The present study aims to design and evaluate performance of chitosan based bio-active containing acrylic materials and investigations towards applicationfor the development of novel bio-active materials with build in capabilities for treatment and prevention of denture stomatitis and associated conditions in denture wearers in vitro.
The bio-active modified polymethyl methacrylate denture resins (PMMA) were prepared by dispersion of the corresponding component in glycerol and acetic acid with the addition of chitosan gelling agent. The release behaviors at physiological pH and also under acidic conditions and stability of the antioxidant-chitosan were also evaluated. Mechanical performance such as tensile strength and compressive strength were measured as well bio-adhesive studies were investigated in order to assess the suitability of these designer materials.
The bioactive modified PMMA hydrogels showed a high adhesive force and were only slightly swelled in the aqueous medium. Bioactive release suggested prolonged release of the therapeutic agent from the hydrogels. The hydrogels also had significant free radical defense capability.
In this study we demonstrated that the newly preparedbio-active modified PMMA resins are suitable novel bio-active materials capable of comparable performance with the conventional PMMA materials with additional benefit of therapeutic bioactive release as well as potential antimicrobial properties to be demonstrated in vitro.
Chitosan, Bio-active, PMMA, Drug delivery systems, Free radicals
Since first polymerized by Walter Bauer in 1936, acrylic resin denture base gradually took the place of traditional metal base and became most commonly used denture base material in clinical fabrication [1,2]. It is a combination of advantages, such as of its excellent esthetics, ease of processing and repair and being economical, rather than one excellent aspect that accounts for its wide usage [3], including its popularity in satisfying aestheticdemands and clearly defined processing method in dentistry application. However, this material is not ideal in every respect [4], especially when meeting with mechanical requirements of prosthesis. Fracture of acrylic resin denture base happens frequently because of the fatigue and chemical degradation of base material [3], which is reflected by a large number of denture repairs annually [5].
The surface of denture base acrylic resin is porous, and denture plague easily adheres to the surface of dentures [6]. The colonization of microorganisms on the denture base acrylic resin occurs rapidly and candida species adheres strongly to denture base materials.
Oral candidosis in the form of candida-associated denture stomatitis is a common disease in some 65 per cent of denture wearers [7]. Candida albicans and related species are believed to play a major role in initiating, maintaining and aggravating the disease. This is the further evidence by the fact that the denture stomatitis is often cured by antimycotic treatment [8].
Chitosan, which is a biologically safe biopolymer as well as an antioxidant, has been proposed as a bio-adhesive polymer and is of continuous interest to us due to its unique properties and flexibility in a broad range of oral applications reported by others and ourselves recently [9-11].
Lentinusedodes, known as shiitake mushroom, has received great attention due to positive health effects, including antitumour and hypocholesterolemic activity [12], related to the presence of β-glucans [13].
Propolis is a resinous substance produced by bees with antibacterial, antifungal, antiviral, and anti-inflammatory activities [14,15]. Propolis antibacterial activity is bacteriostatic and, in high concentration, bactericidal [16]. Propolis has antimicrobial activity against gram-positive bacteria, egS. aureus, but limited action against gram-negative bacteriaand also against some fungi, e.g., C. albicans [17,18].
The copaiba tree is native to Latin America and Occidental Africa [14]. There are more than 20 species of copaiba in Brazil, and the most commonly described effects are anti-inflammatory, analgesic, antibacterial and antitumoral activities [15-17].
The present study aims to design and evaluate performance of chitosan based bio-active containing acrylic materials and investigate applicationsfor the development of novel bio-active materialswith build in capabilities for treatment and prevention of denture stomatitis and associated conditions in denture wearers, while the performance of the material is not compromised in vitro.
Propolis Brazilian (Red, Natura Nectar), Copaiba Oil (Laboratorio Sao Lucas, Brazil) and Shiitake powder (Border herbal health, Australia) were purchased from a commercial supplier (Wholesale Chemist, QLD, Australia) and used without further purification. (-)-Epigallocatechin gallate (EGCG) from green tea, 95%, (-)-epicatechin gallate (ECG) from green tea, 98%, (-)-epigallocatechin (EGC) from green tea, 95%, (-)-epicatechin (EC), 90%, gallic acid (GA) purity not specified, 6-hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid (trolox), 95%, Folin-Ciocalteu reagent, ferric chloride, Tween-20 and 2,20-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS)were purchased from Sigma-Aldrich Company Ltd. (Australia).
Chitosan (Aldrich, Australia), glycerol (Sigma, USA), glacial acetic acid (E. Merck, Germany) were used as received. The degree of deacetylation of typical commercial chitosan used in this study is 87%. Chitosan with molecular weight 2.5 × 103 KD was used in the study. Gelatin in powder form was purchased from Shanghai Chemical Reagent Co., (Shanghai, China) with the number-average molecular weight (Mn) of about 8.7 × 104. The isoelectric point is 4.0-5.0.
The bio-active containing gel was prepared by dispersion of 0.2 grams of commercially available bio-actives (Propolis (Red Brazilian), Copaiba oil or Shiitake powder) in glycerol (5% w/w) (1 ml) using a mortar and a pestle following the earlier reported generic protocol [16]. Ten milliliters of glacial acetic acid (2% w/w) was then added with continuous mixing to the mixture of 0.2 grams of commercially available bio-actives (Propolis (Red Brazilian), Copaiba oil or Shiitake powder) in glycerol (5% w/w) (1 ml) and finally chitosan (10% chitosan w/w) polymer was added and mixed well to form there quire gel and then mixed into a PMMA resin prior to setting. The amount of bioactive component such as propolis, copaiba oil or shiitake mushroom extract respectively was determined to be 0.2 grams of the prepared material (10 grams). The total phenolic concentration was quantified using Rocha et al [19] and described by Waterman & Mole [20] with some modifications.
Total phenols content was estimated by a colorimetric assay based on the same procedure realized by Rocha et al. [19] and described by Waterman & Mole [20] with some modifications. The samples were diluted in distilled water to obtain a concentration of 5 μg/mL of total phenols. The concentration of 5% v/v of Folin-Ciocalteu reagent and 10% v/v sodium carbonate (35% w/v) reagents were added to the samples. After the addition of the reagents, the solutions were kept in the dark at room temperature for 30 min and the absorbance was read at the wavelength of 760 nm in a mini 1240 UV - Vis spectrophotometer (Shimadzu Co., Kyoto, Japan). Gallic acid (Aldrich, Australia) was used as a standard. The analyses were performed in triplicate. The total flavonoids, total phenolics contents as well as the antioxidant activity results were statistically analyzed by the variance analysis (ANOVA) by the Prism 6 software. In all analyses, a 5% significance level was considered.
The swelling/weight loss tests were performed when triplicates of each samples composition (approximately 2 cm2, weight normalized) were immersed in 2 mL of different fluids at 37°C for each time interval studied (1, 2, 4, 24, and 96 h). Two different media were used in accordance with the ISO 10993 - 9 standard. The first medium was Phosphate Buffered Saline (PBS, Sigma Aldrich), intended to mimic the inorganic phase of human plasma [17]. The other media was PBS with a reduced pH which was intended to simulate the local inflammatory environment of the wounds [15,18]. This is termed Solution pH 4.0. The pH was lowered using Lactic Acid (Sigma Aldrich). The fluid absorption of each sample was calculated according to equation (1) to obtain their swelling degree (SD). WS is the weight of the sample at each time interval (swollen weight) and WD is the dry weight before swelling [21]. After 4 days of immersion, the samples were dried and weighed in order to calculate their weight loss (WL) [equation (2)], where WD and WDS are the weight of the dried samples before and after swelling tests, respectively
Equation 1: SD = 100 × (WS-WD)/WD (%)
Equation 2: WL = 100 × (WS-WD)/WD (%)
To analyze the bio-active release (propolis (Brazilian), Copaiba oil and Shiitake mushrooms) based on the total phenolic concentration; the swelling media was analyzed after 1, 2, 24, and 96 h of immersion via UV - Vis spectrometer, from 300 to 800 nm, using polystyrene cuvettes [22]. For quantification of the amount of propolis released, a standard curve was created by diluting the original propolis in isopropanol resulting in several aliquots of known concentration, which were then analyzed in the same wavelength range. The area of the peak of these aliquots (of known concentration of bio-additive) was calculated and used to compare with those of the bio-additive released by the samples.
Tensile testing was conducted using Instron 5565. Following American Standardized Testing Materials Standard D3039, rectangular samples were approximately 6 - 8 mm in length, 1mm in width an 1 mm in thickness, and tested with a gauge length of 3.5 ± 0.4 mm [23]. Samples were elongated at a rate of 1% of gauge length per second. The cross-sectional area of samples was evaluated using Image J image analysis software [24].
Compressive strength test samples were placed in the measuring apparatus in an appropriate manner and cross-sectional area of each sample (mm2) was determined. A compressive load (N) was applied at a crosshead speed of 1.3 mm/min [25]. The compressive strength (MPa) was measured at the sample fracture point.Mean, average, and mode in each group were calculated and normal distribution curve was evaluated. One-way ANOVA, followed by multiple comparison test (Scheffe's test), was used for statistical analysis. Statistical significance was set at P < 0.05.
Bio-adhesion studies were done using a Chatillon apparatus for force measurement [26]. This method determines the maximum force and work needed to separate two surfaces in intimate contact [26]. The hydrogels (0.1 g) were homogeneously spread on a 1 cm2 disk and then the disks were fixed to the support of the tensile strength tester using double sided adhesive. The bio active modified PMMA material was brought into contact with a slice of pig ear skin was established in order to imitate adhesion of the gel to the "oral mucosa prototype system" structure. After a preset contact time of 1 min under contact strength of 0.5 N, the 2 surfaces were separated at a constant rate of displacement of 1 mm/s. The strength was recorded as a function of the displacement, which allowed to determine the maximal detachment force, Fmax, and the work of adhesion, W, which was calculated from the area under the strength-displacement curve [26].
The single electron microscopy (SEM) images were obtained for selective bio-active modified PMMA resins to characterize the microstructure of the freeze-dried samples and are presented in figure 1 SEM observations of PMMA-based samples revealed a smooth surface with the formation of the valleys and crests, which could be attributed to the presence of some aqueous medium in the preparation of modified materials which is consistent with the SEM images previously reported for similar PMMA based materials.
The poly methyl methacrylate has adequate tensile and compressive strength for complete and partial dentures [27]. The compression behavior for composite prosthetic dentures represents the important mechanical properties specialization when using the polymer matrix materials. The compression strength values results obtained from compression tests are carried out for all bioactive prepared materials and results are summarized in the figure 2. The addition of the bioactive compounds such as Shiitake extract, Copaiba oil or Brazilian propolis had not influenced significantly the compression strength of the bioactive PMMA material. However upon incorporation of chitosan: bioactive combination (10% w/w) into the PMMA material the compressive strength of the new bioactive material was significantly lowered in comparison to the standard PMMA material. (Where Acr = PMMA resin materials)
 Figure 1: SEM photographs of interior morphology of the selected gels under investigation for (a) Shiit/PMMA/Chi, (b) Cop/PMMA/Ch, (c) Pr/PMMA/Ch. View Figure 1
Figure 1: SEM photographs of interior morphology of the selected gels under investigation for (a) Shiit/PMMA/Chi, (b) Cop/PMMA/Ch, (c) Pr/PMMA/Ch. View Figure 1
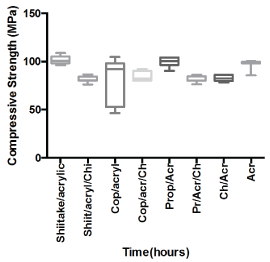 Figure 2: Comparison of compressive strength of bio-active denture materials. Where Acr = PMMA resin. View Figure 2
Figure 2: Comparison of compressive strength of bio-active denture materials. Where Acr = PMMA resin. View Figure 2
The tensile behavior for composite prosthetic dentures represents the important mechanical properties specialization when using the polymer matrix materials and is summarized in figure 3. The addition of the bioactive compounds such as Shiitake extract, Copaiba oil or Brazilian propolis had not influenced significantly the tensile strength of the bioactive PMMA material. However upon incorporation of chitosan-bioactive combination (10% w/w) into the PMMA material the tensile strength of the new bioactive material was significantly lowered in comparison to the standard PMMA material. The slight increase of tensile strength is probably due to the potential action interference of the bio-actives and their antioxidant capacity with the polymerization rates of the PMMA presence of chitosan as well as phenolic compounds which are capable to acts as the protective host of the excess of free radical formation and therefor the slight decrease in the tensile as well as compressive strength is observed. The results are basically consistent with the literature [28,29] and more detailed investigations into mechanistic interaction of chitosan/bioactive/PMMA resin are currently on the way in our laboratory.
 Figure 3: Comparison of tensile strength of bio-active denture materials.
Figure 3: Comparison of tensile strength of bio-active denture materials.
Where Acr = PMMA resin. View Figure 3
The swelling characteristics of the bio-active modified materials at pH 7.0 and pH 4 are shown in figure 4a and figure 4b respectively. Swelling has not been affected significantly in case of either incorporation of bioactive or bioactive/chitosan did not increased in the case at either pH. No definite trend in swelling withcomposition was observed. Though PMMA is a hydrophobicpolymer, the acidic environment may hydrolyse the methacrylate group to some extent conferring hydrophilic nature to the copolymer.
 Figure 4A: Swelling at pH = 4. View Figure 4A
Figure 4A: Swelling at pH = 4. View Figure 4A
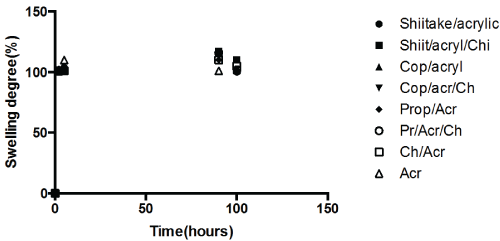 Figure 4B: Swelling at pH = 7. View Figure 4B
Figure 4B: Swelling at pH = 7. View Figure 4B
On the basis of calibration curve for standard total phenolic concentration, the concentration of these bioactive compounds in the propolis (Brazilian, Red), Copaiba Oil and Shiitake mushroom powder sample used in the study was assessed and is summarized in figure 5.The results are consistent with the previously reported values for a hydroalcoholic extracts of the propolis, copaiba oil and shiitake mushroom powder in the literature [30-33]. It is well accepted in the literature that the biological activities for the bio-actives such as Propolis, Copaiba oil and Shiitake extracts for extracts for example are mostly due to the high levels of phenolic acids [30-36].
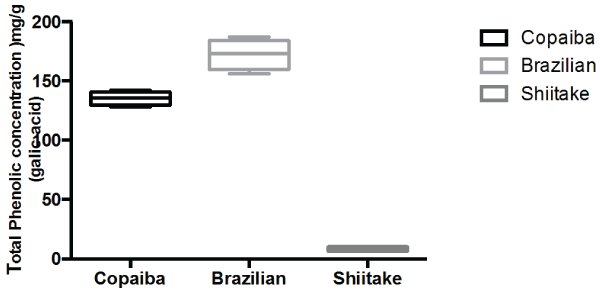 Figure 5: Total phenolic concentration of the bioactive extracts used in the investigation. View Figure 5
Figure 5: Total phenolic concentration of the bioactive extracts used in the investigation. View Figure 5
The amount of bio-actives (such as Brazilian propolis, Shiitake mushroom extract and Copaiba oil) release in swelling media was analyzed after 1, 2, 24, and 96 h of immersion (Figure 6). The propolis release by polymeric systems usually occurs in two steps: the release of certain amounts of propolis in the first day of swelling as well as a prolonged release in some cases [37,38]. A trend could be observed in all curves after 4 days of immersion: there was a high bioactive release in the initial hours and the cumulative release reached constant values up to 1 day of immersion. No prolonged release was observed.
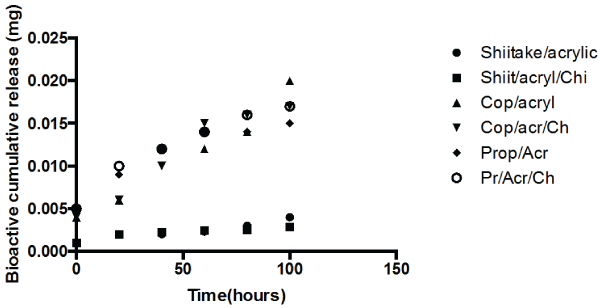 Figure 6: Bioactive cumulative release profile of Ch/PMMA-bioactive samples. The Ch/PMMA-bioactive samples were immersed in (a) PBS and (b) Solution pH 4.0 and the bioactive (such as copaiba oil, propolis and shiitake extract delivered was quantified after regular intervals of time for 4 days.
Figure 6: Bioactive cumulative release profile of Ch/PMMA-bioactive samples. The Ch/PMMA-bioactive samples were immersed in (a) PBS and (b) Solution pH 4.0 and the bioactive (such as copaiba oil, propolis and shiitake extract delivered was quantified after regular intervals of time for 4 days.
Where Acr = PMMA resin. View Figure 6
Higher adhesiveness of the modified PMMA resins is desired to maintain an intimate contact oral mucosa and the prosthetic device such as full or partial denture, therefor bio-adhesion between the newly prepared modified bio-active containing PMMA was tested against pig ear skin structure and results are summarized in table 1 Chitosan hydrogels showed the highest adhesive force and the work of adhesion. This can be expected because of the well-known intrinsic bio adhesive properties of chitosan [39]. The adequate water absorption capacity together with the cationic nature, which promotes binding to the negative surface of skin structure, can also interpret this results.
Table 1: Bio-adhesion table. View Table 1
The first step for successful colonisation of mucosal surfaces or any other surface by C. albicans is adhesion [40]. Although among the functions of Propolis, the fungicidal property has already been shown, in this study, we aimed to verify its effectiveness in inhibiting the adhesion of C. albicans biofilm on a denture surface (Figure 7).
 Figure 7: Surfaces of the materials after exposure to artificial saliva and oxygen as an in vitro model for the biofilm formation after 3 weeks of storage. a. Shiit/PMMA/Chi, b. Cop/PMMA/Ch, c. Pr/PMMA/Ch. View Figure 7
Figure 7: Surfaces of the materials after exposure to artificial saliva and oxygen as an in vitro model for the biofilm formation after 3 weeks of storage. a. Shiit/PMMA/Chi, b. Cop/PMMA/Ch, c. Pr/PMMA/Ch. View Figure 7
The reinforcement and bio-active addition of the copaiba oil, propolis (Brazilian) or shiitake mushroom extract of the tested PMMA dental resin resulted in no statistically significant increase of its compressive of tensile strength. The additional benefits of the functionalized bio-material as preventative measure for the biofilm formation as well as build in bio-active free radical defense capability of the materials make them ideal candidates for further development and application in the materials for prosthetic devices.