Spontaneous echo contrast (SEC) in the left atrial appendage (LAA) is associated with an increased risk of cerebral embolism in patients with atrial fibrillation (AF). Novel oral anticoagulants (NOACs) are increasingly prescribed as first-line therapy and have shown a comparable benefit-risk ratio compared to vitamin K antagonists (VKAs). The aim of this study was to investigate whether NOACs decrease the incidence of SEC compared to VKA.
We retrospectively analysed 542 patients who underwent transoesophageal echocardiography (TEE) prior to AF ablation at our institution. SEC was characterised by dynamic, smoke-like echoes within the LA cavity or LAA on TEE.
SEC was detected in 58 patients (10.7%) and occurred significantly more often in patients on VKA (n = 39; 13.6%) compared to patients on NOACs (n = 19, 7.5%; p = 0.02). Decreased left ventricular function (p < 0.001), arterial hypertension (p < 0.001), LA enlargement (p < 0.001) and AF during TEE (p < 0.001) were significantly more often observed in patients showing SEC. Transient ischemic attack or stroke did not occur in any patient during AF ablation.
The incidence of SEC on TEE in patients prior to AF ablation was significantly lower in patients treated with NOACs as compared to patients treated with VKA. Patients with multiple risk factors showed a higher SEC incidence. The results support the recommendation of NOACs as first-line oral anticoagulation in non-valvular AF.
Spontaneous echo contrast, Atrial fibrillation, Transoesophageal echocardiography
A recently published article indicates no correlation between periprocedural thromboembolic events and spontaneous echo contrast (SEC) in patients undergoing catheter ablation of atrial fibrillation (AF). However, during long-term follow-up, patients with preprocedurally detected SEC showed significantly more strokes/transient ischemic attacks (TIAs) [1]. Additionally, SEC in the left atrial appendage (LAA) is associated with an increased risk of cerebral embolism in patients with AF, even if sufficiently anticoagulated [2].
During past years, new agents other than the established vitamin K antagonists (VKA) entered the arena of oral anticoagulation-the so-called novel oral anticoagulants (NOACs). Previous reports have stated the clinical effectiveness of NOACs in patients with non-valvular AF [3-6]. Due to good compliance and effectiveness, the use of NOACs is increasing, but clinical experience is still limited [7]. In the present study, we investigated whether patients under NOACs exhibit a lower incidence of SEC compared to patients under VKA.
Our retrospective analysis included all patients who underwent catheter ablation for paroxysmal or persistent AF at our centre from January 2013 to December 2014 and who received transoesophageal echocardiography (TEE) to exclude intra-atrial thrombus. All patients were examined for SEC. SEC was characterised by dynamic, smoke-like echoes within the LA cavity or LAA during TEE [8]. Patients taking VKA who were not in the therapeutic window (INR between 2.0 and 3.0) were excluded from the analysis.
Two-dimensional (2D) and three-dimensional (3D) TEE were performed with a commercially available ultrasound system (Phillips iE33; Philips Medical Systems, Andover, MA, USA) using a matrix-array 3D TEE probe (X7-2t; Philips Medical Systems). All TEEs were performed within 24 h prior to the scheduled procedure. SEC was defined as dynamic, smoke-like echoes with swirling motion using optimal gain setting during the cardiac cycle. Thrombus, on the other hand, was defined as a circumscribed and uniformly echo-dense intracavitary mass distinct from the underlying LA or LAA endocardium [8].
Data analysis was performed using the software package SPSS for Windows versions 23.0 (IBM Corp., Armonk, NY, USA). Two sample t-tests were performed in the case of normal distribution of the variables, and Mann-Whitney-Wilcox tests were performed in the case of the remaining distributions. A linear regression analysis was conducted to determine the relationship between the SEC and selected variables (risk factors). For categorical outcomes including relative frequencies, group comparisons were performed using chi-squared tests. For quantitative measures, means and standard deviations for symmetrically distributed data or medians and ranges (minimum to maximum) for skewed data were shown.
A total of 542 patients were included in the study (VKA, n = 287; NOAC, n = 255). Within the NOAC group, 18.4% of patients (n = 47) received dabigatran, 22% (n = 56) apixaban, and 59.6% (n = 152) rivaroxaban. A reduced dose of the NOACs was administered in 11% (n = 28) of patients. No intra-atrial thrombus was found in this population.
SEC was significantly more often present in patients on VKA (13.6%) as compared to patients on NOACs (7.5%) (p = 0.02; Table 1; Figure 1).
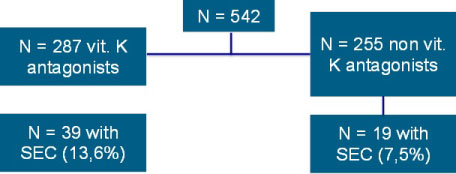 Figure 1: Incidence of SEC (spontaneous echo contrast) in patients on vitamin K-antagonists and NOACs (novel oral anticoagulants).
View Figure 1
Figure 1: Incidence of SEC (spontaneous echo contrast) in patients on vitamin K-antagonists and NOACs (novel oral anticoagulants).
View Figure 1
Table 1: Differences in baseline characteristics in patients with or without SEC. View Table 1
Decreased left ventricular function (p < 0.001), arterial hypertension (p < 0.001), LA enlargement (p < 0.001) and AF during TEE (p < 0.001) were significantly more often found in patients exhibiting SEC (Table 1 and Table 2; pages 12 and 13).
Table 2: Differences in baseline characteristics in patients with or without SEC. View Table 2
The number of previous ablation procedures was not associated with a higher SEC incidence (p = 0.53), but we found a significantly higher SEC incidence in patients with persistent AF versus patients with paroxysmal AF (p < 0.001; Figure 2 and Figure 3; page 15).
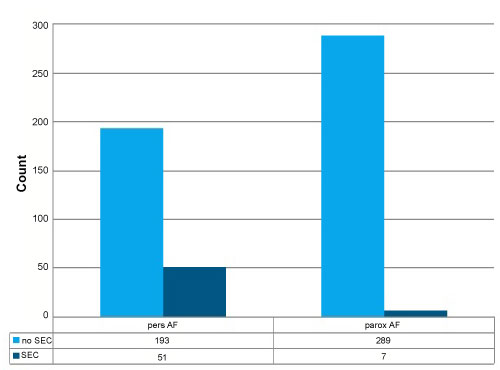 Figure 2: Count of patients with paroxysmal versus persistent AF in context of prevalence of SEC.
View Figure 2
Figure 2: Count of patients with paroxysmal versus persistent AF in context of prevalence of SEC.
View Figure 2
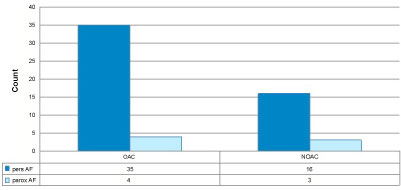 Figure 3: NOAC versus vitamin K antagonists (OAC) in patients with SEC in context of prevalence of pers. and paroxysmal AF.
View Figure 3
Figure 3: NOAC versus vitamin K antagonists (OAC) in patients with SEC in context of prevalence of pers. and paroxysmal AF.
View Figure 3
Logistic regression (Table 3, page 13) demonstrated a clinically relevant increased risk for SEC by age (5.4%/year), total duration of persistent AF (1.7%/month), number of previous cardioversions (22.3%/each), LA diameter (6.5%/mm) and CHADS2 score (6.5%/point). We also found a significant correlation of the CHADS2 and CHA2DSc-VASc scores with the incidence of SEC (p = 0.01/p < 0.01, shown in Table 2, page 12).
Table 3: Logistic regression (IV: age, total AF-duration in months, persistent AF duration in months, number of previous cardioversions, LV-function, LA-diameter, CHADS2 score; dependent variable. View Table 3
The main finding of the study is that patients on NOACs reported significantly less SEC on TEE compared to patients taking VKA in the therapeutic window. All included patients under VKA have been in the target INR range (between 2 and 3). However, it is expected that patients in the lower INR range have a higher risk for SEC than those in the upper target range. In addition, nutritional factors can also lead to strong fluctuations. In terms of safety in thromboembolic and bleeding events, NOACs have been proven to reduce SEC and, thus, the risk of late strokes [3,4,6,9] (Table 4 and Table 5).
Table 4: Chi-square test (independent variable, IV): type of atrial fibrillation (parox/pers), Vitamin-K antagonists versus non-vitamin K antagonists, rhythm at TEE, hypertension; dependent variable: spontaneous echo contrast (SEC). View Table 4
Table 5: Distribution of NOACs. View Table 5
Periprocedurally, we did not encounter any stroke or TIA in our patient group, which is consistent with the work of Gedikli, et al. [1] SEC in LA/LAA, therefore, does not seem to be a contraindication for ablation or cardioversion either. Our findings confirm the results of Di Biase, et al., that TEE before ablation is not absolutely necessary, especially under sufficient NOAC intake [9-12].
There are reports that during long-term follow-up, patients with preprocedurally detected SEC report significantly more strokes or TIAs [1]. It might be speculated that a change from VKA to NOACs preprocedurally might reduce the frequency of TIA and stroke in the long term. A postprocedural switch could also be discussed in patients on VKA and SEC, although no definite recommendations are possible from ours and other's data.
Impaired left ventricular function, arterial hypertension, AF during TEE, and an enlarged LA were identified as predictors of SEC. These results are consistent with previously published studies and are also included in the CHA2DS2-VASc score [13-15]. An improvement in the left ventricular function caused by optimal medical treatment of heart failure as well as a therapy of arterial hypertension according to the guidelines can reduce the SEC risk mentioned above [16].
Interestingly, the number of previous AF ablations in our cohort was not associated with an increased SEC incidence. Data here are not consistent. The number of ablations and associated ablated areas in the LA may impair LA contractility and, thus, increase the risk for SEC [17,18]. Several studies show that left atrial strain is a risk factor for both LA thrombi and SEC [19,20]. In addition, the LA-strain is associated with an increased risk of recurrence after ablation [21]. However, we could not confirm this correlation in the present study.
A new finding of the present study is that with the number of external cardioversions performed before ablation, the probability of SEC increases by almost a quarter (22.3%). It is also proven that the left atrial function correlates with the risk of recurrence of AF after external cardioversion [22]. For patients with multiple cardioversions (and other additional risk factors), this could mean that echocardiographic thrombus exclusion is recommended even if sufficient oral anticoagulation is consistently administered. The CHA2DS2-VASc score also represents a reliable risk stratification score, and a TEE for thrombus exclusion is recommended for values above 3 points (based on available data).
The available data were collected retrospectively and represent the typical patient population at the time of the investigation. Edoxaban was not yet registered on the European market at the time of data collection.
The incidence of SEC in patients undergoing TEE prior to AF ablation is significantly lower in patients treated with NOACs than those on VKA. Further studies are necessary to investigate the long-term effect of NOACs on stroke/TIA following AF ablation. In addition to the CHADS2 score parameter, LA diameter, persistent versus paroxysmal AF, and the number of previous cardioversions were identified as independent risk factors for SEC. The results support the justifiably increased use of NOACs as first-line oral anticoagulation in non-valvular AF.
None of the authors have conflicts of interest to declare.