The pathogenesis of megaesophagus in chronic Chagas disease, which is caused by infection with the parasite Trypanosoma cruzi is compelling. Individuals with megaesophagus often present achalasia and disturbances of peristalsis and neuronal loss. Esophageal samples were obtained from 6 T. cruzi infected individuals with megaesophagus, 6 T. cruzi infected individuals without megaesophagus, and 6 noninfected individuals who underwent necropsy procedures. Using one antibody specific for neuronal Nitric Oxide Synthase (nNOS) and another specific for Protein Gene Product 9.5 (PGP-9.5), which is a pan-neuronal marker, we demonstrated the relative area of nitrergic innervation. Additionally, we analysed the area occupied by Interstitial Cells of Cajal (ICCs) with antibody specific anti-CD117. The analyses presented here show that the relative area of nitrergic innervation is reduced in T. cruzi infected individuals with megaesophagus. Furthermore, we demonstrated reduced CD117-immunostained areas in the esophagus of T. cruzi infected individuals. Statistical analyses revealed a positive correlation between the relative area of nitrergic innervation and the density of ICCs in the same infected individuals. Considering data from the literature, we raised the hypothesis that the loss of nitrergic nerve fibers and ICCs could be view as an interdependent phenomenon occurring in the esophagus of T. cruzi infected individuals and that it could contribute to peristalsis disturbances, to achalasia and to megaesophagus development.
Chagasic megaesophagus, Denervation, Nitrergic nerve fibers, Interstitial cells of cajal
Chagas disease is caused by the intracellular parasite Trypanosoma cruzi and affects approximately 6-7 millions individuals in Latin America still today [1]. The acute infection displays a variable clinical course, with high parasitemia and a high parasite load within affected tissues. In the chronic phase some patients remain asymptomatic but after a indeterminate time course of infection some of them could present symptoms that reflect cardiac and/or digestive tract involvement [2].
The digestive form of Chagas disease is characterised by dilatation of the Gastrointestinal (GI) tract and the esophagus and colon most often been affected, and may progress to megaesophagus and mega colon, respectively [3]. Individuals with chagasic megaesophagus usually present dysphasia, peristaltic dysfunction and achalasia, and partial or absent lower esophageal sphincter relaxation [4] witch develop in consequence of the denervation [5,6]. Structural damage of the Enteric Nervous System (ENS), denervation above 90% in the myenteric plexus [7], ganglionitis and periganglionitis have been widely described in the digestive form of Chagas disease [8,9]. It is well known, however, that motility regulation in the GI tract and lower esophageal sphincter relaxation is influenced not only by neuromediators, as Nitric Oxide (NO), but also by Interstitial Cells of Cajal (ICCs) [10-13].
The production of NO by neurons of the ENS, coupled with its transmission to the muscle in the esophagus, is necessary for sphincter relaxation. This process is essential for muscle relaxation during peristalsis and it allows for the passage of gut contents [14,15]. Commonly observed in several regions of the GI tract, the ICCs are responsible for generating and propagating "slow waves" to the muscle layers, constituting the pacemaker system [16,17]. This system regulates the membrane potential of smooth muscle cells. When peristaltic activation begins, the electrical activity of the ICCs sets the pace, the frequency and the amplitude of contraction waves. ICCs within the muscle layers have been shown to be in close proximity to varicose nerve fibers and smooth muscle cells and have led to the suggestion that this class of ICCs may be involved in or mediate enteric neurotransmission [18].
Studies in mice have suggested that intramuscular ICCs may play a critical role in NO-dependent neurotransmission in the GI tract and the reduction of both have been implicated as cause of dysfunction of the lower esophageal sphincter and esophageal peristalsis in nNOS-/- and ICC-/- mice [19,20]. Decreased nitrergic innervation and loss of ICCs have been observed in idiopathic achalasia and those factors, in conjunction, have been considered to be implicated in such pathological process [21,22]. Previous studies of individuals with the digestive form of Chagas disease, have reported a decrease of ICCs density in the esophagus and colon that were associated with megaesophagus and megacolon, respectively [23,24]. Other study has demonstrated relative decrease in the number of neurons in colon of chagasic patient with megacolon [25].
Therefore the present study was designed in order to evaluate, in the lower portion of esophagus from T. cruzi infected individuals, the relative area of nNOS Immunoreactive (IR) nerve fibers and ICCs density.
Esophageal samples were obtained from 6 T. cruzi infected individuals with megaesophagus, 6 T. cruzi infected individuals without megaesophagus and 6 noninfected individuals who underwent necropsy procedures at Federal University of Triângulo Mineiro (Uberaba, Minas Gerais, Brazil). The individuals previously lived in Uberaba, MG, Brazil, where the natural transmission of Chagas disease was interrupted more than 30 years ago. Research consent was obtained from family members prior to tissue procurement. Serological tests diagnostic for Chagas disease (ELISA, hemagglutination and immunofluorescent tests) were positive in all study individuals.
The noninfected group was composed of individuals with negative Chagas specific serology. These participants were also from the state of Minas Gerais (average age of 62.5 ± 20.42 years) and had an esophagus diameter of 1.4 ± 0.16 cm. Infected individuals without megaesophagus (average age of 55.5 ± 11.88 years) had an esophagus diameter of 1.3 ± 0.23 cm. Infected individuals with megaesophagus (average age of 52.5 ± 9 years) had an esophagus diameter of 3.3 ± 0.79 cm (Table 1).
Table 1: Study group. View Table 1
The diagnosis of megaesophagus was established from clinical data reporting esophagus obstruction and from radiological studies. Tissue samples were collected from the lower third of the esophagus far 5 cm of the cardia (the junction between the stomach and the esophagus). The tissue was processed in 4% neutral buffered formaldehyde solution and embedded in paraffin for immunohistochemical studies. Subsequently, the esophagus rings were cut into serial 5 μm sections and treated with either immunohistochemical or Giemsa staining.
Paraffin-embedded tissues from chagasic individuals and noninfected individuals were immunostained for ICCs and nerves fibers. Esophagus sections (5 μm thick) were deparaffinised with xylene and dehydrated in graded alcohols. Endogenous peroxide activity was inhibited by incubation with 4% hydrogen peroxide and 0.05 M sodium azide for 30 minutes. The slides were then incubated in 2% normal swine serum (SIGMA, MO, USA) in phosphate-buffered saline for 30 minutes and, subsequently, with polyclonal antibodies specific to receptor protein surface tyrosine kinase (c-Kit), anti-CD117 (DAKO, Carpinteria, CA, USA; code A4502, 1:25), PGP 9.5 (DAKO, Carpinteria, CA, USA; code Z5116, 1:50) or anti-nNOS against neuronal nitric oxide synthase (ABCAM, Cambridge, MA, USA; code ab106417, 1:25) and this incubation was followed by a second incubation with peroxidase conjugated rabbit anti-mouse antibodies (DAKO, CA, USA) for 60 min. Peroxidase activity was demonstrated by incubation with 3,3-diaminobenzidine (Sigma) and hydrogen peroxidase for 10 minutes. The slides were counterstained with Gill's haematoxylin (Sigma), dehydrated in graded alcohols and mounted in synthetic mounting media.
A Morphometric analysis of immune-stained areas was performed using an image analysis system (Kontron KS300 version 2.0; Kontron, Port Coquitlam, Canada) on 20 randomly selected fields (total area of 1180 × 103 μm2) of the inner muscle layer using one slide per individual. The relative area of nNOS-IR nerve fibers was calculated as follows: nNOS-IR area/PGP 9.5-IR area × 100.
To quantify the distribution of ICCs, we employed immunohistochemistry using an anti-CD117 polyclonal antibody, which also reacts with mast cells [26]. Therefore, to identify or exclude the presence of mast cells, consecutive sections underwent Giemsa staining and those images were captured and analysed.
After normality test, statistical analysis was performed using a one-way Analysis of Variance (ANOVA) and Turkey's Multiple Comparison post-test. A correlation analysis was performed using the Pearson parametric test. The values are expressed as the mean ± SD (standard deviation) and differences were considered statistically significant at p ˂ 0.05.
The inner muscle layer of the esophagus from serologically negative controls demonstrated abundant nNOS-IR nerve fibers (Figure 1A). A similar distribution of nNOS-IR nerve fibers was also observed in the inner muscle layer of the esophagus from infected individuals without megaesophagus (Figure 1B). However, infected individuals with megaesophagus showed a decrease in nNOS-IR nerve fibers density (Figure 1C). Morphometric analysis of immunohistochemistry treated tissue sections revealed that individuals infected with megaesophagus have a significant reduction in the relative area of nNOS-IR nerve fibers compared to noninfected individuals, but no significant difference was observed between individuals infected without megaesophagus and the remaining two groups (Figure 2).
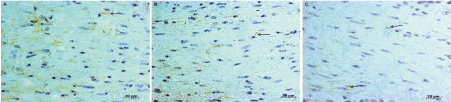 Figure 1: A) immunohistochemical profile of nNOS-IR nerve fibers (arrows) in the inner muscle layer of esophagus from noninfected individuals; B) Infected individuals without megaesophagus; C) Infected individuals with megaesophagus (Original magnification of 400x). View Figure 1
Figure 1: A) immunohistochemical profile of nNOS-IR nerve fibers (arrows) in the inner muscle layer of esophagus from noninfected individuals; B) Infected individuals without megaesophagus; C) Infected individuals with megaesophagus (Original magnification of 400x). View Figure 1
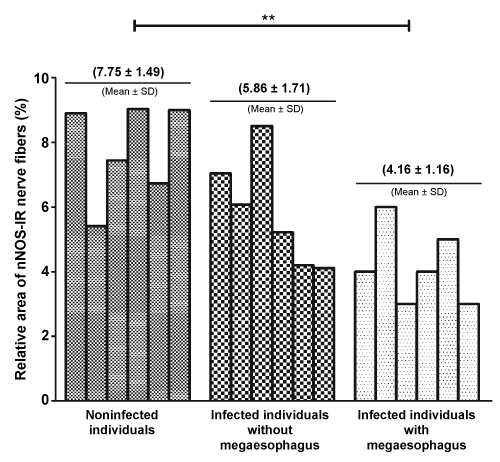 Figure 2: Data from morphometric analysis of relative area of nNOS-IR nerve fibers in the inner muscle layer of esophagus from noninfected individuals, infected individuals with and without megaesophagus. The bars express individual values and the mean ± SD is presented above each group.
Figure 2: Data from morphometric analysis of relative area of nNOS-IR nerve fibers in the inner muscle layer of esophagus from noninfected individuals, infected individuals with and without megaesophagus. The bars express individual values and the mean ± SD is presented above each group.
**Statistically significant differences between the noninfected individuals and the infected individuals with megaesophagus were observed (p < 0.01). View Figure 2
To determine the distribution of ICCs, we employed an anti-CD117 polyclonal antibody, which also reacts with mast cells. Therefore, to identify or exclude the presence of mast cells, consecutive sections were prepared and subjected to Giemsa staining (Figure 3A and Figure 3B). Within the inner muscular layer, CD117-IR ICCs were observed among the smooth muscle cells and exhibited an elongated shape, oval nuclei and prominent bipolar ramifications, which may or may not be associated with other ICCs (Figure 3C and Figure 3D). A morphometric analysis revealed an equally significant decrease in the density of CD117-IR ICCs in both groups of individuals infected, with and without megaesophagus (Figure 4).
 Figure 3: A) ICC (arrow) and mast cell (arrowhead) immunoreactive to CD117 (original magnification of 630x); B) Mast cells observed with giemsa staining (arrowhead; original magnification of 630x); C) Distribution of CD117 ICC-IR in the inner muscle layer of the esophagus from noninfected individuals (arrow; original magnification of 630x); D) Morphological profile of ICC (original magnification of 1000x). View Figure 3
Figure 3: A) ICC (arrow) and mast cell (arrowhead) immunoreactive to CD117 (original magnification of 630x); B) Mast cells observed with giemsa staining (arrowhead; original magnification of 630x); C) Distribution of CD117 ICC-IR in the inner muscle layer of the esophagus from noninfected individuals (arrow; original magnification of 630x); D) Morphological profile of ICC (original magnification of 1000x). View Figure 3
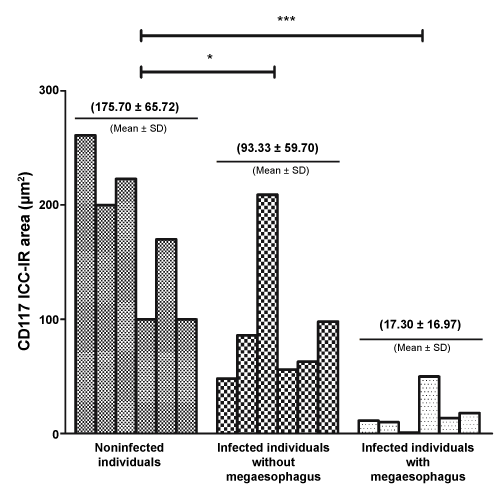 Figure 4: Morphometric analysis of the CD117 ICC-IR area in the inner muscle layer of esophagus from noninfected individuals and infected individuals, with and without megaesophagus. The bars express individual values and the mean ± SD is presented above each group.
Figure 4: Morphometric analysis of the CD117 ICC-IR area in the inner muscle layer of esophagus from noninfected individuals and infected individuals, with and without megaesophagus. The bars express individual values and the mean ± SD is presented above each group.
*Statistically significant differences between the noninfected individuals and infected individuals without megaesophagus were observed (p < 0.05).
***Statistically significant differences between the noninfected individuals and infected individuals with megaesophagus were observed (p < 0.001). View Figure 4
Furthermore, statistical analyses of the data obtained from infected individuals revealed a positive correlation between the relative area of nNOS-IR nerve fibers and the area of CD117-IR staining (r = 0.6263 and p = 0.0293; Figure 5).
 Figure 5: Correlation analysis between relative area of nNOS-IR nerve fibers and CD117 ICC-IR area in esophagus from T. cruzi infected individuals with and without megaesophagus (r = 0.7567 and p = 0.0044). View Figure 5
Figure 5: Correlation analysis between relative area of nNOS-IR nerve fibers and CD117 ICC-IR area in esophagus from T. cruzi infected individuals with and without megaesophagus (r = 0.7567 and p = 0.0044). View Figure 5
We present a semi-quantitative study of ICCs and of the nNOS-IR nerve fibers density in T. cruzi infected individuals with and without megaesophagus. To the best of our knowledge, no previous studies have demonstrated that the distal esophagus of individuals infected with megaesophagus have a decreased relative area of nNOS-IR nerve fibers compared to noninfected individuals. Neural NOS (nNOS) is an enzyme found in neurons supplying the muscle of the gastrointestinal tract. The production of NO by these neurons and the transmission of this NO to the muscle of the gastrointestinal tract, is necessary for sphincter relaxation. This process allows for the passage of gut contents and is essential for muscle relaxation during peristalsis [14,15]. Decreased nitrergic innervation has been observed in idiopathic achalasia (inability of the sphincter to relax) and is considered an important factor in its pathogenesis [21-22]. Here, we found decreased nitrergic innervation in infected individuals with megaesophagus. Previous studies have demonstrated the presence of achalasia in almost 100% of chagasic individuals with megaesophagus and also in some asymptomatic T. cruzi infected individuals [27,28]. Here, we demonstrated that a subgroup of individuals infected without megaesophagus also have a loss of nitrergic nerve fibers in the lower esophagus. Therefore, it is possible that the reduction of nitrergic innervation in T. cruzi infected individuals contributes to the failure of peristalsis and to the inability of the sphincter to relax. Unfortunately, due to the lack of manometric study data and dysphasia complaints within our group, it is not possible to evaluate this hypothesis at this time.
Regarding the analyses of the ICCs, reduced CD117-immunostained areas were observed in the inner muscular layer of the esophagus from infected individuals with megaesophagus, confirming the results from a previous study [23]. One novel finding was the observed decrease in CD117 immuno-staining in individuals infected without megaesophagus. Because these individuals had the initial or the mild form of digestive disease, we propose that the loss of ICCs happens early in the digestive disease before dilation of organ. In the GI tract, ICCs in the inner muscle are thought to mediate neurotransmission between enteric neurons and smooth muscle cells. The ICCs receive neuronal input and subsequently stimulate the smooth muscle cells [18]. Morphological studies corroborate this hypothesis and have shown that within the inner muscle of the GI tract, ICCs are physically interposed between varicosities of nerve endings and adjacent smooth muscle fibers. ICCs both establish synaptic contacts with neurons and form gap junctions with smooth muscle cells and the ICC-to-ICC interactions facilitate neurotransmission [29,30]. Thus, it is tempting to speculate that during early chronic T. cruzi infection, the loss of ICCs can lead to motility alterations and disturbances of sphincter relaxation and, consequently, can contribute to the development of megaesophagus.
The question of whether the loss of ICCs during T. cruzi infection is primarily caused by the accelerated death of mature cells or by the impaired regeneration of these cells was not addressed in this study. It is well known that, like other cells, the number of ICCs is controlled by several factors that regulate proliferation and death and that ICCs continuously undergo apoptosis in the GI tract of healthy individuals [12-31]. Because neurons produce Stem Cell Factor (SCF), which activates the c-kit receptor and stimulates the proliferation, survival and differentiation of ICCs [32], the denervation process may be one of multiple causal factors that lead to ICCs loss in the esophagus of T. cruzi infected individuals.
An interesting observation in this study was that CD117-IR ICC area in T. cruzi infected individuals positively correlates with the relative area of nNOS-IR nerve fibers. Although data from correlation analyses should not be interpreted as causative, those data in conjunction with data from previous studies suggest that alterations in both nitrergic innervation and ICC density may be viewed as inter-dependent events. A decrease in ICCs density in the muscle layers has been associated with reduction of the NO-dependent inhibitory response [33] and several other studies in the literature have implicated ICCs in NO-dependent neurotransmission [11,18,29,34]. Moreover, NO is considered to be a cytoprotective molecule that affects the survival of the ICCs [35,36].
In conclusion, this study of individuals with Chagas disease suggests that the loss of nitrergic innervation and ICCs may be implicated in the development of megaesophagus. Furthermore, our data, which are consistent with data from previous studies, show the inter-dependence between nitrergic innervation and ICCs and suggest the importance of continuing these investigations. Future studies, employing a combination of clinical and pathological research will be essential in furthering our understanding of the natural history of Chagas disease.