A 53-year-old male with end-stage alcoholic liver cirrhosis and portal hypertension presented with hepatic encephalopathy as well as painful ambulation due to a mass on the medial aspect of his knee. The mass was first noted several months prior, concomitant with his presentation of decompensated liver failure. As a part of the pre-transplant work-up, multimodality imaging evaluation of the medial knee mass was completed and revealed a high-flow vascularized soft tissue lesion. Biopsy performed to exclude malignancy confirmed a high-flow vascular malformation, presumably acquired as a sequela of the patient's liver disease. While arteriovenous malformations (AVMs) are common congenital phenomena, this case represents one of the first reported instances of an acquired lower extremity AVM in a patient with end-stage liver disease.
Arteriovenous malformation (AVM), Vascular malformation, Liver cirrhosis
Vascular abnormalities can arise due to a wide range of disease states, happen throughout the body, and depending on their location, can go unnoticed by the patient and the clinician. Congenital vascular malformations commonly arise from errors of embryogenesis. However, vascular malformations can be acquired in the setting of a systemic disease such as liver failure. In this scenario, acquired lesions often present in the pulmonary system, but there are also two case reports of acquired AVMs in the upper extremities of liver failure patients. With an inadequately understood pathogenesis, acquired vascular malformations should be considered in the differential diagnosis for high-flow lesions found in patients with end-stage systemic manifestations of decompensated liver failure.
A 53-year-old man with a past medical history of alcoholic liver cirrhosis and portal hypertension presented with confusion, weakness, and ascites. He also presented with left knee pain accompanied by a large palpable mass that had developed along the medial aspect of the left knee. The patient reported he first noted the painful mass at the onset of his liver failure, 8 to 12 months prior to the current presentation. He stated the mass had continued to slowly grow in size. He asserted no history of surgery or prior mass in this location predating the liver failure. He recalled a minor "injury" to the left knee as a child that subsequently resolved with conservative management and no further work-up or intervention. The patient has no family history of an angiomatous syndrome. He had been admitted two months prior for the emergent banding of bleeding esophageal varices. On physical examination, the patient was cachectic and jaundiced. He had multiple sequelae of cirrhosis, including spider angiomata, hepatosplenomegaly, caput medusae, and palmar erythema. Diffuse lower extremity edema and ascites was also noted. A round, pulsatile, partially compressible mass was found on the left anteromedial distal thigh/knee (Figure 1). Laboratory results revealed thrombocytopenia (93 L/uL), hyponatremia (118 mmol/L), and hyperkalemia (6.5 mmol/K), as well as an elevated creatinine (1.14 mg/dL increased from baseline of 0.63 mg/dL), concerning for developing hepatorenal syndrome.
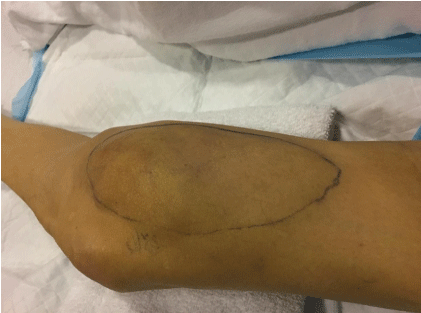 Figure 1: A 53-year-old male with end-stage alcoholic liver disease and a pulsatile painful mass along the medial left distal thigh/knee. Note the blue-purple discoloration of the skin overlying the outlined mass. View Figure 1
Figure 1: A 53-year-old male with end-stage alcoholic liver disease and a pulsatile painful mass along the medial left distal thigh/knee. Note the blue-purple discoloration of the skin overlying the outlined mass. View Figure 1
The primary medical team included joint effusion, hemarthrosis, hematoma, or primary sarcoma in the differential for the medial knee mass and ordered a non-contrast computed tomography (CT) of the left lower extremity. The CT demonstrated a 3.3 × 5.7 × 12 cm (AP × Trv × CC) hypoattenuating mass infiltrating the vastus medialis obliquus and obscuring the soft tissue plane with the adjacent popliteal vasculature (Figure 2). Numerous dilated vessels were noted in the surrounding tissues. There was no periosteal reaction or cortical changes in the adjacent bone.
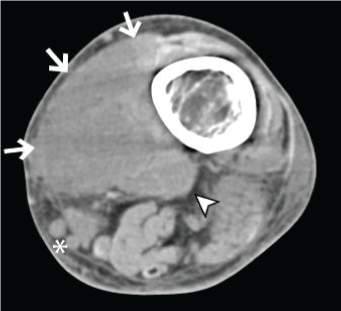 Figure 2: Hypoattenuating mass along the medial aspect of the distal femur (white arrows). The mass infiltrates the medial extensor muscles and abuts the popliteal vasculature (white arrowhead). Dilated subcutaneous draining vessels are noted adjacent to the mass (white asterisk). There is no osseous destruction, erosion, or periosteal reaction. View Figure 2
Figure 2: Hypoattenuating mass along the medial aspect of the distal femur (white arrows). The mass infiltrates the medial extensor muscles and abuts the popliteal vasculature (white arrowhead). Dilated subcutaneous draining vessels are noted adjacent to the mass (white asterisk). There is no osseous destruction, erosion, or periosteal reaction. View Figure 2
The radiological differential diagnosis for this lesion included a neoplasm (both primary soft tissue sarcoma and metastasis), hematoma, or vascular malformation. Based on the imaging appearance, a contrast-enhanced MRI was recommended for more definitive characterization.
MRI showed an ill-defined intermediate T1 (Figure 3a), hyperintense T2 (Figure 3b), homogeneously enhancing infiltrative mass that demonstrated diffuse enhancement on T1-weighted fat suppressed post gadolinium imaging (Figure 3c). Multiple flow voids from high-flow arterial vessels were present within the lesion. Several dilated feeder arteries, primarily arising from the distal profunda femoris and popliteal arteries, as well as enlarged draining veins were noted in the adjacent subcutaneous and deep tissues. Ill-defined portions of the lesion extended into the surrounding soft tissues and into the medial retinaculum. Patchy T2 hyperintense marrow edema was noted in the femoral diaphysis and distal metaphysis without a discrete marrow replacing lesion. Biopsy was recommended to differentiate a high-flow vascular malformation from hypervascular neoplasm, including metastasis.
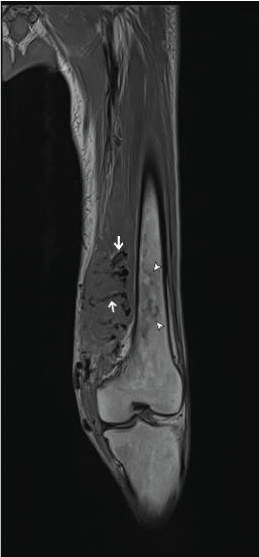 Figure 3A: Coronal T1 (TR:617, TE:13) sequence of the left thigh. Flow voids (arrows) can be seen within the infiltrative isointense mass along the medial aspect of the knee. Note the absence of a marrow replacing lesion the in the adjacent distal femur (white arrowheads). View Figure 3A
Figure 3A: Coronal T1 (TR:617, TE:13) sequence of the left thigh. Flow voids (arrows) can be seen within the infiltrative isointense mass along the medial aspect of the knee. Note the absence of a marrow replacing lesion the in the adjacent distal femur (white arrowheads). View Figure 3A
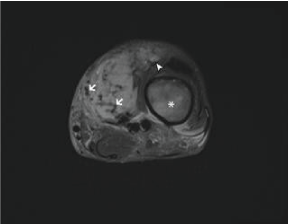 Figure 3B: Axial fat-suppressed T2 (TR: 5830, TE: 74) image shows a heterogenous hyperintense infiltrative mass, again with multiple low signal intensity flow voids throughout the lesion (white arrows). The mass extends into the vastus medialis muscle (white arrowhead) and there is reactive edema within the distal femoral diaphysis (white asterisk). View Figure 3B
Figure 3B: Axial fat-suppressed T2 (TR: 5830, TE: 74) image shows a heterogenous hyperintense infiltrative mass, again with multiple low signal intensity flow voids throughout the lesion (white arrows). The mass extends into the vastus medialis muscle (white arrowhead) and there is reactive edema within the distal femoral diaphysis (white asterisk). View Figure 3B
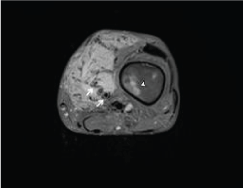 Figure 3C: Axial gadolinium-enhanced T1 fat-suppressed sequence (TR: 726, TE: 11) shows homogeneous enhancement of the mass and dilated feeding and draining vessels (white arrows). There is patchy enhancing reactive marrow edema in the adjacent femoral diaphysis (white arrowhead). View Figure 3C
Figure 3C: Axial gadolinium-enhanced T1 fat-suppressed sequence (TR: 726, TE: 11) shows homogeneous enhancement of the mass and dilated feeding and draining vessels (white arrows). There is patchy enhancing reactive marrow edema in the adjacent femoral diaphysis (white arrowhead). View Figure 3C
Pre-procedure planning ultrasound revealed a highly vascular infiltrative mass extending into the vastus medialis obliquus. Innumerable, dilated, tortuous veins and arteries with connection to the distal femoral vessels were seen throughout the mass (Figure 4a, Figure 4b, and Figure 4c). There was no extension of the lesion into the knee joint. An ultrasound guided fine needle aspiration was performed. Rapid onsite evaluation by cytopathologist revealed rare spindle cells. Core biopsies demonstrated irregular distribution of arteriole and venous-like vessels within a loose myxoid stroma with no atypia, supporting the diagnosis of AVM (Figure 5).
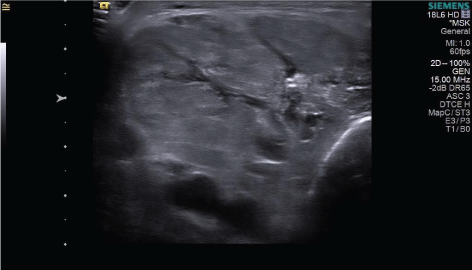 Figure 4A: Transverse B-mode ultrasound of the left medial knee mass for pre-procedure planning shows a heterogeneous, isointense mass with multiple intralesional anechoic vessels. The intervening soft tissue had fatty striation and a fascicular pattern typical of muscle. View Figure 4A
Figure 4A: Transverse B-mode ultrasound of the left medial knee mass for pre-procedure planning shows a heterogeneous, isointense mass with multiple intralesional anechoic vessels. The intervening soft tissue had fatty striation and a fascicular pattern typical of muscle. View Figure 4A
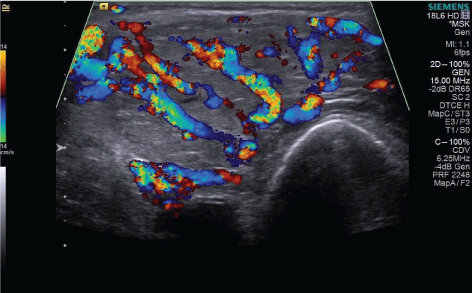 Figure 4B: Transverse color Doppler ultrasound shows dilated high flow arterial and venous vessels throughout the lesion. View Figure 4B
Figure 4B: Transverse color Doppler ultrasound shows dilated high flow arterial and venous vessels throughout the lesion. View Figure 4B
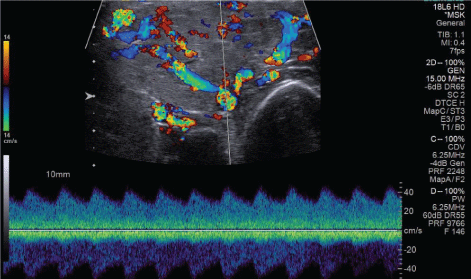 Figure 4C: Spectral Doppler tracing of a high-flow vessel within the mass. Velocities reach as high as 40 cm/s with a low-resistance waveform. View Figure 4C
Figure 4C: Spectral Doppler tracing of a high-flow vessel within the mass. Velocities reach as high as 40 cm/s with a low-resistance waveform. View Figure 4C
 Figure 5: Core biopsies of the lesion stained with hematoxylin and eosin. Irregular distribution of arteriole and venous-like vessels within a loose myxoid stroma support the diagnosis of AVM. View Figure 5
Figure 5: Core biopsies of the lesion stained with hematoxylin and eosin. Irregular distribution of arteriole and venous-like vessels within a loose myxoid stroma support the diagnosis of AVM. View Figure 5
Multiple classification systems of vascular malformations have been proposed in the literature. Mulliken and Glowacki [1] first classified vascular anomalies into hemangiomas and malformations, with the former being present at birth and involving proliferative and involutive phases, and the latter growing as the patient ages without regression. Vascular malformations can be divided into high-flow and low-flow lesions [2,3], which aids not only with etiology, but also with potential treatment.
High-flow vascular malformations include arterial vessels that bypass normal capillaries and drain into tortuous, low-pressure systems, forming AVMs consisting of a tangle of abnormal capillaries and venous structures fed by the arterial system [4-6]. Vascular morphogenesis is believed to take place during the 4th to 10th week of gestation [7]. For congenital AVMs, arrest of normal vascular development occurs due to an inadequate removal of primitive mesenchymal tissue. The remnants of immature tissue remain, leading to haphazard angiogenesis as the patient grows to adulthood [1,3,8]. In cases of acquired AVMs, however, the pathogenesis can vary with the underlying disease state.
The systemic effects of chronic liver disease are well known, though their mechanisms are not fully elucidated. Common sequelae of the disease, such as jaundice, gynecomastia, ascites, cutaneous lesions, and variceal vessel formation can be evident to the examiner. Spider telangiectasias, palmar erythema, and gynecomastia are thought to occur due to an increase in the estradiol to free testosterone ratio [9,10]. Pulmonary manifestations of chronic liver disease tend to be severe. Of the graver lesions, AVMs within the pulmonary vasculature can lead to hepatopulmonary syndrome. This syndrome typically occurs in patients with a higher Model for End-stage Liver Disease (MELD) score and can indicate a poor prognosis [11,12]. One mechanistic theory proposes bacterial translocation leads to localized toxin release in the pulmonary microvasculature, stimulating nitric oxide release and vasodilation, as well as promoting angiogenesis [13-15]. Another theory states that the damaged liver's inability to clear pulmonary vasodilators, in addition to the production of vasodilators released into circulation by the liver, can result in systemic vasodilation [16]. Prolonged exposure to these vasodilatory effects may lead to intrapulmonary AVM formation, as well as hypoxia due to right-to-left shunting [17].
In contradistinction to pulmonary arteriovenous malformations, cases of acquired malformations in the extremities of liver failure patients have rarely been reported. An extensive literature review presents two cases of acquired AVMs in the digits of the upper extremities presented by Alcolado, et al. [18]. These malformations were classified as high-flow lesions after angiography of the hand demonstrated systemic arterial vessels feeding the mass of capillaries with venous drainage. Both cases reported the AVM increased in size as the liver function deteriorated, and after orthotopic liver transplantation, one patient saw complete regression of the lesion.
The etiology of this acquired extremity AVM remains unclear. Prior research has described the role of Ephrin-B2 and EphB4 in delineating arterial and venous boundaries in developing tissue. Their expression has been implicated in mediating angiogenesis and hepatic vascular structure formation during cirrhosis [19-23]. This may be an interesting avenue of future research for extremity vascular malformations.
In the case we present, by patient report, the lower extremity malformation appeared with his diagnosis of liver failure and increased in size over time. He had no recollection of a mass or pain in the left knee prior to his diagnosis of cirrhosis. While a malignancy was the primary concern for the treatment team, MRI findings elucidated the vascular nature of the lesion. Despite the somewhat mass-like appearance on MRI, the infiltrative, ill-defined nature of the lesion and abnormal dilated feeding and draining vessels is typical for a vascular malformation [24,25]. The surrounding soft tissue and adjacent bone marrow edema are uncharacteristic for a primary soft tissue sarcoma or soft tissue metastatic lesion without direct osseous invasion. The MRI findings and subsequent biopsy solidified the diagnosis of AVM. From our review, this appears to be the first reported case of the development of a lower extremity high-flow AVM in a patient with liver cirrhosis described in the literature.
The authors declare that they have no conflicts of interest.
Informed consent was obtained from the patient prior to including any de-identified images in this manuscript.