Cutaneous sarcoidosis is a common presentation for patients with sarcoidosis. Rarely, patients can present with psoriasiform lesions mimicking chronic plaque psoriasis. Here, we present a case of psoriasiform sarcoidosis in a pregnant patient. Pregnancy represents a unique challenge to systemic treatments if topical management fails. Tumor necrosis factor alpha inhibitors warrant special consideration during pregnancy.
Sarcoidosis, Psoriasis, Tumor necrosis factor-alpha, High-risk pregnancy
Sarcoidosis is a systemic inflammatory disease characterized by granuloma formation in multiple organs of the body, with a predilection for the lungs, skin, and eyes. Cutaneous sarcoidosis presents in myriad forms, appearing in about 25% of patients, while psoriasiform lesions are a rare manifestation in only about 0.9% [1]. We report the case of a 40-year-old woman at 10-weeks’ gestation with psoriasiform sarcoidosis.
A 40-year-old African-American woman at 10-weeks gestation with a history including active pulmonary sarcoidosis and diabetes was referred for several-year history of skin lesions by maternal-fetal medicine, which recently became scalier. Her medications included hydroxychloroquine 200 mg qday, prednisone 20 mg qday, and insulin. Physical examination revealed large, well-demarcated, hyperpigmented plaques with micaceous scale on her upper extremities, knee, neck, and back (Figure 1). There were also thin hyperpigmented plaques on her temple, nose, cheeks, and chin. A punch biopsy of the arm showed granulomatous inflammation consisting of histiocytes and lymphocytes in discrete aggregations and abutting the hyperkeratotic and psoriasiform epidermis (Figure 2). Clinical pathologic correlation was consistent with psoriasiform sarcoidosis.
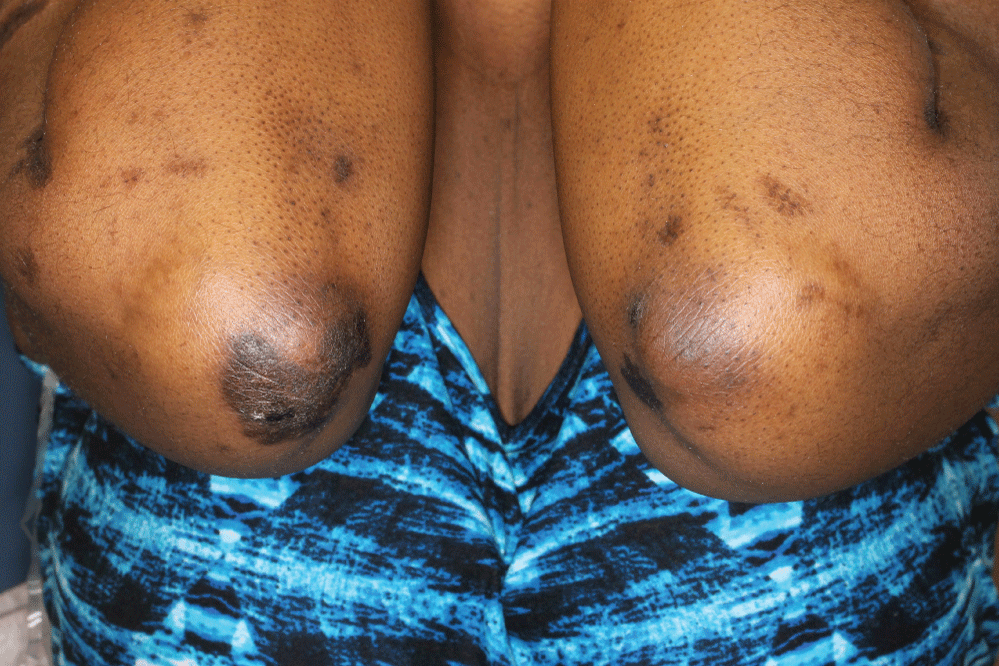 Figure 1: Well-demarcated, hyperpigmented plaques with micaceous scale on upper extremities. View Figure 1
Figure 1: Well-demarcated, hyperpigmented plaques with micaceous scale on upper extremities. View Figure 1
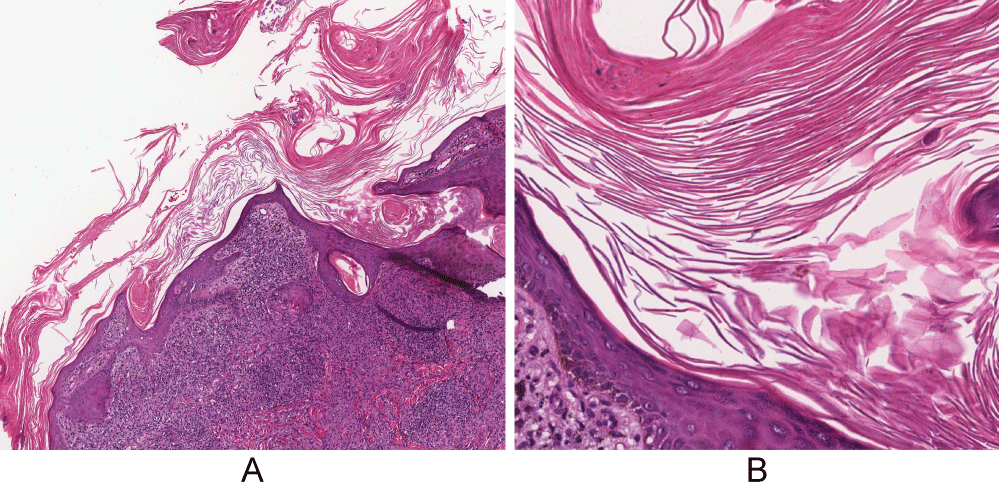 Figure 2: Granulomatous inflammation consisting of histiocytes and lymphocytes in discrete aggregations and abutting the hyperkeratotic and psoriasiform epidermis. (A) H&E, original magnification x4; (B) H&E, original magnification x20. View Figure 2
Figure 2: Granulomatous inflammation consisting of histiocytes and lymphocytes in discrete aggregations and abutting the hyperkeratotic and psoriasiform epidermis. (A) H&E, original magnification x4; (B) H&E, original magnification x20. View Figure 2
The patient was started on topical corticosteroids and continued on hydroxychloroquine. The prednisone was to be tapered. At follow-up, there was moderate improvement of skin lesions with near complete resolution of scaling, but still with significant classic sarcoidal appearance. Due to active pulmonary lesions, her treatment was to be discussed with pulmonology and maternal-fetal medicine regarding the decision to increase hydroxychloroquine or start a tumor necrosis factor alpha (TNF-α) inhibitor.
While the etiology of sarcoidosis has yet to be fully elucidated, environmental exposures and infectious agents have been suspected [2]. The presence of non-caseating granulomas is firmly established as the key pathologic finding. These characteristic sarcoidal granulomas consist predominantly of macrophages and epithelioid cells with peripherally surrounding lymphocytes. The pathological process begins when an unknown sarcoidal antigen is phagocytosed by antigen-presenting cells. This subsequently stimulates CD4+ T cells into secreting Th1-associated cytokines, such as IL-12, IL-15, IL-18, interferon gamma, and TNF-α, leading to the formation of the characteristic sarcoidal granulomas [3]. Many of these cytokines are also elevated in psoriasis, producing irregular hyperproliferation and differentiation of keratinocytes with histopathology demonstrating parakeratosis, intraepidermal neutrophilic infiltrations, and hypogranulosis [4].
Investigations into the overlap between sarcoidosis and psoriasis have focused heavily on the similar pathologic roles of Th1 cells. Both disorders have been associated with specific gene polymorphisms, such as IL23R, and psoriasis itself has been linked with 14 other autoimmune conditions, further strengthening the argument for shared pathways [5,6]. Paradoxically, sarcoidosis and psoriasis have both been reported after the initiation of TNF-α inhibitors, hinting again at homologous pathologic factors [7]. A previous report theorized that overexpression of TNF-α from both diseases may be responsible for the psoriasiform lesions arising in areas of cutaneous sarcoidosis [8].
For now, the goal of treating sarcoidosis focuses on suppressing symptoms, as the underlying cause remains elusive. First line therapy consists of topical and intralesional corticosteroids, whose benefits can often be seen in a matter of weeks [9]. While methotrexate and anti-malarials are also widely used, evidence for their efficacy is limited [10]. Recently, the spotlight has focused on TNF-α inhibitors, such as infliximab and adalimumab. The pivotal role of TNF-α in mediating the formation and maintenance of sarcoidal granulomas makes it an exciting therapeutic target [11]. Numerous studies have shown extremely promising benefits for patients recalcitrant to first and second line treatments [12-15].
Our patient presented at 10-weeks gestation with psoriasiform sarcoidosis, and she showed an incomplete response from topical steroids and hydroxychloroquine. The ensuing discussion with a multi-disciplinary team should revolve around increasing the dose of hydroxychloroquine versus considering a TNF-α inhibitor. However, pregnancy requires special consideration due to additional concerns regarding the risks to the fetus. Studies comparing TNF-α inhibitors to placebo in pregnant patients with irritable bowel disease and rheumatoid arthritis have not consistently shown strong associations with rates of preterm birth, spontaneous miscarriage, and congenital anomalies [16-19]. However, it has generally been recommended that TNF-α inhibitors be stopped in the second or third trimester when placental transfer is greatest. Certolizumab, a fragment of a monoclonal antibody specific to TNF-α, may not need to be stopped, since it does not as readily cross the placenta [19-21]. The decision to start TNF-α inhibitors in recalcitrant cases should be made on a case-by-case basis with multi-disciplinary input.
All authors have contributed equally to this work.
None.
None.