Diabetic foot ulcers are one of the major complications of patients with diabetes mellitus. And due to their high susceptibility to microbial infections, are the leading cause of hospitalization and amputation of lower limbs. It has been well studied the antimicrobial properties of silver nanoparticles (AgNPs), therefore their use in biomedicine is a trend. Herein we present for the first time the use of AgNPs for the treatment of diabetic foot ulcers of grade II and III of Wagner classification. Ulcers were treated by topical administration of AgNPs (at 1.8 mg/mL of metallic silver) in addition to conventional antibiotics. In all the cases presented in this study, a significant improvement in the evolution of ulcers was observed upon AgNPs administration. The edges of the lesion reached the point of closure. These results constituted the basis for further studies on the use of AgNPs for the treatment of chronic ulcers from different origins.
Diabetic foot ulcers, Diabetes mellitus, Silver nanoparticles, Nanomedicine, Chronic ulcers
AgNPs: Silver nanoparticles; DFU: Diabetic Foot Ulcers; PVP: Polyvinylpyrrolidone; Ag-SD: Silver Sulfadiazine; DM: Diabetes Mellitus
Diabetes mellitus (DM) is one of the most common chronic and metabolic diseases. According with the World Health Organization (WHO) in 2014 there were 422 million of diabetic people and 1.5 million of people have died due to this illness in 2012 [1,2]. In Mexico DM is the second major cause of death [3]. A frequent ailment among DM patients is the complications of lower limbs also known as diabetic foot or Diabetic Foot Ulcers (DFU) [4]. DFU are characterized by infection, ulceration and destruction of the deepest tissues of lower limbs, associated with neurologic alterations and diverse degrees of peripheral vascular disease [5]. Between 12 and 25% of patients with DM are affected with DFU, being the main cause of hospitalization and lower limbs amputation [6-8].
Infection of DFU with Multi-Drug Resistance (MDR) microorganisms may increase the time of wound healing, hospitalization, treatment costs and patient mortality [9]. To overcome this, silver has been extensively used to treat bacterial infection and to prevent wound sepsis due to its well-known antimicrobial properties.
Silver has been incorporated as topical antibacterial agent in a wide variety of products on the market (i.e. Silver Sulfadiazine (Ag-SD), Flammazine™, silver sulfadiazine/chlorhexidine (Silverex®), Ag-SD with cerium nitrate (Flammacerium®) and Ag-SD-impregnated lipid colloid wound dressing, Urgotul SSD®) [10,11]. Moreover, topical silver antimicrobials and dressings are facing difficulties that involve their low silver release rate, the lack of tissue penetration, and the rapid consumption of silver ions and pro-inflammatory side effects of silver nitrate in cream-based products. It has been reported that Ag-SD delays the wound-healing process and has cytotoxic effects on various host cells impairing the re-epithelialization process [10,12]. To overcome this, various types of silver dressings have incorporated silver within it, instead of being applied as a separate component. For example, Acticoat™ and Silverlon® have a prolonged release of silver nanocrystals (with a size < 20 nm) to the wound area. This application requires a less frequent dressing change. With this it is possible to reduce the risk of nosocomial infection, care cost and patient discomfort [10,13].
However, silver dressings are currently used before the wound exhibits a bacterial infection, but in the case of DFU, wounds are already infected with several strains of microorganisms including MDR. Therefore, it is necessary to develop efficient antibacterial therapies against MDR microorganisms that allow the rapid healing of DFU, without the cytotoxic effects of silver ions while effectively eliminating microbial infections.
Nanotechnology is generating new applications for biomedicine and biotechnology through the synthesis of nanomaterials. Their nanometric scale (between 1 to 100 nm) confers them their unique physicochemical properties [14]. Thus, nanotechnology offers an alternative to overcome infectious diseases through the use of antimicrobial nanomaterials [15]. Silver nanoparticles (AgNPs) resulted to be one of the most used nanomaterials in medical products such as bandages, wound dressings, catheters and textiles due to their excellent microbicidal activity against wild and nosocomial strains of MDR microorganisms [16-20]. The infections presented in DFU are mostly polymicrobial and in many cases the presence of MDR microorganisms increases the risk for a chronic infection, limb amputation and morbidity [5].
Taking in consideration the antimicrobial properties of AgNPs, the aim of this study is to investigate the potential use of AgNPs for the treatment of DFU in patients. In this work, we presented tow cases of patients with DFU classified as Wagner ulcers II and III, who were treated with AgNPs solution at 1.8 mg/ml of metallic silver topically administered. The evolution of the wound healing during treatment was documented by photography, which clearly shows that the DFU disappeared or significantly reduce its area without developing infection after treatment with AgNPs.
After comparison of different AgNPs commercially available, we concluded that only Argovit preparation resulted to have multiple certificates for their usage in veterinary and human applications [21]. Argovit (Scientific and Production Center Vector-Vita, Russia) is a preparation of highly dispersed silver nanoparticles with an overall concentration of 200 mg/mL (20%) of PVP-coated AgNPs in water. The content of metallic silver in Argovit preparation is 12 mg/mL, stabilized with 188 mg/mL of Polyvinylpyrrolidone (PVP). AgNPs dilutions were calculated according to metallic silver content in Argovit preparation. Solutions of AgNPs were prepared with distilled and sterile water and were kept at 4 ℃ in darkness.
Size distribution and morphology of AgNPs were determined on the basis of the results obtained by High-Resolution Transmission Electron Microscopy (HRTEM) using a JEOL-JEM-2010 microscope. Hydrodynamic radius and Zeta potential were measured by using Dynamic Light Scattering (DLS) (Malvern Instruments Zetasizer Nano NS model DTS 1060, UK) equipped with a green laser operating at λ = 532 nm at 25 ℃. AgNPs were characterized by UV-Vis spectroscopy in the range of 200 to 900 nm using a Cary 60 UV-Vis spectrophotometer (Agilent Technologies, Santa Clara, CA, USA). Further characterization of lyophilized Argovit was performed with Fourier Transform Infrared Spectroscopy-Attenuated Total Reflectance (FTIR-ATR) analysis in the range of 400 to 4000 cm-1 with a resolution of 2 cm-1 on a universal diamond ATR top plate accessory (Perkin Elmer, USA); the sample spectrum was compared with that of standard solid PVP (Mw 100 kD).
Topical administration of AgNPs for DFU treatment was developed by Med. Cesar Alejandro Almonaci Hernández.
The research protocol for this study was approved by the Bioethic Committee of the Instituto de Servicios de Salud Publica del Estado de Baja California, Seccion Hospital General de Tijuana (No.000056, Septiembre de 2011). All the patients reported in this study signed an informed consent letter; in this case they were included in the study. Here we presented two clinical cases of diabetic patients with different grades of DFU according with Wagner classification.
The first patient is a 48-years-old male with type 2 diabetes controlled with insulin and oral hypoglycemic agents. He presented a third grade Wagner classification ulcer of 1.5 cm at the internal face of the first toe in the right foot. Edges are adjoining with four single stitches. A perilesional edema is presented with purulent and fetid material drainage after digital pressure. Pedium and tibial pulse are present, also sensitivity to monofilament 8/10, vibratory sensitivity was not evaluated and non-pathological Achilles reflex was observed.
Second patient is a 54-years-old male with type 2 diabetes controlled with oral hypoglycemic agents. With systemic arterial hypertension in treatment with acetylcholinesterase inhibitors and peripheral vascular insufficiency treated with pentoxifylline. Patient presents dermatitis of 6 weeks of evolution at the plant of left foot, treated with miconazole without any improvement. There is a loss of hair on the dorsum of the left foot. Also, loss of subcutaneous tissue and hyperkeratosis are observed in pressure areas. It is evident bony prominence at the outer edge of the plantar arch. Patient has two plantar ulcers, the first one of approximately 2 cm of diameter at the level of first metatarsal, classified as second-grade Wagner. The second ulcer of circa 0.5 cm of diameter is presented at the bony prominence. Both ulcers exhibited necrotic edges and hyperkeratosis, with a brown-red colored centers and blue-violet areas, with no apparent purulent exudate.
The treatment of diabetic foot ulcers with AgNPs, started after all patients signed an informed consent letter given their approval to be included in the protocol.
The treatment consisted of a complete exploration of the wound area, debridement when necessary and cleaning with surgical soap solution. After this, a topical administration of AgNPs solution (with a metallic silver concentration of 1.8 mg/ml) was done first, and this treatment was repeated every 24 h for minimal 7 days. The amount of metallic silver to be applied in the injury was calculated on the basis of the content of metallic silver present in the AgNPs solution Argovit, as mentioned in methodology section. To record the evolution of the wound healing process, photographs were taken before and after the treatment with AgNPs.
Physicochemical characteristics of AgNPs have been recently reported by our group [22] and are summarized in Table 1. Metallic silver nanoparticles are spheroidal in morphology, the distribution of size goes from 1 to 90 nm with an average size of 35 nm, and their hydrodynamic diameter considering both the PVP covering and the metallic silver nanoparticle is 70 nm. Zeta potential of AgNPs is -15 mV and their surface plasmon resonance absorbance maximum is at 420 nm wavelength. A TEM micrograph of the AgNPs used in this study is shown in Figure 1.
 Figure 1: Spheroidal morphology of AgNPs showed by a TEM image.
View Figure 1
Figure 1: Spheroidal morphology of AgNPs showed by a TEM image.
View Figure 1
Table 1: Physicochemical characteristics of Argovit AgNPs. View Table 1
The evolution of the wound healing process of DFU treated with AgNPs was documented by photography. In Figure 2 it is observed the chronological evolution of DFU from the first patient. In Figure 2A it is shown the initial appearance of a third-grade Wagner ulcer present at the internal face of the first toe of the right foot. After wound cleaning (Figure 2B) the stitches were removed and discharge of fetid and purulent material was observed. Then wound rinse with physiological solution was achieved. Afterwards a brown-red colored wound is observed. Necrotic areas and friable tissue were removed. An open wound of approximately 2 cm length and 1 cm depth was exposed for a second intention closure. Then, daily topical administration of AgNPs at 1.8 mg/ml of metallic silver started. After 8 days of treatment with AgNPs solution (Figure 2C), an open wound is observed with a decrease in diameter and depth, and with trends to closure. Ulcer exhibited a bright-red color, with abundant presence of granular tissue. Active edges without data of infection or necrosis are observed at the borders of the ulcer. Moreover, patient referred pain diminishment. After this, topical administration of AgNPs twice a week is indicated. Figure 2D depicts the evolution of the healing process after 21 days of topical administration of AgNPs solution. A progressive diminishment of the lesion extent is noticeable. The zone of the ulcer shows a diminishment of its diameter, the coping is observed along the edges culminating in the closure of the injury where no necrosis or inflammation was observed. Indeed, the pigmentation of the tissue confirms the imminent reepithelialization process with active edges that favoring closure of the injury.
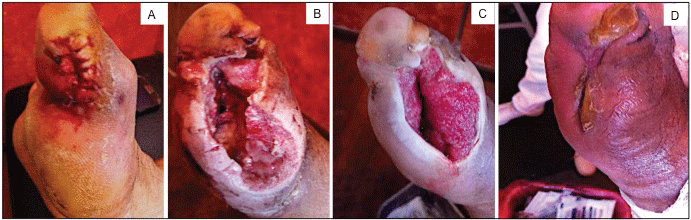 Figure 2: Chronological evolution of the lesion of a diabetic foot ulcer classified as Wagner grade III of 48-years-old male with controlled type 2 diabetes. A) Initial appearance of the ulcer; B) Ulcer after debridement, evolution of the DFU after; C) 8 days; D) 21 days of treatment with AgNPs solution.
View Figure 2
Figure 2: Chronological evolution of the lesion of a diabetic foot ulcer classified as Wagner grade III of 48-years-old male with controlled type 2 diabetes. A) Initial appearance of the ulcer; B) Ulcer after debridement, evolution of the DFU after; C) 8 days; D) 21 days of treatment with AgNPs solution.
View Figure 2
In the case of the second patient, the evolution of the healing process was monitored as shown in Figure 3. The DFU represents a grade II ulcer according with the Wagner classification. The wound is located at the plantar region of the left foot. As observed in Figure 3A, patient presented two plantar ulcers, the first of approximately 2 cm of diameter at the level of first metatarsal, classified as second-grade Wagner. The second ulcer is presented at the bony prominence. Patient exhibits hyperkeratosis and dermatitis. Daily treatment of topical administration of AgNPs solution was started after the debridement of the lesion. After 11 days of AgNPs administration (Figure 3B), the ulcer showed a decrease in hyperkeratosis and dermatitis. The ulcer previously located at the bony prominence is almost imperceptible and the area has regained its normal appearance. The plantar ulcer shows a decrease of edema and an improvement of the pigmentation indicated an active edge of healing, indeed, no fetid odor was perceived. After 12 days of AgNPs treatment (Figure 3C), the pigmentation of the wound is adequate. No necrosis data are detected and granulation tissue at the center of the ulcer and active edges are also observed. Twenty-six days after the beginning of treatment with AgNPs (Figure 3D), hyperkeratosis is minimal at the surrounding of the wound. However the skin has regained its normal characteristics. The ulcer edges have been addressed, culminating in the closure of the injury where no necrosis or inflammation was observed and normal re-epithelialization process and a normalization of the pigmentation were evident.
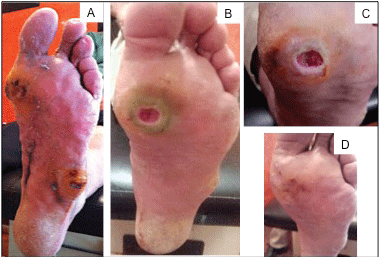 Figure 3: Chronological evolution of an ulcer Wagner grade II of a diabetic male patient of 54-years-old with type 2 diabetes. A) Initial appearance of the ulcer. Evolution of the DFU after; B) 11 days; C) 12 days; D) 26 days of treatment with AgNPs solution.
View Figure 3
Figure 3: Chronological evolution of an ulcer Wagner grade II of a diabetic male patient of 54-years-old with type 2 diabetes. A) Initial appearance of the ulcer. Evolution of the DFU after; B) 11 days; C) 12 days; D) 26 days of treatment with AgNPs solution.
View Figure 3
According with the WHO, it is estimated that by the year of 2025 the persons with diabetes will be 330 million, and the prevalence will be focused in developing countries [23]. Severe complications of DM are the ulcers in lower limbs. And as the number of diabetic patient continues to rise, so will be the number of DFU.
Therefore, DFU has been recognized as a worldwide health problem, especially in developing countries [23].
Because DFU are multifactorial, can be originated by minor trauma such as shoes compression, ingrown toenail and hits. In those cases diabetic patients are not able to perceive pain due to the peripheral neuropathy and the loss of propioceptive sensitivity. Therefore an injury could easily developed and make diabetic patients more vulnerable to foot infections by multi-drug resistance microorganisms [24,25].
Despite of the well-studied antimicrobial properties of AgNPs, silver-containing dressings are exclusively used to avoid microbial infections during wound healing process and tissue engineering. However, their usage for the treatment of infected wounds are not included within their advantages [10,26].
It has been reported that the delay of DFU are correlated with low levels of growth factor in wounds [27]. To overcome this, it has been proved that AgNPs are able to promote wound healing process through the proliferation and migration of keratinocytes [28]. Worth to mention is that AgNPs promote wound healing due to induction of fibroblast differentiation to myofibroblasts [28]. Thus, AgNPs could exert wound healing due to the abolishment of infections by Gram-negative or Gram-positive bacteria [29]. This results in a diminishment of edema along with the differentiation of cell lineages that promote a faster closure and reepithelialization of the wound.
Argovit AgNPs formulation used in this work has been approved by international instances as food supplement, cosmetic, coating of medical devices for human surgery and veterinary medicine [21]. Additionally, Argovit has another advantage being the only AgNPs formulation proved to have non-genotoxic effects on cell lines for concentrations close to the IC50 (3.5 μg/mL of metallic silver) [22,30]. This allows us to use this AgNPs formulation as an alternative to conventional drugs and safe formulation for the treatment of DFU [22,30].
To the best of our knowledge, this work represents the first nanomedicine approach study performed in the world for the treatment of DFU with AgNPs. It is important to indicate that the most ideal way to solve the problem of diabetic foot ulcers is to prevent their occurrence. Great advance in this area was made with development of a portable system which monitors the microclimate in footwear and foot interfaces [31].
Regarding with our study, further analysis of the type of microorganisms associated to the DFU, vascular and histopathological analysis of the injury will be the second phase of our work to validate our findings of the improvement of DFU healing by the application of AgNPs. Therefore, this study lays the bases for further systematic clinical and basic research on the usage of AgNPs for the treatment of chronic ulcers such as DFU. The study in wound healing murine models will be performed to explain the cellular and immune mechanisms involved during the ulcer healing process exhibited on the diabetic patients treated with Argovit is envisaged. These studies will permit further optimization of the healing procedure.
According to our knowledge in this study we present for the first time a successful treatment of diabetic foot ulcers of Wagner classification degrees II and III with AgNPs administered topically into the wound. Daily topical administration of AgNPs solution with metallic silver concentration of 1.8 mg/ml causes an improvement of the wound healing in average in less than 25 days of treatment.
The authors wish to thank the funding through the CONACYT project No. 279889 and DGAPA PAPIIT-UNAMIT200114 project. Also to Dr. Leonel Cota for his support with studentships. Authors acknowledge Dr. Vasily Burmistrov from the Scientific and Production Center Vector-Vita (Russia) for kindly donated Argovit AgNPs formulation. K. Juarez-Moreno was awarded by the National Council of Science and Technology of Mexico as a CONACYT Research Fellow through the CONACYT project No.1073. Authors wish to thank Dr. Oxana Martyniuk and Sr. Francisco Ruiz Medina for their technical support for TEM acquisition images and Med. Isis Cabrera, Alejandra Rembao Hernández, Nora Carricury Chequer and Carmen Enriques Enriques for their support in wound healing procedures.
Ethical approval to report this case series was obtained from Instituto de Servicios de SaludPública del Estado de Baja California, Sección Hospital General de Tijuana (No. 000056 Septiembre de 2011). Also, written informed consent was obtained from the patients for their anonymized information to be published in this article.
The authors claim there is not any conflict of interest.