Intracranial meningeal hemangiopericytomas (HPCs) represent a rare and aggressive intracranial neoplasm located along the dural sinuses, tend to recur and metastasize, and account for less than 1% of intracranial tumours and approximately 2~4% of all meningeal tumors. We discuss herein the dynamic of HPCs and review the literatures from its clinical symptoms, imaging features, histopathological features, differential diagnosis and treatment and prognosis, and present a case of a 35-year-old male with metastatic recurrence of an intracranial hemangiopericytoma, who underwent resection of primary tumor in the right side of the sphenoid ridge 7 years ago.
HPC, Recurrence, Metastasize, Symptom, Feature
Perivascular cell tumor, also known as Hemangiopericytoma, is a soft tissue and mostly single tumor, resulting from the capillary wall peripheral cells. It most commonly occurs in middle-aged people with no gender differences, and mostly locate in the lower extremity, retroperitoneum, and pelvic cavity, and it may also occur in the head, neck, trunk, upper extremity soft tissue, organs and nervous system. The cause of hemangiopericytoma is still not clear so far, which may be related to trauma, long-term use of corticosteroids, pregnancy and hypertension. Generally, the small and superficial tumors locate on the skin and subcutaneous tissue are always with good prognosis, while those large and deep tumors are with poor prognosis. Hemangiopericytomas have the malignant potential, and are easy to recurrence and metastasis to bone, lung and liver.
Intracranial meningeal hemangiopericytomas (HPCs) are rare aggressive tumors with a tendency for local and metastatic recurrence [1]. Approximately 300 cases of HPC have been reported since Stout and Murray described HPC as "vascular tumors arising from Zimmerman's pericytes" in 1942 [2]. After further characterization, the WHO reclassified HPC as a fibroblastic/myofibroblastic tumor [3]. HPC was defined as "a rare disease" by the Office of Rare Diseases (ORD) and the US National Institutes of Health (NIH). It constitutes less than 1% of all intracranial tumors and approximately 2~4% of all meningeal tumors [4]. There was no relevant statistics report in domestic, while in the United States, the number of patients with HPC was fewer than 200,000 in 2014 reported by NIH.
The tumors, in contrast to meningiomas, do not stain with epithelial membrane antigen. They have a grade 2 or 3 biological behavior, and need to be distinguished from benign meningiomas because of their high rate of recurrence (68.2%) and metastases [5,6]. The incidence of HPC in men is slightly more than women (60% male), and the average age of the patients was from 40 to 45-years-old. The tumors are more inclined to occur in the brain (95%) coupled with the pachymeninx, especially sinus sinks nearby, but rare in the meninges.
HPCs usually occur in the same position with meningiomas, which mostly located in the tentorium cerebelli (62%) and minorly located in the tentorium cerebelli and posterior fossa (less than 20%) and the spinal cord (less than 14%). Though multiple HPCs are extremely rare, the simultaneously meningeal hemangiopericytomas are also happened on both the posterior fossa and the thoracic spinal epidural space [7]. The different size and location of HPCs always lead to the various clinical signs and symptoms.
HPCs located in intracranial always tend to oppress the patients' brain tissue with the symptoms of headache, nausea, vomiting, papilledema, paralysis and seizures. As the tumor grows fast and the high intracranial pressure syndrome is often obvious, the patients always present to hospital because of a sudden headache and vomiting. The HPCs located in posterior fossa are often accompanied with the symptoms of unsteady gait and ataxia. Some patients may have subarachnoid hemorrhage or HPC-induced brain hemorrhage as the first symptom [8,9].
Dissimilar with other primary intracranial tumors, the most characteristic clinical manifestation of HPC is the strong tendency of extracranial metastasis. The main extracranial metastasis paths of HPC are blood transfer and intraspinal spread [10], and the most common metastasis position is followed by bone, lung and liver. The lung metastasis patients are often taken with the symptoms of dyspnea, cough and night sweating. The average metastasis time is 84 to 99 months, across the range of 1 to 20 years. The metastasis rates after operation are 13%, 33%, and 64% in 5 years, 10 years, and 15 years respectively [11]. The metastasis can also exist in patients without tumor recurrence after operation [12]. The case of multiple metastases is very common in HPC sufferer, which may release IGF-Ⅱ, resulting in obvious hypoglycemia [13,14].
High recurrence rate is also another feature of HPCs. In a large sample analysis [1], the median recurrence-free period of HPC patients was 50 months (from one month to 26 years). Another statistics showed the recurrence rates after surgery were 65%, 76%, and 87% in 5 years, 10 years, and 15 years respectively, and the multiple recurrence interval times become shorter and shorter. The average time of the second, third, and fourth relapse time are 38, 35 and 17 months respectively [1].
According to the report [15], the median survival for all patients was 83.5 months after date of diagnosis, with 1-year, 5-year, and 10-year survival rates of 100%, 94.4%, and 72.2%, respectively. 41.86% of patients had HPC recurrence. The median time of recurrence was 72.24 months, with 1-year, 5-year, and 10-year progression-free survival rates of 98%, 51%, and 29%, respectively. 11.62% of patients developed extracranial metastasis.
The imaging features of HPCs and meningiomas are very similar and it is difficult to distinguish them. Whether the skull has been thickened or infiltrated can be identified by the skull plain film. The CT scanning of HPCs show the slightly high-density, more clear boundary and lobulated lesions with coherences significantly (Figure 1). The adjacent skull is always accompanied with the osteolysis destruction in HPC caused by their aggressive growth characteristics, while often with appear adjacent skull proliferation in meningioma. The calcification is rarely seen in hemangiopericytoma. Therefore, it can be distinguished with HPCs from meningiomas by the osteolysis or proliferation and calcification of the adjacent skull [16].
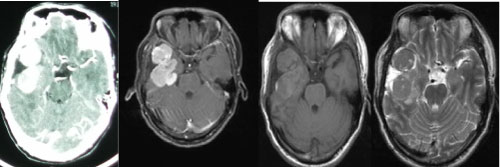 Figure 1: The cranial CT test of HPC. View Figure 1
Figure 1: The cranial CT test of HPC. View Figure 1
On MRI, the tumors are characterized by equal signal intensity on T1-weighted images, increased signal intensity on T2-weighted images, and obviously enhanced but nonuniform on T1-weighted enhanced images.
On the enhanced MRI, the degree of enhancement and the duration of the reinforcement of HPCs are more than meningiomas. The signs of vascular flow void and the necrotic cystic signal also appeared because of the fast grow and necrosis of the tumors [17]. However, the signals of "dural tail", caused by the tumors connected to the dura mater with narrow base, were rarely presented because of the relatively short growth time and the large germinating volume of hemangiopericytomas.
Unlike meningiomas, the DSA (digital subtraction angiography) showed half of the hemangiopericytomas were supplied by the internal carotid artery, and minority hemangiopericytomas took the vein of early emptying phenomenon, with the characteristic features of a venous phase prolongated, spiral vascular structure, resembling AVM. Both DSA and preoperative embolization were recommended to examination for clearing the blood supply and making the definite diagnosis of hemangiopericytomas.
Positron emission tomography (PET) demonstrated a six fold increased uptake of methionine and hyperperfusion in HPCs, whereas glucose utilization was decreased in this area, which is the significant difference from the hyperplasia of affluent meningioma cells and the increased glucose utilization in meningiomas [18]. Recently, the specimens of HPCs and meningiomas were tested by magnetic resonance spectroscopy (MRS), which showed the inositol content in HPCs was significantly higher than meningiomas [19].
Both HPCs and gliomas show a mixed or slightly higher density, irregular enhanced ring lesion in CT scan. The former occur outside the brain, while the latter occur in brain parenchyma with a more obvious peritumoral edema. The position of the tumor, which was used to distinguish HPCs and gliomas, can be identified by the signal changes and multi-directional scanning of MRI.
On the naked eye observation, HPCs are slightly lobulated, spherical, with clear tumor boundary, fish-shaped section, ash red or red color, rich in blood vessels, easy bleeding, and maybe with a thin envelope or pseudocapsule, a necrosis cystic degeneration, and hard or soft texture inequality (Figure 2). HPCs are adhered to endocranium tightly, but with no brain tissue invasion, no disk-shaped growth, and few intratumoral calcification.
 Figure 2: The photo of HPC. View Figure 2
Figure 2: The photo of HPC. View Figure 2
On histology, HPCs are characterized by vascular adventitial cell hyperplasia, extremely rich in blood vessels within the tumor, and dilated sinus-like or "antlers" shape, which may also damage surrounding brain and bone tissue by invading. The tumor is consisted of the consistent polygonal mast cells, which is lack of cytoplasm. The tumor cells are separated into alone or in clusters by rich intratumoral reticular fiber. The dense reticulin stretch into every tumor cells, while the reticulins are surrounded by cells in meningiomas. The tumor is always nuclear atypia and nuclear fission, and often accompany by hemorrhage and necrosis (Figure 3).
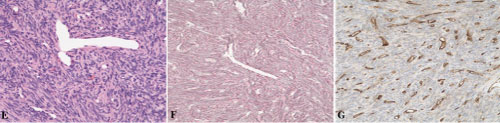 Figure 3: Pathological examination of HPC. View Figure 3
Figure 3: Pathological examination of HPC. View Figure 3
In the terms of ultrastructural, the tumor cells are lack of organelles, and take a plurality of protrusions, significantly silk and micro beam in the cytoplasm. The adventitial cells and the adjacent endothelial cells are separated by the multilayered basement membrane in tumor. The phenomenon of trapped nerve-corpuscle, finger-like tuber of cell membrane, the gap-junctions and the desmosome junctions are undetected in HPCs, which are completely different with meningiomas.
Mena, et al. [20] reviewed 94 cases of HPC, and classified HPC into highly differentiated and anaplastic tumor, according to a number of histological changes in the tumor. The anaplastic tumor was diagnosis on the presence of necrosis or brisk mitotic activity (≥ 5/10 HPF) and two more of the followings: Tumor cells were moderate or more rich, the nucleus variation on moderate to severe, and with hemorrhage symptom. In brief, HPC is different with the atypical or malignant meningiomas on histology. The latter two take the structural performance characteristics of meningiomas, but are often accompanied with various degrees of structural features atypical or undifferentiated, such as structure disappeared, cytoplasm increased, nuclear complex type, mitosis, necrosis and brain invasion.
In immunohistochemistry, it's generally considered that only the mesenchymal marker-vimentin, without the epithelial markers-keratin antigen (CK) and the epithelial membrane antigen (EMA) are expressed in HPC. And both CK and EMA can be found in meningioma. The vessel marker of CD34, CD99 and Bcl 22 are positive in mostly HPC, while CD34 was negative in meningiomas. The different characteristics mentioned above in immunohistochemistry are conducive to diagnose HPCs and meningiomas. The immunohistochemical study showed that vimentin can be expressed both in intracranial and extracranial HPC, while EMA, desmin and S-100 protein cannot. Therefore, both the intracranial and extracranial HPCs are considered to be derived from Zimmerman's vascular skin cells.
In molecular biology, the neurofibromatosis-2 (NF2) gene of 28 cases of intracranial meningeal hemangiopericytomas (iHPCs), 10 case of peripheral hemangiopericytomas (pHPCs), and 26 case of meningiomas were detected, and no NF2 mutations were found in either iHPCs or pHPCs, whereas 35% of meningiomas had NF2 gene alterations (P < 0.001). The research suggested that HPCs are distinct from meningiomas at the molecular genetic level [21].
Because of anaplastic meningiomas (MIIIs) and meningeal hemangiopericytomas (HPCs) display significant morphologic and immunohistochemical overlap, including occasional cases of otherwise classic HPC with focal epithelial membrane antigen (EMA) positivity, ancillary immunohistochemistry (IHC) and fluorescence in situ hybridization (FISH) were used to examine several new biomarkers of MIIIs and HPCs. Three conclusions were as follows [22].
1) EMA, CD99, bcl-2, and claudin-1 IHC and 1p, 14q, NF2, and 4.1B FISH are particularly useful for distinguishing anaplastic meningiomas from meningeal HPCs.
2) Focal EMA expression does not preclude a diagnosis of HPCs.
3) The characteristic FXIIIa staining pattern reported for HPCs also is encountered frequently in anaplastic meningiomas and therefore is nonspecific in this diagnostic setting.
In the differential diagnosis of HPCs, Image test is used to show the exact location, size and proliferation level of tumor, and biopsy is used to identify the cancerization. Generally speaking, blood tests, including complete blood count (CBC) should be taken, and the lower limb bone visible tumor can also be scanned. Other detection, including CT and MRI and so on, also need to be considered in the comprehensive diagnostics.
In clinical practice, the differential diagnosis of HPCs need to be distinguished with the followings tumors first.
1) Hemangioma meningiomas: The imaging of which show homogeneous enhancement, calcification and bone hyperplasia, dural tail caused by the slow growth, significantly different from the imaging features of HPCs. Secondly meningioma tumor cells aggregate into concentrically intimate whirlpool, with psammoma bodies visible. EMA is expressed in meningiomas, while CD99 and Bcl-2 are not. In addition, E-cadherin and catenin's are positive in meningiomas, but negative in HPCs [23]. Comparing with meningiomas, much higher levels of myoinositol in the hemangiopericytomas is certified by MRS [24].
2) Solitary fibrous tumors of the central nervous system (SFTs) [24,25]: SFTs are well-circumscribed, often with attached fragments of dura mater, and have smooth, glistening pseudo capsules. The typical histologic features are spindle cells arranged in a pattern less architecture or in interlacing fascicles, prominent collagenous bands, and branching vascular channels with thin walls. Although CD34 and Bcl-2 can be expressed in both SFTs and HPCs, but the later is weak expression or only partially positive, while the former is diffuse expression. Furthermore, SFT is usually less recurrence, while HPC is relatively easy recurrence and metastasis.
3) Hemangioblastomas: Always occurred in the cerebellum with symptoms of intracranial hypertension and ataxia, and the obvious tendency of familial heredity. It's also the mark of the Vonhippel-Lindau (VHL) syndrome. The tumor boundary was clear and rarely infiltrated the surrounding tissue. The lipid-rich vacuolated mesenchymal cells and capillary network can be found by microscopy, and the NSE and EGFR can be expressed in mesenchymal cells.
4) Mesenchymal chondrosarcoma [26]: The tumors are liable to develop at dura mater cerebralis when it happened at extraskeletal, with the non-specific imaging. When the cartilage components were lack, the histological features of undifferentiated small round cells are similar to those of HPCs, but these small round cells are poorly differentiated, with obvious heteromorphism, and express Leu7 and S-100. So It's not difficult to distinguish them.
The treatment of intracranial hemangiopericytomas is given priority to surgical treatment, and the postoperative radiotherapy should be carried out further. Surgery is the common method to treat this rare tumor, and the recurrence of HPC is associated with the growth index. The low Ki67 index indicates the long recurrence interval, the low tumor metastasis rate, and the long survival time. The aggressive surgical resection, postoperative radiation therapy and extensive follow-up can reduce the risk of tumor in-site recurrence and delayed extracranial metastases [27].
It's believed that the radiation dose of 50~55 Gy is appropriate for small to medium-sized lesions and recurrent lesions [28]. Although postoperative radiotherapy has been widely accepted and affirmed, but the risk of radiation-induced sarcoma is still present (< 1%). Statistics show that 85% to 91% tumors could be recurrent in 15 years after surgery, and 64% to 68% of the tumors could be metastasis. The average survival time is 2 years when the tumor metastasis happened [29].
The far distant metastases of bone, lung, liver, soft tissue, as well as retroperitoneal organs are common, while skin metastases are rare. So no matter how serious the lesions is, the long-term follow-up after surgery is needed.
A 34-year-old man presented to hospital in 2006 with headache, tinnitus, intermittent nausea for one months. Cranial computed tomography (CT) demonstrated the right sphenoid ridge lesions (Figure 4), and underwent surgical excision of the right sphenoid ridge tumor under general anesthesia (Figure 5). Meningioma was diagnosed post surgically. In 2010, a following up computed tomography of the patient revealed a small recurrence of meningeal tumor and then treated with Gamma knife (Figure 6 and Figure 7).
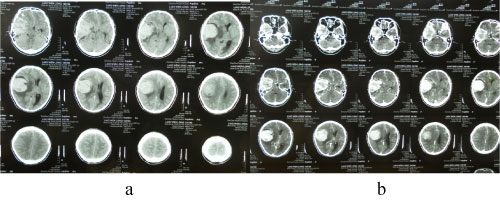 Figure 4: The cranial CT test before surgery (2006). View Figure 4
Figure 4: The cranial CT test before surgery (2006). View Figure 4
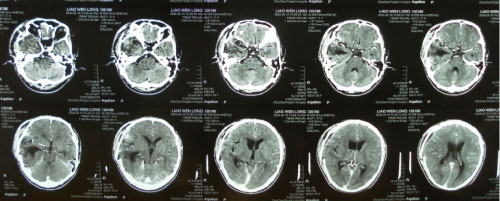 Figure 5: Postoperative cranial enhanced CT scan (2006). View Figure 5
Figure 5: Postoperative cranial enhanced CT scan (2006). View Figure 5
 Figure 6: The cranial CT test before the Gamma knife therapy (2010). View Figure 6
Figure 6: The cranial CT test before the Gamma knife therapy (2010). View Figure 6
 Figure 7: CT after the Gamma knife therapy (2010). View Figure 7
Figure 7: CT after the Gamma knife therapy (2010). View Figure 7
7 years after the right sphenoid ridge meningioma surgery, the patient presented to us again with back pain for 2 months. The magnetic resonance imaging (MRI) and CT (2013) (Figure 8) showed the patient had a surgical scar at the right frontal temporal, a frontal temporal skull defects, and the tenderness and percussion pain on thoracic. The limb muscle strength was level 5, the bilateral limb skin' feeling was normal, the limb muscle tension was not high, and the bilateral Babinski, Brudzinski, and Klinefelter signs were negative. The CT, MRI and CTA examination showed the intracranial meningeal tumor recur again and metasted to vertebra thoracalis and vertebra lumbalis (Figure 9). Three surgery were taken for the recurrence and metastases of the meningeal tumor. Firstly, thoracic tumor resection and thoracic pedicle screw fixation were taken for the thoracic metastasis on general anesthesia (Figure 10) and then the second operation was perform for the resection of the recurrence of meningeal tumor; At last, the lumbar tumor resection and lumbar pedicle screw fixation were taken for the lumbar metastasis on general anesthesia (Figure 11) and the tumor was further diagnosed to be intracranial meningeal hemangiopericytoma.
 Figure 8: The cranial CT test in 2013 (tumor recurred). View Figure 8
Figure 8: The cranial CT test in 2013 (tumor recurred). View Figure 8
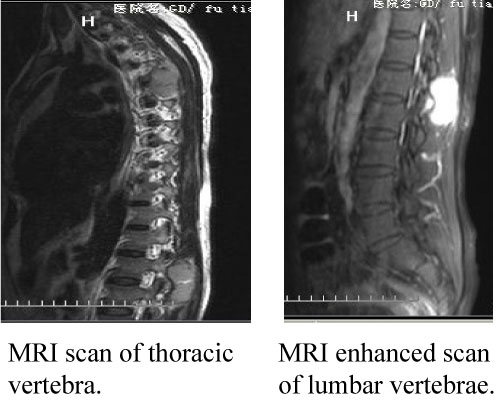 Figure 9: The MRI test of vertebrae. View Figure 9
Figure 9: The MRI test of vertebrae. View Figure 9
 Figure 10: The MRI test of the first surgery. View Figure 10
Figure 10: The MRI test of the first surgery. View Figure 10
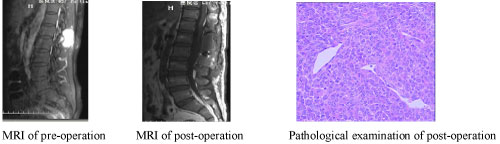 Figure 11: The MRI test and the pathological examination of the third surgery. View Figure 11
Figure 11: The MRI test and the pathological examination of the third surgery. View Figure 11
The case mentioned above not only certified the similarity and the confused clinical features of HPCs and meningiomas, but also suggested the recurrence and the extracranial metastasis of meningeal hemangiopericytomas. Much more histopathological features, clinical features, and imaging features need to be considered in differential diagnosis. The patient was followed up till the present paper was summated, he is normally survival.