Cerebral venous thrombosis is not a frequent condition and may follow unsuspected, with unspecific manifestations. Neuroimaging studies are mandatory in differential diagnosis, which includes arterial strokes and other benign and malignant causes of intracranial hypertension, headaches, seizures, and hemiparesis. Characteristic images are triangle and the empty delta signs, and loss of flow void. Images of total or partial empty sella can be associated with secondary or idiopathic intracranial hypertension. Primary type occurs in up to 35% of cases, mainly affecting middle aged obese and hypertensive women. Membranous subaortic stenosis is a congenital or acquired change of the left ventricular outflow tract, with connections to the aortic and mitral valves; aortic regurgitation usually develops. Partial empty sella, sagittal sinus thrombosis and membranous subaortic stenosis are herein described in an obese woman, aiming to enhance the interest for new studies to better clear the possibility of some causal relationship among them.
Empty sella, Sagittal sinus thrombosis, Sub aortic stenosis
Cerebral Venous Thrombosis (CVT) is relatively uncommon, but with significant neurological sequels and up to 20% of mortality [1,2]. Because of the unspecific manifestations, clinical suspicion is challenging and imaging studies are needed to establish early diagnosis and adequate control [1-3]. CVT can origin Intracranial Hypertension (IH) and main symptoms are headaches, seizures, and hemiparesis; major neuroimaging signs are brain edema, cerebral hematoma and subarachnoid hemorrhage [1-5]. Secondary IH (SIH) may be due to tumor, infection, obstruction of CSF flow, and arteriovenous fistula [1-3,6-10]. CVT cab be confirmed by images of triangle and empty delta signs, and loss of flow void [1,10,11]. CVTs are less common than arterial thromboses, but under diagnosis occur without imaging evaluations; arterial stroke and other benign and malignant disorders are differential diagnosis [1-3].
The sella turcica may be partially or totally filled with cerebrospinal fluid and the hypophysis is compressed; in cases of shrinking or flattening of the gland, Magnetic Resonance (MR) study can show the sella appearing totally or partially empty - the empty sella syndrome [3,4,6-13]. This condition may be related to SIH or Idiopathic IH (IIH), but the gland volume has no correlation with Intracranial Pressure (ICP) [3,4,9]. IIH (or pseudotumor cerebri), affects up to 35% of population, mainly middle aged obese and hypertensive women (5:1 compared to men) [3-10,12]. Obesity is one major risk factor of IIH, and has growing prevalence in Western countries; with visual involvement in up to 90% of cases, causing severe deficits in up to 24% of them [12]. Disorders of arterial inflow and venous outflow on the mechanisms of IIH include narrowed venous sinuses without thrombosis, and can be related to features of total or partial empty sella [4,5,10]. Both types of IH should be investigated by neuroimaging studies including MR and MR/CT venography, because sinus venous occlusions can be the etiology of isolated intracranial hypertension in more than 30% of cases [4,10].
A 25-year-old woman was admitted on April 27 with inappetence, vomiting and weakness for 30 days, in addition to mild oppressive chest pain, dyspnea on functional class NYHA II/ III. She was using a beta-blocker and a Total Atrioventricular Block (TAVB) was detected; there was also behavioral and emotional changes (unmotivated laughter, dyslalia, and discordant speech). After sudden intense left temporoparietal headache she had a generalized seizure and clinical evaluation discarded TAVB, blood pressure changes, or hypoxemia [Blood Pressure (BP): 110 × 60 mmHg, Heart Rate (HR): 51 bpm, and SatO2: 98%]. She denied allergy, tobacco smoking and alcoholism; as well as familial cardiovascular diseases, but had subaortic membrane stenosis submitted to surgery two years before. With six years of age she had bilateral spontaneous dislocation of the crystalline and a heart murmur. Prompt surgical correction of her crystalline disorder was successfully performed, and she did not present remarkable visual impairment during the current admission. The dyspnea started ten months before and worsened to functional class NYHA III, associated with palpitation and dizziness. She utilized oral anticoagulant during four years and stopped in the last two months; used paroxetine, valproic acid and quetiapine for one year; and was in regular use of AAS (100 mg daily) and sertraline (50 mg daily) for three months. On physical examination: conscious, oriented, afebrile, without cyanosis or jaundice; BMI: 30.45 kg/m2, HR: 48-52 bpm, BP: 100 × 60 mmHg; respiratory rate: 16 irpm, SatO2: 98%; temperature: 36.1 ℃; regular heart rhythm; systolic aortic murmur (grade 3); jugular veins; lungs, abdomen and extremities unremarkable. Routine blood determinations are shown in Table 1. Cardiac catheterization on February 2017: Absence of coronary lesions, Left Ventricle (LV) with normal contractile function, and stenosis of the LV outlet via with a gradient of 50 mmHg. Cardiac Magnetic Resonance (MR) on March 2017: Normal contractile function of the LV, preserved myocardial integrity, and significant turbulence of the blood flux at the subaortic region due to the presence of a membrane. Holter on March 2017: Predominance of sinus rhythm with AVB type 2:1 and periods of AV dissociation during the sleep; maximal HR: 72 bpm, mean HR: 49 bpm, and minimal HR: 38 bpm; QRS within normal range, and six ventricular extra-systoles during the exam. Other studies of the heart and nervous system were done on May 2017. Transthoracic echocardiogram: Accentuated residual subaortic stenosis; concentric remodeling of the LV with preserved global and segmental function [LV Ejection Fraction (EF) Teicholz: 84%]; discrete mitral and aortic insufficiency. Transesophageal echocardiogram: Sub aortic stenosis measuring 3 mm × 3 mm (Figure 1); systolic peak gradient: 43 mmHg, with maximal velocity: 328 cm/second; discrete aortic, mitral and tricuspid insufficiency; preserved biventricular systolic functions; absence of thrombus and of vegetation; systolic pressure of pulmonary artery: 34 mmHg, and LVEF: 72.51%. Electrophysiological study: Sinus rhythm; abnormal AV conduction, fixed AVB 2:1 without response to atropine; and normal conduction by the His-Purkinje and intraventricular systems. Computed Tomography (CT) of the brain: Diffuse calcification of falx cerebri and choroid plexus, and suggestive image of empty sella and absence of bleeding sign (Figure 2A and Figure 2B). Angio-CT of the brain: extensive filling failure on superior sagittal sinus, consistent with venous thrombus and left frontal ischemia showed by cortical and subcortical hypoattenuating areas (3.1 × 3.0 × 2.8 mm); and normal polygon of Willis (Figure 2C and Figure 2D). MR of the brain: Focus of hyper-signal in T2-weighted sequences in the cortical-subcortical white and gray matter, at the transition of the left frontal and parietal lobes in the middle cerebral artery territory measuring about 3.7 mm, with restriction in the sequence of diffusion, indicative of ischemic vascular event; and absence of other abnormalities (Figure 2E, Figure 2F and Figure 2G). Angio-MR of the brain: Images in the sagittal plane showed failure of sagittal sinus filling indicative of thrombus, and presence of the pituitary gland (Figure 2H). Control blood tests (normal values in parenthesis) on May 2017: HBsAg: Negative; anti-HBs: Positive; IgG anti-HBc: Negative; IgM anti-HBc: Negative; anti-HBe: Negative; anti-HCV: Negative; HIV test: Negative; CMV IgM: Negative; CMV IgG: Negative; anti-Toxoplasma gondii IgG and IgM: Negative; anti-Epstein Barr virus IgG: Positive; and IgM: Negative; anti-Treponema pallidum: Negative; anti-ADN: Negative; anti-Ro (< 7): 0.4 U/ml, anti-La (< 7): 0.3 U/ml, anti-Sm (< 7): 0.8 U/ml, anti-RNP (< 5): 0.9 U/ml; IgA anti-cardiolipin (< 14): 10 APL U/ml; IgM anti-cardiolipin (10-40): 18 MPL U/ml; lupus anticoagulant: Negative; antithrombin III (79-112): 57.75; beta-2-glycoprotein I IgA (< 20): 9 U/ml; beta-2-glycoprotein I IgM (< 20): 11 U/ml; factor X (70-120): 87.6%, functional C-protein (72-150): 90.2%, S-protein (60.1-113.6): 102.6%, and factor V Leiden: Negative. After diagnosis of sagittal sinus thrombosis and pace-maker implant, full enoxaparin and warfarin were used; she evolved without neuropsychiatric or cardiac symptoms. Currently, her follow-up is on neurology and arrhythmology services.
Table 1: Routine exams of a 25-year-old female with sagittal sinus thrombosis. View Table 1
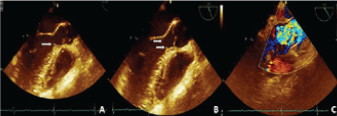 Figure 1: A,B) Echocardiography showing Left Ventricle (LV) with normal dimensions and sigmoid septum measuring 10 mm, with a small residual subaortic membrane of 3 mm × 3 mm (arrows); C) (color-Doppler) Turbulence in the LV outflow tract (with peak systolic pressure gradient of 43 mmHg and maximal velocity of 328 cm/s). View Figure 1
Figure 1: A,B) Echocardiography showing Left Ventricle (LV) with normal dimensions and sigmoid septum measuring 10 mm, with a small residual subaortic membrane of 3 mm × 3 mm (arrows); C) (color-Doppler) Turbulence in the LV outflow tract (with peak systolic pressure gradient of 43 mmHg and maximal velocity of 328 cm/s). View Figure 1
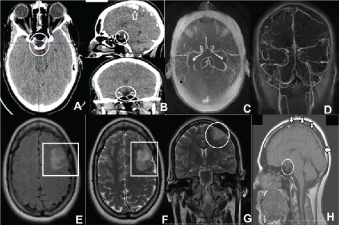 Figure 2: A,B) CT of the brain with contrast, and reconstruction in sagittal and coronal planes, showing the empty sella (encircled) and calcification of the cerebral falx (arrow); C,D) Angio-CT of the brain revealing a filling defect in the superior sagittal sinus and left frontal ischemic changes characterized by cortical and subcortical hypoattenuating areas; and normal polygon of Willis; MR of the brain with emphasis in the areas of hypoperfusion: E) In T2 flair; F) In T2 axial with diffusion; G) In T2 coronal plane; H) Angio-MR of the brain in T1 sagittal plane showing the filling defect in the sagittal sinus indicative of thrombosis, and the hypophysis (encircled). View Figure 2
Figure 2: A,B) CT of the brain with contrast, and reconstruction in sagittal and coronal planes, showing the empty sella (encircled) and calcification of the cerebral falx (arrow); C,D) Angio-CT of the brain revealing a filling defect in the superior sagittal sinus and left frontal ischemic changes characterized by cortical and subcortical hypoattenuating areas; and normal polygon of Willis; MR of the brain with emphasis in the areas of hypoperfusion: E) In T2 flair; F) In T2 axial with diffusion; G) In T2 coronal plane; H) Angio-MR of the brain in T1 sagittal plane showing the filling defect in the sagittal sinus indicative of thrombosis, and the hypophysis (encircled). View Figure 2
In 1994, for the first time Haisa, et al. reported the association between empty sella and superior sagittal sinus thrombosis [8]. The 61-year-old woman had a dural arteriovenous malformation and was in use of medroxyprogesterone acetate after her surgical treatment for breast cancer [8]. The authors emphasized the major role of thromboembolic events due to drug side-effects, and commented the hypothesis of a relationship between the arteriovenous malformation and the sinus thrombosis [8]. Moreover, thrombosis might be related to cancer-associated hypercoagulable state.
In this case, the main risk factors for thrombosis included: Female gender (in spite of young age and no contraceptive drugs); congenital or acquired thrombophilic disorders (deficiency of proteins C and S, high levels of fibrinogen and of factors VII, VIII and X, hyperhomocysteinemia, factor V Leiden mutation, and antiphospholipid antibodies). None of these risk factors was confirmed. Although not discarded, the sagittal sinus calcification seems did not have a significant role in the CVST of this patient. A concern might be about changes on blood brain barrier allowing exposure to blood of prothrombin stored in neural tissue, which might initiate local thrombosis.
The young obese woman herein described presented acute neuropsychiatric disturbances associated with severe temporoparietal headache followed by generalized seizure. The diagnosis of sagittal venous thrombosis with SIH was characterized by the evaluation of neuroimaging procedures including MR and MR/CT venography studies. There was diffuse calcification of falx cerebri and choroid plexus, partial empty sella, and absence of hemorrhage. The panel of blood routine determinations related to prothrombotic disorders revealed unremarkable results. Therefore, idiopathic CVST with obstruction of the sagittal sinus was considered the most probable diagnosis; hypothesis that is in accordance with the literature about this condition affecting obese young females [3,5,7-10,12]. Worthy of note were the psychiatric manifestations that might be associated with the empty sella syndrome [6]. The patient underwent low-molecular-weight-heparin and warfarin schedule and improved of neuropsychiatric changes, and during admission there was no hyponatremia; but its episodic role was not ruled out in the present case [13]. One must remember that up to 50% of patients with empty sella syndrome have endocrine dysfunction, in special hypopituitarism with recurrent hyponatremia that can be the cause of anxiety, depression, mania, and psychosis [13].
CVT on the sinuses (CVST) may have poor outcome and accounts for up to 20% of strokes in young Indians [2]. This condition represents approximately 30% of all strokes in that population and 60% occur in the postpartum [2]. CVST is related to infection; dehydration; prothrombotic disorders as factor V Leiden polymorphism, protein C and S resistance, antithrombin III deficiency; and vasculitis of systemic lupus erythematosus or polyarteritis nodosa [2]. Patil, et al. evaluated 50 patients with cerebral sinus thrombosis, 58% were females with mean age of 29 ± 7 years, and the main manifestations were headache and vomiting (90%), seizures (26%), and hemiplegia (24%); the occlusive thrombi were found in sagittal sinus (46%), sigmoid/transverse sinus (32%), and multiple sinus (20%) [2]. The authors described the superior sagittal sinus thrombosis as the most often affected site; 30% of women were in peripartum period; infection was a major risk factor; and morbidity and mortality was lower than arterial strokes [2]. Ridha, et al. compared 60 patients with abnormal images on MR venography, 29 with IIH (28 women, median age: 28, and median BMI: 34) and 31 with CVT (19 women, median age: 46, and median BMI: 29), for evaluation of venous sinus stenosis or cerebral venous thrombosis, and the measurement of the sella turcica [4]. All patients with IIH had bilateral transverse sinus stenosis; among the group of CVT patients, 3 had thrombosis of the sagittal, 3 of sigmoid, 1 of cavernous, 7 of unilateral transverse, 16 of multiple sinuses, and 1 of cortical veins [4]. The total or partial empty sella was more common in IIH (3/29 and 24/29) than in CVT groups (1/31 and 19/31) [4]. Tang, et al. reviewed 17 patients with CVT showed by MR venography; all had sagittal sinus thrombosis with or without transverse sinus thrombosis; none had straight sinus or cortical vein thrombosis [12].
Other concern is about Discrete Membranous Sub aortic Stenosis (DMSS) at left ventricular outflow tract; a congenital or acquired change with aspect of normal endocardium and fibrous connections with the aortic and mitral valves, with etiopathogenic mechanisms not well-understood [14,15]. DMSS is considered a progressive condition that may evolve from a thin membrane less than 3 mm like a fibrous diaphragm on the anterior portion of the left ventricle to a muscular obstruction [15]. The high-pressure subaortic jets may injure aortic cusps causing aortic regurgitation; hemodynamic sequels vary from mild peak gradients (< 30 mmHg) to severe (> 70 mmHg) [15]. The patient herein described presented a DMSS measuring 3 mm × 3 mm, and the systolic peak gradient was 43 mmHg; and the noninvasive option of management was for use of ivabradine 5 mg twice daily and watchful expectancy. Butany, et al. reviewed clinical and morphological features of excised DMSS of 19 adults (26 to 75 years, 68.4% females), and 21% of them had other congenital heart disease [14]. Only four patients had murmurs or membranes in childhood, and the membranes developed on the ventricular surface of anterior mitral leaflet 99.4% of cases [14]. The lesions had five layers: Endothelial, acid mucopolysaccharide-rich subendothelial, collagen-rich fibrous, fibroelastotic, and smooth muscle. Aortic regurgitation was a very common complication (63%) of DMSS, and conduction abnormalities due to correction by septal myectomy were observed in 47.3% of the patients [14,15]. DMSS may develop in adults following some surgical procedure for underlying congenital heart disease [14,15], and may play a role in the CVST by the occurrence of endocarditis and cerebral embolism. The present case study could have clinical usefulness, if one considers that some clinical combinations of the cerebral venous sinuses thrombosis would allow timely and adequate diagnosis of severe disorders of the venous circulation of the brain. With the inherent weaknesses of a single case study, it was not possible to establish consistent relationship between DMSS and the pathogenesis of CVST in this patient. In fact, DSSM can be considered a challenging entity with unresolved uncertainties with respect to origin, pathogenesis, natural history, treatment, and recurrences [15].
In conclusion, the coexistence of empty sella, sinus thrombosis, and subaortic stenosis, might enhance the interest for researches about some causal relationship.
• Santos VM, MD, PhD drafted the case report, critically revised the text, and approved all corrections made to the manuscript, including the final version.
• Weiber RP, MD critically revised the case presentation and made substantial corrections to all the drafts.
• Cardoso RAR, MD obtained the images in the case study, critically revised the text and made a substantial contribution to the final draft.
• Helbingen MFS, MD and Amaral BCR, MD critically revised all data pertaining to the patient's time in the hospital and made important corrections to the initial draft.
All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Yes.
None to declare.
None to declare.