Knowing the predictors of quality of life (QoL) of children with osteogenesis imperfecta (OI) can optimize their treatment. The present study evaluated a longitudinal cohort of children and adolescents in a reference center for OI. The results showed that we must target reducing fractures and pain and focus on rehabilitation.
Purpose: To evaluate the predictors of quality of life (QoL) of children and adolescents with osteogenesis imperfecta (OI).
Methods: Descriptive cross-sectional study. Data were taken from a longitudinal cohort of children and adolescents with OI from 2 to 16-years-old on Pamidronate treatment in a reference center for OI in Southern Brazil. Pediatric Quality of Life Inventory 4.0 (PedsQL 4.0) was used for the evaluation of QoL.
Results: Twenty-four patients answered the questionnaires. Patients with more severe forms (types III and IV) had lower scores than those with mild form (type I) in the physical domain of QoL. Those with greater duration of treatment, total number of fractures and pain intensity had worse QoL scores.
Conclusions: Children with severe forms of OI presented lower QoL in the physical domain than those with mild forms. QoL scores were negatively correlated with duration of treatment, total number of fractures and pain intensity.
Osteogenesis imperfecta, Children, Quality of life, Fractures
Osteogenesis Imperfecta (OI) is a rare hereditary disease characterized by bone fragility or low bone mass. It is caused by a qualitative or quantitative defect of type 1 collagen, synthesized by osteoblasts [1,2]. The estimated incidence of OI is 1 case per 10,000 births [3]. Most patients diagnosed with OI have a mutation in one of the two genes, COL1A1 or COL1A2, which encode type 1 collagen, the main protein found in bone [4]. The presence of abnormal structural protein determines bone fragility. In this disease, there are fractures with minimal trauma and bone deformities. The severity of the findings is quite variable, from lethal forms of intrauterine fractures to fractures that will only occur in adolescence or adulthood. Some patients have impaired stature, as well as dentinogenesis imperfecta, blue sclera and ligament laxity, since type 1 collagen is also present in teeth, skin, tendons and sclera [1].
According to the clinical presentation, the classification of types I to IV by Sillence, et al. in 1979 is the most used so far [5,6]. Types V, VI, and VII have recently been included, and although the defect is not in the collagen gene, they are also characterized by bone fragility [7-9].
The diagnosis of OI is predominantly clinical and should be considered in any child with repetitive fractures at minimal trauma. Family history, clinical examination, and radiological findings are important for diagnostic confirmation [3].
Treatment involves physical therapy, rehabilitation, orthopedic surgery and, more recently, the use of bisphosphonates [10]. Intravenous pamidronate was the pioneer drug and is still the most widely used in pediatric patients, since younger children have difficulty swallowing the oral forms, often associated with gastroesophageal reflux, esophagitis and dyspepsia. Studies have shown pain reduction, improved mobility, increased bone mass, and decreased fracture rates [4,11].
Regarding the quality of life (QoL) of OI patients, we know that this condition can cause great physical limitation to patients, but there are few studies exploring this issue in children with OI [12,13].
This is a descriptive cross-sectional study. Data were taken from a longitudinal cohort of children and adolescents with OI from 2 to 16-years-old on Pamidronate treatment in Hospital Infantil Joana de Gusmão, a reference center for OI in Florianópolis, Southern Brazil. The variables analyzed were age, gender, type of OI according to Sillence classification, age at diagnosis and at beginning of treatment, fractures at birth and in the last year, total number of fractures, physical conditions, pain intensity and body mass index (BMI).
Pediatric Quality of Life Inventory 4.0 (PedsQL 4.0) is a modular approach to measuring health-related QoL. It integrates both generic core scales and disease-specific modules, and can be used in healthy children and adolescents and those with acute and chronic health conditions. Divided by age group, it presents 23 items that address the physical, emotional, social and school dimensions [14,15]. It's also validated in Brazil, in a study with 240 healthy children and adolescents from São Paulo (SP, Brazil) and 105 patients with chronic rheumatic diseases [16].
The PedsQL tools generate results by domains: physical, emotional, social and school functioning [14]. The items were scored inversely and transposed linearly on a scale of 0-100 (0 = 100, 1 = 75, 2 = 50, 3 = 25, 4 = 0). Thus, the higher the score, the better the QoL. Scale scores were computed as the sum of items divided by the number of items answered.
For children 2 to 4-years-old, the questionnaire was answered by the caregiver and for children 5 to 7-years-old, by the child using illustrative figures.
Pain intensity was assessed through a 0 to 10 scale, with "0" being no pain and "10" being the worst pain they have ever felt.
A total of 24 patients answered the questionnaire. Patients' ages ranged from 2 to 16 years, with a mean of 9 years and 6 months, with a slight majority being male (58%). The profile of patients is shown in Table 1.
Table 1: Profile of the patients according to the type of osteogenesis imperfecta (values in means and standard deviation). View Table 1
Patients with more severe forms (III and IV) had lower scores than those with mild form (I) in physical domain (48.7 ± 18.7 × 69.9 ± 18.8 respectively, p = 0.02), but there was no statistical difference in emotional, social and school domains (Table 2).
Table 2: PedsQL 4.0 scores according to the type of osteogenesis imperfecta (values in means and standard deviation). View Table 2
Total number of fractures had a negative correlation with physical (p = 0.04, r = -0.42), emotional (p = 0.006, r = -0.54), social (p = 0.003, r = -0.57) and school (p = 0.02, r = -0.47) domains of PedsQLTM 4.0 (Figure 1). However, there was no statistical difference in the QoL between the groups who have had or not fractures at birth, or in those who have had or not fractures in the last year.
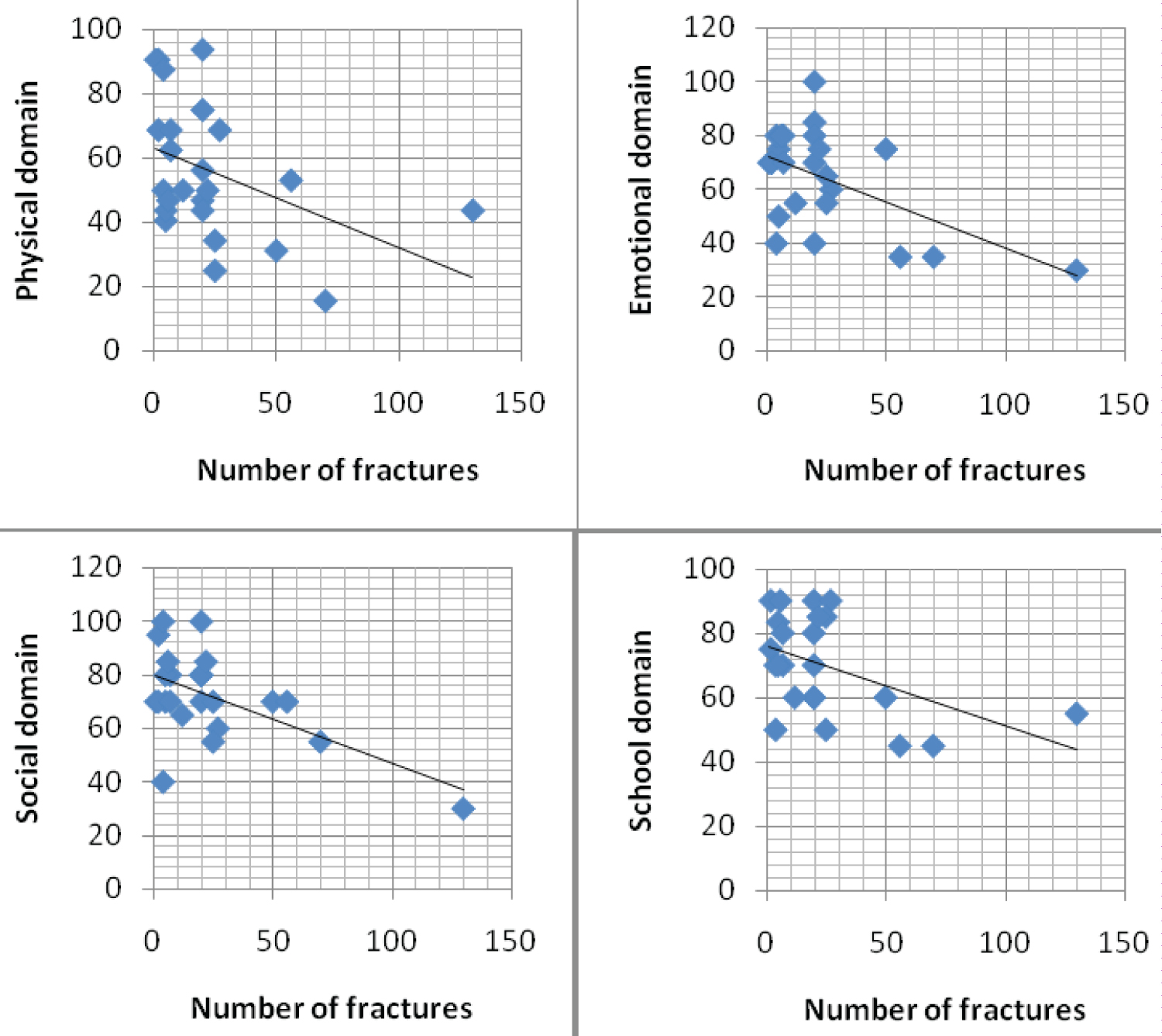 Figure 1: PedsQLTM 4.0 scores in physical (A), emotional (B), social (C) and school (D) domains according to the total number of fractures. View Figure 1
Figure 1: PedsQLTM 4.0 scores in physical (A), emotional (B), social (C) and school (D) domains according to the total number of fractures. View Figure 1
PedsQLTM 4.0 scores had a negative correlation with duration of treatment in physical (p = 0.0039, r = -0.57), social (p = 0.03, r = -0.43) and school (p = 0.01, r = -0.5) domains.
PedsQLTM 4.0 scores correlated negatively with pain intensity in emotional (p = 0.04, r = -0.41) and school (p = 0.02, r = -0.49) domains.
QoL of patients who were normal weight had no statistically difference from those who were overweight or obese. There was also no statistical difference in the quality of life between the sexes.
The type of OI had an impact only in the physical domain of QoL, with worse scores in more severe forms. Similar result was found by Fano, et al. evaluating the QoL of 65 children using the PedsQL. They found a decrease in the physical domain in types III and IV of OI, but not in the social domain [17].
The study of Dogba, et al. evaluated the impact of severe forms of OI of 12 adolescents and their parents with semi-structured interviews. Although the diagnosis can be devastating to families, most parents and patients have developed a new mentality, with a new concept of "normal life" characterized by ups and downs, challenges and changes as well as positive experiences [18].
A systematic review on QoL in OI of Dahan-Oliel, et al. involved 10 studies, 6 of them with children with OI, 3 with adults with OI and 1 with parents of children with OI. The physical dimension of QoL appeared to be lower than that of the general population, especially in more severe types of OI. However, in mental and psychosocial issues, individuals with OI had equal or higher QoL [19].
Lower number of fractures was associated with better QoL scores, as well as in the study by Fano, et al. [17]. Pain intensity was associated with worse scores in emotional and school domains. Hill, et al. conducted a qualitative study to propose a specific QoL questionnaire for patients with OI, identifying as important six themes: Feeling safety (particularly in relation to fractures), functional reduction, pain, fear, isolation and independence. The study suggests that the item "feel safe" should be explored in these patients [20].
Children with severe forms of OI presented lower physical functioning scores than those with mild forms. Duration of treatment, total number of fractures and pain intensity were negatively correlated with QoL scores.
No funding was secured for this study.
Juliana van de Sande Lee, Genoir Simoni, Ana Paula Vanz, Têmis Maria Félix, João Carlos Xikota, Sílvia Modesto Nassar, Maria Luiza Cimardi Rupp and Maria Marlene de Souza Pires declare that they have no conflict of interest.
Data is available upon formal request.
Data is available upon formal request.
Dr. Maria Marlene de Souza Pires conceptualized and designed the study, analyzed the database, drafted the initial manuscript, and reviewed and revised the manuscript; Dr. Juliana van de Sande Lee conceptualized and designed the study, collected data, analyzed the database, drafted the inicial manuscript, and reviewed and revised the manuscript; Dr. Silvia Modesto Nassar analyzed the database, drafted the inicial manuscript, and reviewed and revised the manuscript; MD, Genoir Simoni, Msc. Ana Paula Vanz, Dr. Têmis Maria Félix, Dr. João Carlos Xikota and Maria Luiza Cimardi Rupp drafted the inicial manuscript and reviewed and revised the manuscript.
All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Approval was obtained from the local hospital ethics committee. Informed consent was obtained from all individual participants included in the study.
Informed consent was obtained from all individual participants (or their legal guardians) included in the study.
Informed consent regarding publishing this study was obtained from all individual participants (or their legal guardians) included.
The authors acknowledge Universidade Federal de Santa Catarina, Hospital Infantil Joana de Gusmão and the patients with OI and their caregivers, for the cooperation to the present study.