Managing mechanical ventilation in patients with bronchopleural fistula (BPF) can be hazardous, especially in case of large air leaks and underlying parenchymal lung disease. The fistulous tract offers low airflow resistance, and air escapes through the BPF to the detriment of the tidal volume, jeopardizing the minute ventilation and oxygenation. We report the management of a 33-month-old girl in which neurally adjusted ventilatory assist (NAVA) avoided the use of extracorporeal membrane oxygenation (ECMO). NAVA is a respiratory support in which pressure assistance is provided in proportion to and synchronous with the electrical activity of the diaphragm. In this child, when increasing pressure settings to expand lungs and to offset hypoxemia, gas flow increases through a large BPF. When decreasing pressure settings, oxygenation was even worse. The use of NAVA helped break the deadlock. When switching to NAVA, gas leakage decreased and the air bubbling in the chest drainage system, that was previously continuous, persisted only at expiration. After four hours with NAVA, blood gas was normalized and FiO2 decreased to 30%. In a ventilated child with a large BPF and air leaks, NAVA can provide effective ventilation while reducing air leak.
Electrical activity of diaphragm, Interactive ventilatory support, Neurally adjusted ventilatory assist, Respiratory distress syndrome, Respiratory tract fistula
BPF: Bronchopleural fistula; ECMO: Extracorporeal membrane oxygenation; NAVA: Neurally adjusted ventilatory assist
Bronchopleural fistula (BPF) is a direct communication between the airway and the pleural space producing air leaks. BPF is a relatively rare but threatening complication of several pulmonary illnesses. The etiologies are varied: Postoperative complication for pulmonary resection, necrotic lung complicating infection, chemotherapy or radiotherapy for lung cancer, persistent spontaneous pneumothorax, and tuberculosis [1]. The air leaks induce incomplete expansion of the affected lung and derecruitment of the unaffected lung, compromise pulmonary gas exchange, and can lead to severe hypoxemia and respiratory acidosis. The supportive management of this potentially fatal disorder is challenging. Intercostal chest tubes are required to clear out the air accumulating in the pleural space and to drain coexisting pleural effusions. Spontaneous ventilation should be maintained whenever feasible [2]. When mechanical ventilation is required, the strategies for support rely on minimizing the air leak and maintaining acceptable gas exchange. The treatment includes keeping the airway pressure below the critical opening pressure of the fistula, optimizing pleural suction pressures and preventing further lung injury. High-frequency ventilation may be effective, especially in infants [3]. Extracorporeal membrane oxygenation (ECMO) is a rescue support for refractory hypoxemic respiratory failure, including in children with necrotizing pneumonia [4]. Nevertheless, the potential seriousness of complications associated with ECMO justifies finding other effective supports for children with refractory hypoxemia.
Neurally adjusted ventilatory assist (NAVA) ventilation provides pressure assistance in proportion to and synchronous with the electrical activity of the diaphragm [5]. NAVA was effective in a child with severe respiratory distress associated to pneumothorax and diffuse subcutaneous emphysema [6]. We present a case of the successful application of NAVA to a child with BPF and refractory hypoxemia despite conventional ventilation.
A 33-month-old girl (12 kg) with congenital cystic adenomatoid malformation was referred to our unit for refractory hypoxemia to consider rescue therapy. Although the pulmonary malformation was previously diagnosed with a large cyst in the right upper lobe, the child did not receive medical care earlier due to prolonged migration out of his native country. Surgery had been scheduled but the child developed a respiratory distress after spontaneous emptying of an abscess in a large bronchus. Once antibiotic therapy was initiated, a chest tube had been placed with evacuation of 500 ml of purulent liquid. Subsequently air was bubbling through a BPF into the chest drainage system. Respiratory management became more and more difficult due to a large air leakage.
On arrival, she was invasively ventilated with inspiratory pressure 30 cmH2O, expiratory pressure 8 cmH2O, respiratory rate 34/min, FiO2 84% with SpO2 94% (oxygenation saturation index [kem]: 13). Initial blood gas showed: pH 7.15, PvCO2 101 mmHg, PvO2 48 mmHg, lactate 0.8 mmol/L. Thoracic X-ray showed a voluminous aerial cavity filling almost the entire right hemithorax, with atelectasis of the right lower pulmonary lobe, and poor ventilation of the left lung (Figure 1A). A new 18-French-size tube was surgically placed allowing the mediastinum to regain median position with an improvement in left lung aeration (Figure 1B). Nevertheless, a large air leakage by the BPF remained with a continuous flow of air bubbling into the chest drainage system. Minute volume measured at expiration (VMe) was 1.5 L/min for a minute volume at inspiration (VMi) of 2.7 L/min (leakage VMi-VMe/VMi = 44%). Respiratory acidosis persisted with oxygen needs about 70%. When increasing pressure settings to expand lungs, gas flow increases through the fistula. Conversely, all attempts to decrease inspiratory pressures or chest drain depression altered oxygenation forthwith.
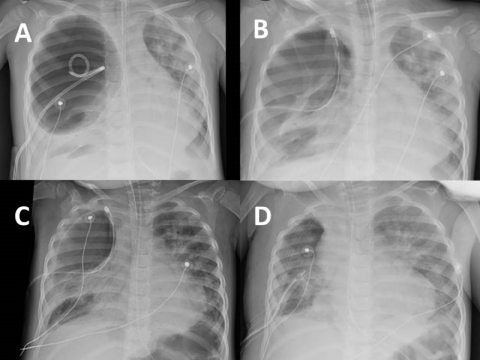 Figure 1: The patient's thoracic X-rays: A) At admission, voluminous aerial cavity filling almost the entire right hemithorax, ventilation mode: Pressure control; B) After new 18-French-size tube surgically placed, ventilation mode: Pressure control; C) Ventilation mode: NAVA; D) After surgery, ventilation mode: Pressure control. View Figure 1
Figure 1: The patient's thoracic X-rays: A) At admission, voluminous aerial cavity filling almost the entire right hemithorax, ventilation mode: Pressure control; B) After new 18-French-size tube surgically placed, ventilation mode: Pressure control; C) Ventilation mode: NAVA; D) After surgery, ventilation mode: Pressure control. View Figure 1
Therefore, we decided to use NAVA before resorting to extracorporeal membrane oxygenation (ECMO). Starting NAVA required the initial correct positioning of the "NAVA" gastric tube. This commercially available feeding tube equipped with sensors (Edi catheter, Maquet Critical Care, Solna, Sweden) permitted the recording of electrical activity of the diaphragm (Edi) via a Servo-U Ventilator (Maquet Critical Care, Solna, Sweden) using a standardized method [5]. Sedation was thus decreased with introduction of dexmedetomidine. In few hours, the child began to recover a spontaneous ventilation (Figure 2) and diaphragmatic electrical activity. When switching to NAVA ventilation, inspiratory pressures decreased dramatically from 31 to 18 cmH2O (Figure 2). In the same time, gas leakage decreased by 30%. The air bubbling in the chest drainage system, previously continuous, persisted only at expiration. After four hours with NAVA, blood gas showed: pH 7.41, PvCO2 71 mmHg, PvO2 34 mmHg, lactate 1.0 mmol/L. FiO2 was decreased to 30% resulting in SpO2 96%. In less than 24 hours, the thoracic X-ray showed an improvement of the aeration of the pulmonary parenchyma (Figure 1C).
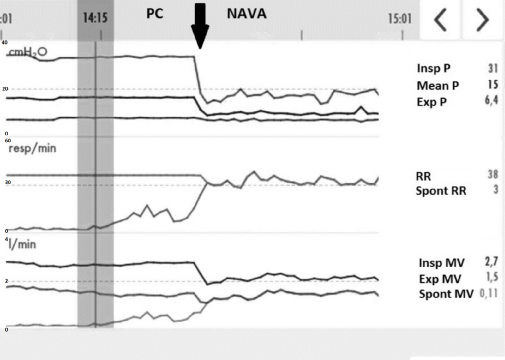 Figure 2: Screenshot with trends over 1 hour.
Figure 2: Screenshot with trends over 1 hour.
The downward vertical arrow indicates the switch from Pressure controlled mode (PC) to NAVA at 14:25. Last column on the right shows values collected next to the vertical bar (at 14:14) before the switch. Previously to NAVA, sedation was adjusted to permit some spontaneous ventilation (last bottom curve). The graph shows the decrease in peak inspiratory pressure and in inspiratory minute ventilation after the switch of ventilation.
Abbreviations: PC: Pressure controlled; NAVA: Neurally adjusted ventilatory assist; Insp P: Inspiratory pressure; Mean P: Mean airway pressure; Exp P: Positive end-expiratory pressure; Spont RR: Spontaneous respiratory rate; RR: Respiratory rate; Spont MV: Spontaneous expiratory minute ventilation; Insp MV: Inspiratory minute ventilation; Exp MV: Total expiratory minute ventilation.
View Figure 2
Thereafter right lobectomy could be achieved with the excision of a large cyst (Figure 1D). Three days later, she was successfully extubated and discharged from the intensive care unit. The parents gave us a written consent for publication.
BPF is a challenging complication where mechanical ventilation may be required but worsens the air leaks. The fistulous tract offers low airflow resistance, and air escapes through the BPF to the detriment of the tidal volume, jeopardizing the minute ventilation and oxygenation. In ventilated patients, using spontaneous respiration with low airway pressure could be an effective approach to reduce fistula flow and allow the leak to heal [2]. Nevertheless, spontaneous breathing under mechanical ventilation with pneumatic trigger is quite impossible in patients with a large BPF because of autotriggering [7]. The usefulness of a neural triggering was previously reported in a patient with serious air leakage, already stabilized by ECMO [7].
In ventilated children, NAVA often induced a significant decrease in inspiratory airway pressures [8,9]. Consistent with these data, NAVA was considered as ventilation mode to support a child with a large BPF and refractory hypoxemia despite conventional ventilation. Before switching to NAVA, sedation was alleviated with the use of dexmedetomidine allowing the child to recover spontaneous ventilation and a diaphragmatic electrical activity [10]. In a few minutes of NAVA ventilation, inspiratory pressures decreased dramatically with a reduction in leakage from 44% to 30%. In a few hours, oxygen requirements decreased from 84% to 30%, and blood pH was normalized.
Several factors could explain the beneficial effects observed with NAVA. First, NAVA provides assistance in synchronization, as well as in pressure assistance in proportion to the measured electrical activity of the diaphragm. Lower airway pressure reduces fistula flow. Secondly, it is likely that the excellent synchronization of mechanical ventilation with the spontaneous breathing leads to a better performance of ventilatory support, that is to say a better alveolar recruitment. Finally, with recovering spontaneous ventilation, air leaks occurred only at expiration. Although pleural or esophageal pressures were not recorded, it is likely that NAVA ventilation allowed a spontaneous pleural pressure generation ideally synchronized with the mechanical inspiratory pressures.
To our knowledge, it is the first case where NAVA was used as rescue ventilation in a child with life-threatening BPF. When switching to NAVA, inspiratory pressures and consequently the air leaks decreased. NAVA provided effective ventilation and succeeded to recover efficient pulmonary gas exchange. Further studies are required to determine the clinical impact of NAVA in patients with large air leaks.
No financial support was received for this work.
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Each author listed on the manuscript has seen and approved the submission of this version of the manuscript and takes full responsibility for the manuscript.