Melanoma is the most aggressive skin cancer and its incidence continues to increase worldwide. Molecular targeted kinase inhibitors have improved survival and tumor response for those patients with BRAF mutant disease.
We describe the case of a patient under treatment with BRAF and MEK inhibitors (BRAFi and MEKi) who presents cutaneous toxicity grade 3, as an adverse effect due to treatment.
A 51-years-old woman with no personal history to note was assessed at Dermatology department in September 2017 because of a bloody lesion on left foot for three months. No other symptoms.
Dermatologist described in physical exploration a lump of 2 × 2 cm on the left foot with drainage. No palpable adenopathies.
Biopsy was compatible with stage III melanoma lesion (pT4aL0V1R1 N0). BRAF study showed mutation for V600.
Study of extension with Positron Emission Tomography (PET-CT) showed a left inguinal lymphadenopathy suggestive of tumoral infiltration. Brain magnetic resonance without significant lesions.
Surgical removal was taken with large edges and inguinal lymphadenectomy with transposition of sartorius muscle and close with drainage. Afterwards, she was remitted to Oncology Department and we started nivolumab as adjuvant treatment (240 mg every two weeks).
After one cycle, transaminases increased upping up to five times normal value (hepatitis grade 3). Corticotherapy mg/kg/day was needed until toxicity resolution one month later.
PET-CT on the 13th of July described lymph node relapse inside of the left thigh and the left external iliac chain, thus, treatment schedule was changed to vemurafenib (iBRAF) 960 mg twice every day and cobimetinib (iMEK) 60 mg daily.
Three weeks later (first treatment cycle completed), she was admitted at the Emergency Room presenting erythematous lesions in lower limbs, fever and cough without expectoration.
Patient was valued by dermatologist, impressing of urticariforme exanthema in context of respiratory infection. Analytical parameters and chest radiography were normal. Biopsy of one of the lesions was performed and the patient started antihistamine, amoxicillin-clavulanic acid and was cited in medical oncology queries within 48 hours. After two days, the patient presented worsening of the skin lesions (maculopapular rash with desquamative lesions) and extension to the trunk and upper limbs (respects palms, plants and oral mucosa) (Figure 1a and Figure 1b).
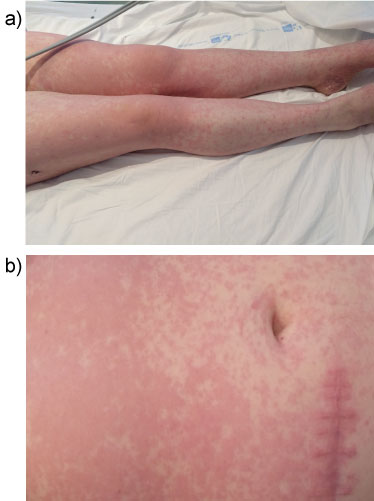 Figure 1: a,b) Maculopapular rash in limbs and abdomen. View Figure 1
Figure 1: a,b) Maculopapular rash in limbs and abdomen. View Figure 1
No conjunctivitis or rhinorrhea, however persistence of fever so she was admitted into the hospital and BRAFi and MEKi were suspended.
In the income analytics: ALT transaminase 113 UI/L (10-43), enzyme lactate dehydrogenase 825 UI/l (105-333), C-reactive protein 5.38 mg/L (0-4), leukocytes 3 × 103 µL, hemoglobin 15.3 g/dL and platelets 90 × 103 µL. With the previous findings, toxicodermia grade 3 or viral exanthema were suspected. Treatment with antibiotic and corticosteroids (prednisolone 0.5 mg/kg/day) was taken.
During admission exanthema diminished until its total resolution and standardization of analytical parameters. Eipstein Bar virus, Cytomegalovirus, parvovirus B19 and Mycoplasma virus serologies were negative. Pathology sample showed a slight infiltrated inflammatory full of lymphocytes perivascular surface. The observed histopathological findings were compatible with the clinical diagnosis of toxicodermia.
The patient remained asymptomatic, so she continued surveillance at home. After the toxicodermia presented by iBRAF and iMEK, it was decided to keep on this treatment, but reduce the dose (vemurafenib 480 mg twice every day and cobimetinib 40 mg daily). The patient presented excellent tolerance and without incidence of new reactions adverse up to three months.
Melanoma is one of the tumors whose incidence has increased most rapidly in recent decades. It represents 3% of all malignant tumors.
Despite the increase in its incidence, mortality has remained stable possibly due to a greater number of diagnoses in early stages.
Approximately 50% of advanced melanomas have BRAF V600 mutations that result in constitutive activation of the mitogen activated protein kinase (MAPK) pathway.
The development of targeted agents to block MAPK pathway activation, BRAFi and MEKi, has resulted in significant clinical benefit in patients with BRAF V600 - mutant melanomas (locally advanced and metastatic). These oral agents are associated with characteristic patterns of drug-related toxicities [1].
Characteristic of treatment with BRAFi is the emergence of drug resistance via activation of parallel signalling pathways. Inhibition of MEK, downstream of BRAF, has been tested as a strategy to bypass resistance.
The combination regimen using full doses of both agents appeared to generate fewer and skin adverse effects (AEs) compared with BRAFi monotherapy. BRAFi-induced hyperproliferative skin lesions occurred less frequently with combination vemurafenib plus cobimetinib compared with vemurafenib alone (hyperkeratosis, 10% vs. 28%; keratoacanthoma, 1% vs. 8%) [2]. AEs which occur more frequently with combination therapy are: Fever, chills, fatigue, diarrhea, hypertension and vomiting.
Skin toxicity is lower with dual therapy than with monotherapy. This is due to BRAFi because they do not initiate tumorigenesis but rather accelerate the progression of preexisting subclinical cancerous lesions, with paradoxical MAPK pathway activation, which is inhibited by treatment with a MEKi [3].
Up to 60% patients with BRAFi present skin toxicities (rashes and other skin irritations, acneiform dermatitis, hyperproliferative skin disorders and photosensitivity). Monitoring of cutaneous eruptions must be undertaken by the prescribing team. In our case, skins lesions (rash without photosensitivity) appeared before the first month of treatment. Other likely etiologies (viral exanthem) have been wiped out by serology. The result of the skin biopsy was compatible with toxicodermia.
Treatment is aimed mostly at alleviating the symptoms, including the use of emollients, antihistamines, and analgesics; a short course of steroids may also be appropriate. For intolerable grade 2 or grade 3-4 cutaneous AEs, dabrafenib and trametinib alone or in combination should be withheld for 3 weeks. If the AE improves, the drug(s) can be resumed at a lower dose [4].
Proper proactive management of these cutaneous AEs is critical to avoid drug delays, interruptions, or discontinuations. In our case, skin toxicity was grade 3 (affecting more than 50% of skin surface) so dermatological toxicity management was: Stopping kinase inhibitor until complete resolution, antihistamines and oral steroids for 10 days.
Patient was able to continue with vemurafenib and cobimetinib at a lower dose without presenting new skin toxicities.
Molecular targeted kinase inhibitors have been developed for the treatment of metastatic and locally advanced melanoma and have dramatically improved outcomes for those patients with BRAF mutant disease.
Skin toxicities are the most common AEs associated with BRAFi.
Proactive toxicity management is important to ensure maximum treatment benefit and avoid unnecessary treatment discontinuation.
Assessment by a trained dermatologist is recommended for toxicities grade 3 and 4.
Skin biopsy is not mandatory but may be a support in the clinical assessment.