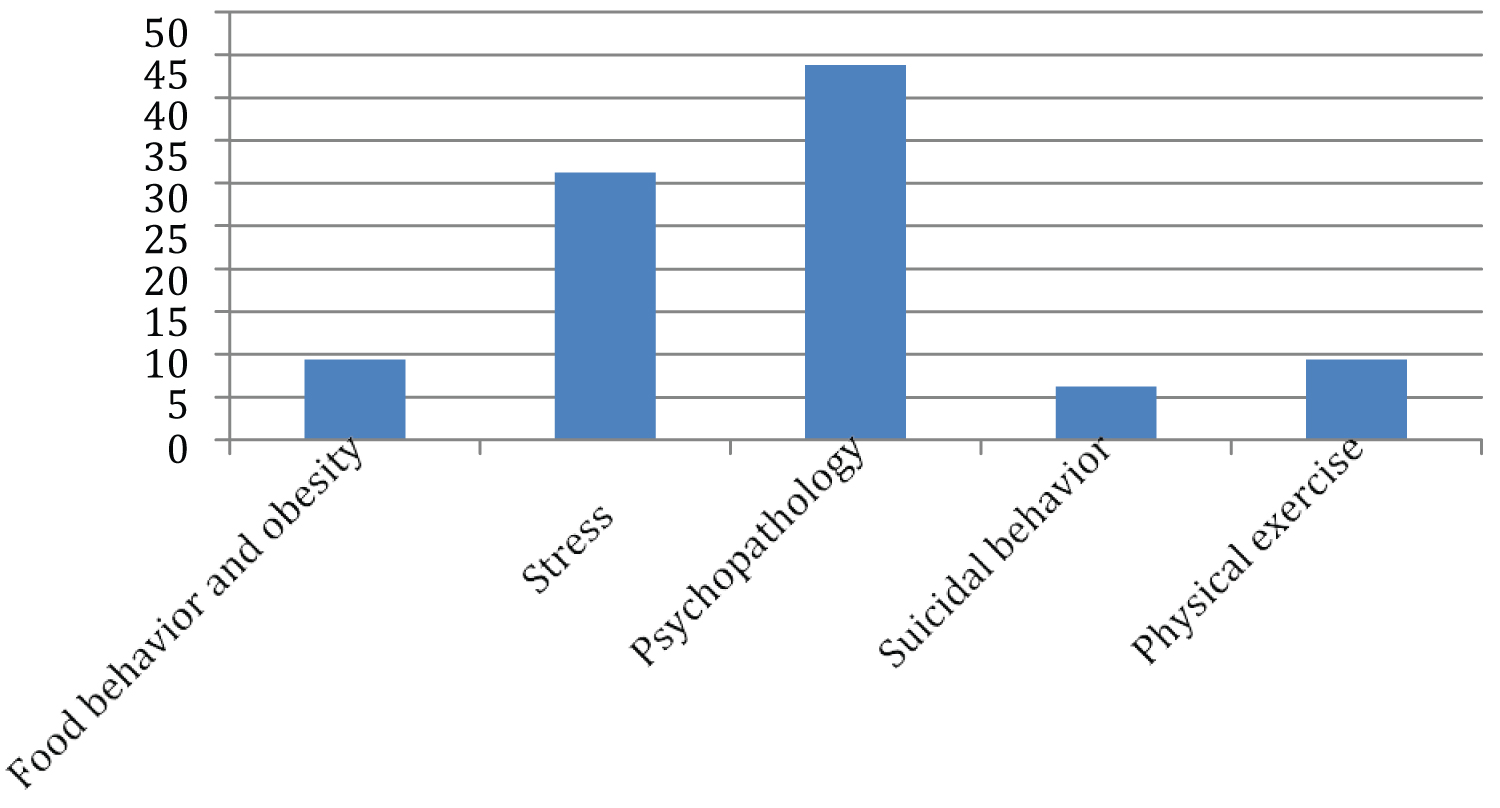 Figure 1: Percentage of articles surveyed by descriptors.
View Figure 1
Figure 1: Percentage of articles surveyed by descriptors.
View Figure 1
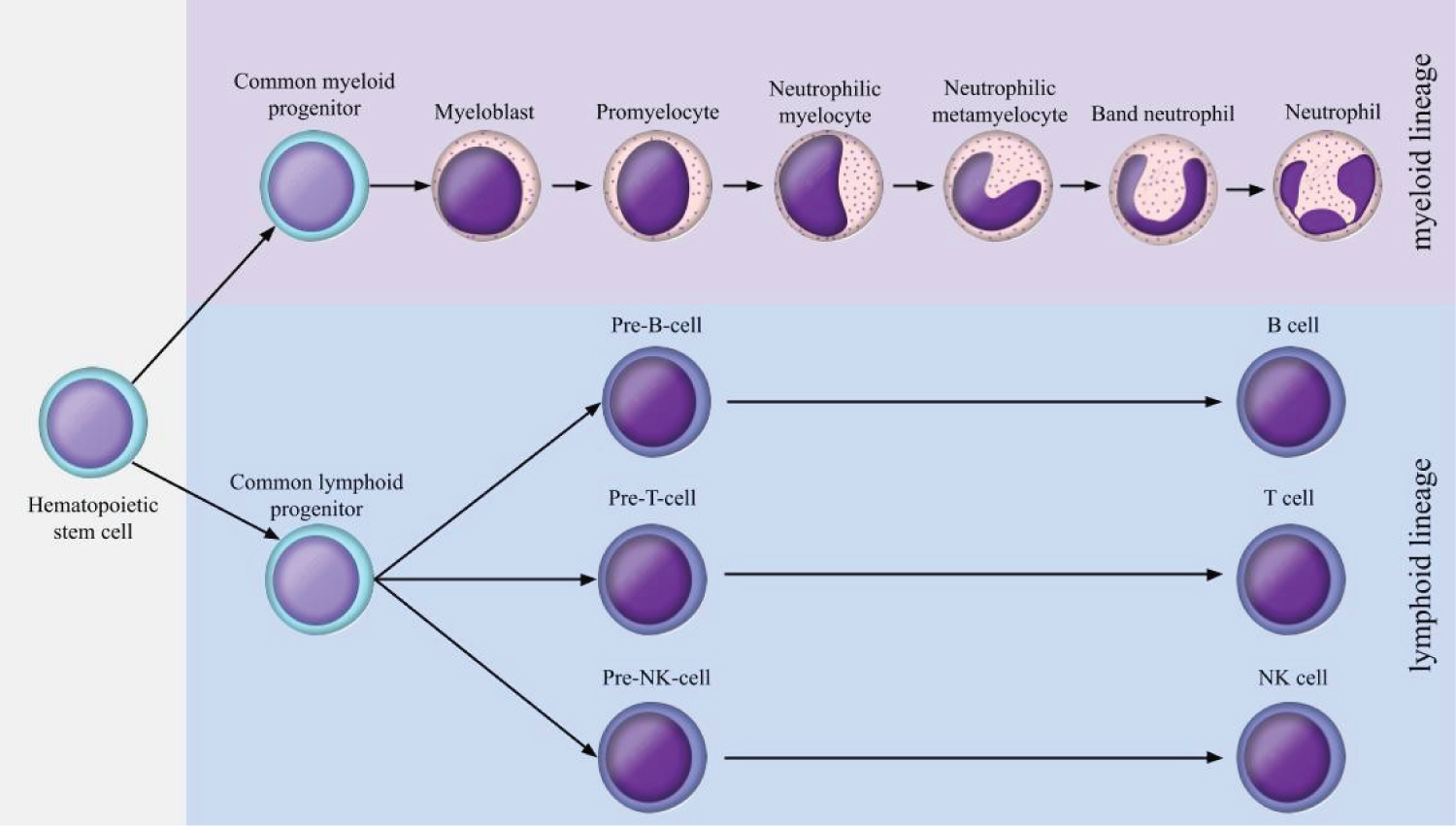 Figure 2: Synthesis and maturation of neutrophils and lymphocytes.
View Figure 2
Figure 2: Synthesis and maturation of neutrophils and lymphocytes.
View Figure 2
All blood cells originate from pluripotent hematopoietic stem cells [27]. Such cells generate the lymphoid cell lineage, which gives rise to lymphocytes, and the myeloid cell lineage, which gives rise to erythrocytes, monocytes, platelets and granulocytes (eosinophils, basophils and neutrophils) [28].
The synthesis of neutrophils occurs in the bone marrow, being stimulated by G- CSF (Colony Stimulating Factor), a hematopoietic cytokine produced by fibroblasts, endothelial cells and macrophages [29]. The neutrophil maturation process has six distinct stages, which are: myeloblast, neutrophilic promyelocyte, neutrophilic myelocyte, neutrophilic metamyelocyte, rod-nucleated neutrophil and segmented neutrophil [28,30].
Neutrophils, once in the bloodstream, mediate the initial phases of inflammatory reactions, and present cytoplasmic granules and receptors important for their functions. The primary granules, also called azurophiles, have the enzyme myeloperoxidase, which is an acid hydrolase, a type of defensin, which has a function analogous to antibodies, and the antimicrobial peptide cathelicidin, associated with the elimination of pathogens. The specific, or secondary, granules contain several types of enzymes and antimicrobial peptides [27-29]. Tertiary granules have two types, one of these types has phosphatases, while the other type has metalloproteinases, which facilitate the migration of neutrophils to connective tissue [28]. Furthermore, during the inflammatory process, neutrophils secrete certain factors, such as cathepsin G and azurocidin, chemoattractant molecules that act in the recruitment of monocytes/macrophages [31-33].
Multipotent lymphoid cells that express the transcription factor GATA-3 become T lymphocytes. Such cells leave the bone marrow as pro T lymphocytes for the thymus, where their complete differentiation occurs. In contrast, the transcription factor Pax5 activates specific genes that cause the transformation of multipotent lymphoid cells into B lymphocytes, with the function of producing immunoglobulins [28].
T lymphocytes are subdivided according to the antigen expressed on their membranes. Cytotoxic lymphocytes or T8 express the CD8 antigen on the membrane, while helper lymphocytes, or T4, express the CD4 antigen, and can also be divided into T helper 1, sensitive to IL-2 secretion, and T helper 2, the secretion of IFN-γ and/or IL-4. There are also regulatory lymphocytes that express CD25 and FoxP3, in addition to the CD4 antigen [34]. CD8+ T cells have the ability to recognize antigens on any nucleated cell type, becoming activated in the process to kill the target cell. CD4+ helper T cells recognize antigens, which lead to their activation and production of macrophage activating molecules [29].
Regarding Natural Killer (NK) cells, they differentiate under the influence of IL-2 and IL-15 interleukins into immature pre-NK cells, becoming mature cells after acquiring effector functions [28]. NK cells have the function of eliminating virus-infected cells and tumor cells by inducing apoptotic events as a result of the combined action of perforins and granzymes, present in their cytoplasmic granules. In addition, NK cells also produce IFN-γ, an antiviral agent that enhances the ability of macrophages to kill pathogens that have been phagocytosed [28,28].
Emotional stress has two major response pathways, acute and chronic [35-37]. In acute stress, upon hypothalamic activation, there will be an intense release of a special type of monoamines, the catecholamines (noradrenaline and adrenaline, and, in specific situations, acetylcholine), synthesized in brain regions (acetylcholine) and in the adrenal glands (noradrenaline and adrenaline), through activation of the Autonomic Nervous System (ANS) [35,36]. Once in the bloodstream, such catecholamines will be responsible for mobilizing organic responses to an actual or potential stressor stimulus [36].
Its effects will depend on which portion of the ANS is activated. In general, the main response to acute stress is the sympathetic pathway, mediated by noradrenaline and adrenaline, causing tachycardia, increased respiratory rate, pupil dilation, increased glycolytic metabolism, inhibition of the gastrointestinal tract, among other effects [36]. However, parasympathetic activation may occur, which will promote inverse effects to those obtained in the sympathetic response, being mediated by the monoamine acetylcholine, and responsible for freezing behaviors [37,38].
Adrenaline and noradrenaline have the potential to temporarily stimulate immune response mediators [39], increasing the synthesis of cytokines, acute response proteins and lymphocyte proliferation [40]. There is an increase in the population of neutrophils and NK cells [41-43]. Concomitantly, interleukin-1 (IL-1), Tumor Necrosis Factor α (TNF-α), Interferon-α (IFN-α) and Interferon-γ (IFN-γ), from lymphocyte secretion, induce release adrenal catecholamines. It is noteworthy that IL-1 has mimetic effects to neurotransmitters capable of triggering the activation of the Hypothalamus-Pituitary-Adrenal Axis (HPA) [44,45].
As stress becomes chronic and persistent, neuroendocrine responses predominate [35,46,47]. These are initiated by the activation of the hypothalamus. The hypothalamus secretes Corticotropin-Releasing- hormone (CRH). CRH stimulates the adenohypophysis to release Adrenocorticotropic Hormone - ACTH, which in the bloodstream, mobilizes the release of glucocorticoids by the adrenal cortex, the main one being Cortisol [5]. This will try to ensure the maintenance of homeostasis even in physiological conditions of resistance to a stressor stimulus [5]. Some of the strategies for this are: Maintenance of vascular tone, increased lipolysis in adipose cells, increased mobilization of glycerol and fatty acids, increased gluconeogenesis, glycogenesis and hepatic glycogen storage, mobilization of amino acids in skeletal muscle tissue, protein degradation and amino acid utilization in gluconeogenesis, inflammation inhibition, and immunosuppression [4,5].
In this sense, attention is drawn to the role of cortisol on immunity [4-6,48]. In studies involving glucocorticoids on the immune system and blood count parameters, it is observed that cortisol, due to its ability to alter gene transcription, affects the circulation and activity of lymphocytes, especially T helpers (the Th1 subpopulation, due to suppression of IL-2, which is the most affected) and NK cells. The effect of this process is global immunosuppression [4-6,48].
In addition, glucocorticoids, when in high serum concentration, also depress the concentration and activity of macrophages, causing a reduction in cytokine synthesis and decreasing granulocyte migration [49,50]. Therefore, while acute stress has an immunostimulatory effect, chronic stress has an immunosuppressive effect [4-6,48].
Cortisol, from the induction of lipocortin synthesis, inhibits phospholipase A2, and thus the synthesis of arachidonic acid. This prevents the supply of precursors of leukotrienes and prostaglandins, responsible for inflammatory responses [51,52]. Thus, cortisol, in addition to the depression of proliferation and activity of T lymphocytes and IL-2, also inhibits the populations of mast cells, platelets, in addition to histamine and serotonin [53-55].
Based on the above, it is observed that cortisol is a chronic stress, which favors an increase in the population of neutrophils and a reduction in lymphocytes, and thus, increases the NLR index [8]. As will be shown below, several comparison studies involving populations exposed to some type of chronic stressor have attested to this.
There are several studies that have shown an association between the increase in NLR and psychopathologies. This has been observed for patients with Schizoaffective Disorder and Schizophrenia [12], Attention Deficit Hyperactivity Disorder [16], Obsessive Compulsive Disorder [56], Hypomania in Bipolar Mood Disorder [10] and Major Depressive Disorder [57,58].
In depressive patients, Li, et al. [57] observed that the NLR correlated with levels of depression (r = 0.634, p < 0.05), and, after a flow cytometry study, they noticed that the reduction of lymphocytes was mainly from the CD3 and CD4 subpopulations. Even after drug treatment, these subpopulations remained as before, however, the NLR decreased (p < 0.05).
The effect of reducing NLR as depressive symptoms improve was also observed in other studies, being more specific to say that NLR was directly associated with increased severity of symptoms. Arabska, et al. [15] observed that NLR is higher in patients with more severe symptoms compared to the group with milder symptoms, before medication use (2.10 ± 2.13 vs. 2.01 ± 0.75, p = 0.004). In this same study, it was considered that a RNL was higher in patients with a first depressive episode, when compared to recurrent depression (2.11 ± 1.76 vs. 1.64 ± 1.04, p < 0.05).
Steiner, et al. [57] observed that, prior to psychotic crises in schizophrenic patients, the number of neutrophils was higher compared to the control group (p < 0.001), while the number of eosinophils was lower (p = 0.021). Normalized values of neurophils decreased in relation to the response to treatment (ρ = 0.186, p = 0.015), while those of eosinfolios increased ρ = -0.161, p = 0.036).
Karatas, et al. [56] noted that the NLR was increased in patients with Obsessive Compulsive Disorder before the diagnosis was received (p < 0.001), and that this index remained increased in patients already diagnosed and who had disease crises, suggesting that the NLR has prognostic value and monitoring for patients with different psychopathologies.
Still regarding gravity, Agglia, et al. noted that individuals with high lethality suicide attempts had a higher number of neutrophils (6.35 ± 3.07 vs. 5.23 ± 2.47 vs. 4.87 ± 2.16, p < 0.001), lower number of lymphocytes (1.81 ± 0.69 vs. 2.16 ± 0.81 vs. 2.37 ± 0.86, p < 0.001) mean platelet volume (8.99 ± 1.22 vs. 7.92 ± 1.25 vs. 8.00 ± 1.14, p < 0.001), NLR (4.00 ± 2.72 vs. 2.62 ± 1.31 vs. 2.21 ± 1.02, p < 0.001), platelet-lymphocytes ratio (157.83 ± 76.71 vs. 126.16 ± 59.86 vs. 110.94 ± 50.63, p < 0.001), in relation to both those with low-lethal suicide attempts and the control group. Ucuz, et al. [59] also noted a direct relationship between NLR and the occurrence of suicide, when they observed an increase in the inflammatory profile in blood counts of patients prior to committing fatal self-injury (p < 0.001).
Thus, an important reflection to be raised is: is NLR a good predictor of mental health changes? Several studies already presented in this review suggest that it is. Furthermore, Ahmed, et al. [60] noted increased NLR in patients who developed mental health complications after severe episodes of COVID-19 (5.26 ± 2.76 vs. 3.45 ± 3.73 vs. 1.71 ± 1.13; p < 0.0001).
Interestingly, having high NLR seems to favor clinical improvement in depressed patients. Llorca-Bofí, et al. [61] found a positive correlation between NLR and patients' positive clinical response (r = 0.41; p = 0.003). This association was maintained in women who received antidepressant treatment with tricyclics and norepinephrine and serotonin reuptake inhibitors, antipsychotic treatment with olanzapine/quetiapine, and in those who did not receive electroconvulsive therapy. Clomipramine, a tricyclic antidepressant, was responsible for reducing NLR in dogs after transport stress (p < 0.05) [62].
The relationship between NLR and changes in cognitive functions is still unclear. Aykut, et al. [9] were unable to find patterns regarding cognitive dysfunction in patients with TBH and NLR. In an extensive literature review, Sayed, et al. [14] suggest that further studies are important in investigating the topic, given that, for example, in Alzheimer's disease, inflammatory bases are known and thus the NLR emerges with potential in terms of diagnosis, prognosis and clinical management in coping with the disease of the disease.
A question that may arise is: In the case of diseases that directly affect the NLR, what is its predictive value in relation to mental health? Yi, et al. [63] investigated patients with diffuse large B-cell lymphoma and observed that depressed patients, even with a hematologic malignancy, had increased NLR values compared to non-depressed patients (p < 0.05).
Something very important in terms of clinical applicability is the determination of reference values. In this sense, an important question arises: What is the value of NLR for mental health prediction? Despite being scarce, some studies showed significant cut- off points in mental health assessment. Velasco, et al. [64] suggest NLR cut-off ≥ 1.3 for discrimination of suicidal behavior in patients with Major Depressive Disorder (sensitivity 75% and specificity 35%). Sunbul, et al. [65] propose that NLR ≥ 1.57 could be indicative of severe or very severe depression for symptomatic patients (sensitivity 61.4% and specificity 61.2%). Regarding hospitalized elderly patients, the NLR value ≥ 3.636, with a sensitivity of 75.2% and specificity of 63.4%, was considered ideal for predicting psychotic delusions [66].
Studies linking NLR with eating behavior and obesity are still scarcer, however, the results are promising.
Regarding the Body Mass Index (BMI), Furuncuoğlu, et al. [19] found no association between BMI and NLR, but with other markers from the blood count, such as global leukocyte count (p < 0.01), lymphocytes (p < 0.05), platelet distribution width - PDW (p < 0.05), and red blood cell distribution width - RDW (p < 0.05).
Confirming the above results, Yu, et al. [20] found a direct association between WBC count and the prevalence of visceral obesity (p < 0.001) and increased risk of cardiovascular disease (p < 0.001) in overweight Korean adults.
Poor diet quality, which is one of the factors influencing body weight gain, is also associated with NLR [67]. In this study, it was found that Spanish adults (≥ 50 years) with NLR ≥ 2.6 had a lower score on the Global Food Score (GFS), an index related to diet quality, compared to individuals with NLR values < 2.6 (22, 65 ± 6.26 and 23.56 ± 6.41, respectively) (p < 0.05).
The smoking habit is related to the systemic pro-inflammatory profile [24,68]. Thus, studies have pointed to the existence of a positive relationship between the use of cigarettes and the increase in NLR. Gumus, Solak and Eryilmaz [69] studied the NLR and some ratios derived from the complete blood count (RPL and Mean Platelet Volume/Platelet Ratio - MPV/PLt) and found that smokers had significantly higher NLR (2.10 ± 1.42 vs. 1.87 ± 0.83). p = 0.031), in addition to higher NPV/PLt (109.76 ± 90.77 vs. 118.66 ± 40.20, p = 0.001).
The increase in NLR in smokers was also observed by Tulgar, et al. [23]. The authors pointed out that in smokers, several blood count indices in addition to the NLR (leukocyte, neutrophil, basophil and eosinophil count; Mean Corpuscular Volume - MCV, RDW) were significantly higher when compared to the non-smokers group (p < 0.05). They also found a positive linear correlation between the number of cigarette packs consumed per year with the RNL and the MPV (p < 0.05).
However, the NLR seems to be a versatile and flexible marker to the conditions of the environment. Individuals who definitively stopped smoking had a reduced NLR when compared to smokers (before: 1.8, after: 1.7; p < 0.05) [24]. This study also confirms the elevated NLR in smokers, corroborating previous studies.
It is noteworthy that studies involving NLR and smoking seem to point to the consensus of increased NLR in smokers and as tobacco consumption increases, however, other relationships, such as those derived from platelet counts, still need to be elucidated by the scientific community.
Studies linking NLR and physical activity practice are scarce. Those who bring it up suggest that the practice of high-intensity and maximum effort exercises have the potential to increase NLR [7,21,22]. In a study with horses under extreme physical exercise, Cywińska, et al. [21] suggested that exercise-induced stress resulted in neutrophilic leukocytosis and lymphopenia, as well as an increase in NLR.
NLR, however, was not responsive when physical exercise is not high-intensity or is combined with other interventions, such as food-based ones. Renzo, et al. [70] evaluated a protocol containing a personalized Mediterranean diet and physical activity as therapy in reducing cardiovascular risk markers in Italian women. Several cardiovascular risk markers (Atherogenic Plasma Index (AIP), Lipid Accumulation Product (LAP) and Fatty Liver Index (FLI)) were found to decrease after the intervention, but NLR and RPL did not have significant reductions. This seems to suggest that the pro-inflammatory stimuli may not have been stopped, and thus no responding NLR was found. The cessation of stimuli that generate inflammation appears to be essential for the reduction of NLR.
It is plausible to conceive that physical activity, due to its initial increase in cortisol levels, local inflammation, and autonomic activity, will not contribute, in an acute response, to the fall in RNL, but perhaps this occurs in a chronic way, due to the ability to healthy modulation of serotonergic, dopaminergic and endogenous opioid pathways, generating benefits for improving mood, well-being and quality of life [71-73].
Several other questions also intrigue researchers regarding NLR and behavior. To answer some of them, Penz, et al. [26] investigated the kinetics of leukocyte response to stressful life events. They proposed that such events trigger immunological changes that persist over time and thus promote a continuous effect on leukocyte distribution. The authors showed that changes in neutrophil populations were related to changes in capillary cortisol, which suggests a close relationship between the neuroendocrine and immune systems.
In 1998, Zuri, Gottreich and Terkel suggested elevated NRL in subordinate rats in dominance hierarchy relationships. Among the explanations for the phenomenon are the facts that subordinates receive greater stress vis-à-vis their dominants, since preferential access to food and physical violence against subordinates make up the dominance behavioral repertoire, as a way of affirming the social relationship.
The NLR has wide applicability of studies in psychobiology and behavior of different orders. It is fair to say that his study is interesting for applied areas, such as psychiatry and multidisciplinary mental health assessment. In addition, the NLR, as a marker of inflammation, can provide a psychophysiological view of the stress to which the individual is inserted and its modulation in the face of the modification of stress eliciting stimuli.
Not only the NLR presents itself as a marker of interest for studies on behaviors. The investigation of platelets and their reasons also seem very promising as a perspective for future studies.
That the potential of these lines of study be transformed into research with high methodological rigor, in order to offer substantially reliable results that can be clinically used and scientifically corroborated. In this review, it became evident that some topics still need to be studied, in order to improve understanding and glimpse the infinity of relationships between the nervous, endocrine and immune systems.
The authors report no conflict of interest.
Together, the authors declare that they all contributed to the present work and agree with its submission.