Although rare, spontaneous esophageal perforation or "Boerhaave syndrome", carries a high case fatality rate. The 'classic' presentation of chest pain, vomiting and subcutaneous emphysema is actually uncommon clinically, with the literature reporting variable initial presentations. Early recognition is critical for intervention, given its high mortality. Here we review reported presentations of Boerhaave syndrome, and discuss a rare case of Boerhaave syndrome presenting with right sided tension pneumothorax at our tertiary care center. A 56-year-old male was discovered unresponsive and brought by family to the local community emergency department. He was in respiratory distress, hemodynamically unstable, and tension pneumothorax was diagnosed with Boerhaave syndrome. He was immediately transferred to our tertiary care center, and underwent thoracotomy and primary repair of esophageal rupture. He was discharged home in stable condition after a 1-month recovery in the intensive care unit and thoracic surgical ward.
Boerhaave syndrome is spontaneous transmural esophageal perforation resulting from sudden increase in esophageal pressure, combined with negative intrathoracic pressure. First described by Hermann Boerhaave in 1724, it is rare (3.1/1,000,000 cases per year) with high mortality up to 50% [1,2]. Delay in diagnosis and treatment is directly associated with poor outcome. The 'classic' presentation of Mackler's triad includes chest pain, subcutaneous emphysema and vomiting; but clinical presentation is often vague and nonspecific [1]. In a review by Brauer, et al. the most commonly reported symptoms were chest and abdominal pain in the context of retching, with associated clinical findings of dyspnea and septic shock [3]. However, review of the literature reveals several case reports of atypical and rare ways that Boerhaave syndrome can present, including: hoarseness of voice, back pain, hematemesis, and enterococcal bacterial pericardial effusion [4-7]. In addition, Malik, et al. describes a case of a patient presenting with chronic non-specific symptoms associated with an intrathoracic cyst secondary to Boerhaave syndrome [8]. Emergency physician awareness of the various ways that Boerhaave syndrome can present is key for early recognition. Currently available diagnostic imaging includes water-soluble upper Gastrointestinal (GI) contrast study and Computed Tomography (CT) with oral contrast. These unfortunately cannot be completed for some patients due to hemodynamic instability or decreased level of consciousness, leading to delayed diagnosis, morbidity and mortality. In this report, we present a patient who was discovered unresponsive in respiratory distress, with right tension pneumothorax. The tension pneumothorax was secondary to Boerhaave syndrome, repaired successfully with surgery, allowing return to daily activities on discharge.
A 56-year-old male with history of hepatitis C as well as alcohol and cocaine dependence was discovered unresponsive at home. He was brought to the local community hospital by a relative, and no further history was available at the time. In the emergency department he was found to be in respiratory distress with wheezing, and tachycardia up to 180 beats per minute. Chest X-ray revealed large right-sided hydro pneumothorax. Adenosine was given and he subsequently developed further hemodynamic instability with hypotension. He was diagnosed with tension pneumothorax, and a right thoracostomy tube was urgently placed with immediate return of 2 liters (L) of bilious content. He was intubated, resuscitated with intravenous fluids and broad-spectrum antibiotics, and transferred to a tertiary care center for definitive management (Figure 1).
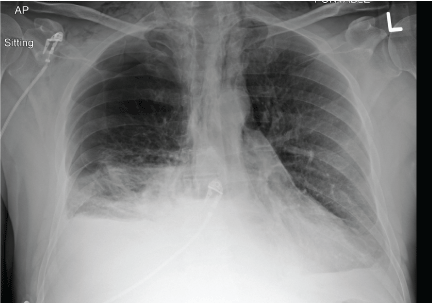 Figure 1: Chest x-ray on initial presentation at peripheral community hospital demonstrates large right hydropneumothorax with subcutaneous emphysema. View Figure 1
Figure 1: Chest x-ray on initial presentation at peripheral community hospital demonstrates large right hydropneumothorax with subcutaneous emphysema. View Figure 1
He arrived at our tertiary care center intubated on inotropic support. Bilious output was noted from the right-sided thoracostomy tube without air leak. Subcutaneous emphysema was present over the chest wall. The remainder of the physical examination was non-contributory.
Blood work revealed white blood cell count of 7.3 × 109/L (Ref. 4.0-11.0) and hemoglobin of 158 g/L (Ref. 135-170). He was acidotic with pH 7.19 (Ref. 7.34-7.44) on arterial blood gas. Lactate was 11.5 mmol/L (Ref. 0.5-2.2) and creatinine was 224 umol/L (Ref. 60-115).
It was not possible to perform a contrast swallow study or give oral contrast for CT scan since the patient was sedated and intubated. Contrast enhanced CT chest revealed right apical pneumothorax, moderate left complex pleural effusion, and thickened distal esophagus with extensive pneumomediastinum tracking to the lower esophageal region, concerning for Boerhaave syndrome. CT head and abdomen showed no acute abnormalities (Figure 2).
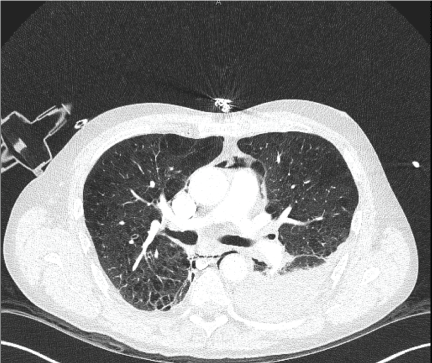 Figure 2: Contrast enhanced CT chest demonstrating left pleural effusion with lower peri-esophageal mediastinal fat stranding and pneumomediastinum suspicious for esophageal perforation due to Boerhaave syndrome. View Figure 2
Figure 2: Contrast enhanced CT chest demonstrating left pleural effusion with lower peri-esophageal mediastinal fat stranding and pneumomediastinum suspicious for esophageal perforation due to Boerhaave syndrome. View Figure 2
Ongoing resuscitation involved IV boluses of ringer's lactate, piperacillin-tazobactam, fluconazole and levophed. A left thoracostomy tube was placed. He was taken to the operating room immediately after CT scan, and underwent flexible bronchoscopy, to rule out tracheobronchial injury as well as esophagoscopy. This confirmed a long esophageal perforation with open communication to the right pleural space. A large air leak was noted from the right thoracostomy tube during insufflation via the esophagoscope. There was no evidence of other esophageal pathology such as: malignancy, stricture, ulceration or candida noted on endoscopy. He then underwent right posterolateral thoracotomy, revealing a large lateral perforation extending from 30-35 cm in the esophagus. Primary repair of the perforation was performed in two layers with intercostal muscle and pericardial fat patch. The patient was transferred to the Intensive Care Unit (ICU) post operatively.
Following his surgery, the patient's course in ICU was complicated by ongoing sepsis, Deep Vein Thrombosis (DVT), delirium and alcohol withdrawal seizure. He improved gradually, and was eventually transferred to the thoracic surgical ward after 27 days. Water-soluble contrast swallow study showed no leak, and he was started on an oral diet and gradually advanced. He was repatriated to his community hospital for further rehabilitation, and was ultimately discharged home uneventfully.
Our unusual case presentation of tension pneumothorax demonstrates that early recognition of Boerhaave syndrome is critical for survivorship. The classic Mackler triad of chest pain, subcutaneous emphysema and vomiting is present in less than 30% of patients [9]. Literature review reveals the presenting complaints are more often vague and non-specific, thus making diagnosis difficult for experienced emergency department physicians, risking delays in definitive life-saving treatment. There are also instances where the patient is unable to provide any history to help guide investigations, thus further delaying diagnosis. It is therefore important to maintain a high index of suspicion, and be aware of the atypical ways that Boerhaave syndrome can present.
While the initial approach to management of tension pneumothorax is constant, with rapid clinical recognition followed by emergent decompression, recognizing the potential for an atypical underlying cause is vital. Boerhaave syndrome has been recognized in the medical literature to present in unusual ways, however presentation with tension pneumothorax is exceedingly rare. There is only one previously reported case in North America, and 5 cases worldwide [10-14]. In contrast to our case, all of these cases reported history of preceding abdominal pain and vomiting, alerting the physicians to the possibility of esophageal rupture as the source of the pneumothorax. This again illustrates the importance of keeping Boerhaave syndrome in the differential diagnoses when approaching patients with tension pneumothorax, even in the absence of risk factors such as history of pain and vomiting, as shown by our case. Additionally in Boerhaave syndrome, the rupture typically occurs at the left posterolateral distal esophagus, thus causing left sided pleural effusion, however our patient further demonstrates the rare case of right-sided tension pneumothorax [15].
The preferred diagnostic test for esophageal perforation is water-soluble contrast imaging, with a dynamic swallow study or CT chest with oral contrast [16]. However, in unstable patients or in those who are unconscious, these may not be feasible. Complicating an already difficult clinical picture, the false negative rate of contrast swallow study for esophageal injury is reported at 10%-25%, though this may be improved with additional testing with insoluble contrast such as thin barium [16]. There are also reports of the use of presence of visible food particles from thorascopic drainage to diagnose Boerhaave syndrome [15,17]. Our case provides further support for careful clinical inspection of chest tube output to detect biliary or gastric contents as a fast and efficient way to clinically diagnose Boerhaave syndrome in patients without obvious external trauma, or in those for whom oral contrast enhanced imaging in not feasible.
Boerhaave syndrome can present with broad range of typical and atypical signs and symptoms, including respiratory distress and hemodynamic instability in an unconscious patient secondary to tension pneumothorax.
In a patient presenting with tension pneumothorax without the usual risk factors such as trauma or positive pressure ventilation, Boerhaave syndrome must be on the differential diagnosis.
In patients for who moral contrast studies are not feasible, bile or gastric content draining from the chest tube in the absence of trauma may be used to diagnose Boerhaave syndrome. Thus ensuring timely delivery of definitive care, allowing emergency physicians to maximize esophageal perforation survivorship.