The factors influencing the prognosis of children with acute disseminated encephalomyelitis ADEM) are still poorly known and understood, especially for the occurrence of sequelae after the acute episode.
A retrospective cohort of 25 patients with ADEM was obtained (mean age: 5.8 years, mean duration of follow-up: 18 months). 11 of them (44%) presented sequelae. Various clinical, biological, and radiological elements have been taken into consideration to compare the patients with sequelae and those without sequelae.
Statistically significant differences were obtained for the MRI findings. Compared to patients without sequelae, patients with sequelae presented on the first MRI more asymmetric lesions (p = 0.006) and more temporal (p = 0.021), parietal (P = 0.002) and occipital (p = 0.043) lesions. The presence of frontal (p = 0.011) and parietal (p = 0.031) lesions on the second MRI was more common in patients with sequelae. No statistically significant differences were demonstrated for the clinical and biological features.
The magnetic resonance imaging seems to provide elements about the prognosis of children with ADEM, especially the asymmetry of lesions and their location. The assessed clinical and laboratory factors do not seem to influence the occurrence of sequelae. This study is a preliminary work for future studies about the role of imaging in the understanding of the complex physiopathology of ADEM.
ADEM, Children, MRI, Neuroimaging
ADC: Apparent Diffusion Coefficient; ADEM: Acute Disseminated Encephalomyelitis; EEG: Electroencephalography; FLAIR: Fluid Attenuated Inversion Recovery; HUDERF: Hôpital Universitaire des Enfants Reine Fabiola (Children's University Hospital Queen Fabiola); IgIV: Intravenous Immunoglobulin's; MRI: Magnetic Resonance Imagery; MDEM: Multiphasic Demyelinating Encephalomyelitis; MeSH: Medical Subject Headings; NS: Not Significative; LP: Lumbar Punction; MS: Multiple Sclerosis; CNS: Central Nervous System
The acute disseminated encephalomyelitis (ADEM) is an inflammatory demyelinating disease, probably autoimmune, with acute or subacute onset and presenting multifocal damages of the central nervous system [1-3]. In children, ADEM is preceded in 71 to 77% of patients by an infection or a vaccination [4-6]. The incidence of this disease is 0.07 to 0.4/100,000/year for a paediatric population [7-9]. The sex ratio M:F is between 1.2:1 and 1.6:1 [4,8,10]. The diagnosis is based on clinical and radiological features [11]. The survival rate is close to 100% [4-6,10]. Residual deficits persist in 17 to 43% of patients: Motor, sensory, visual, or cognitive disorders, behavioural changes, or seizures [4,6,10]. There is also a risk of relapse or evolution to multiple sclerosis (MS). By definition, ADEM is monophasic [1,11]. But a recurrence can be observed up to 25% of cases. Therefore, we speak of multiphasic disseminated encephalomyelitis or MDEM (Disseminated Encephalomyelitis Multiphasic) [4,6,10,12]. The proportion of patients who will evolve to MS can be up to 33% [4,13,14].
The factors that may influence the risk of developing MDEM or MS after ADEM have been investigated in several studies [4,6,13-15]. But the factors involved in the occurrence of sequelae and residual deficits have not been identified yet. The determination of those factors would allow a better understanding of the complex physiopathology of ADEM and could have implications in the medical care for these patients.
This work's goal is to determine if some clinical, biological, and radiological elements could have a predictive interest in the occurrence of sequelae in children with ADEM.
The articles that served as basis of this work were obtained by an exhaustive research on the Medline database via the PubMed interface, using a combination of the following MeSH terms: "ADEM" or "acute disseminated encephalomyelitis", "paediatric", "MRI" or "magnetic resonance imaging", "diagnosis", "clinical", "laboratory", "neurophysiology", "outcome", "treatment", "prognosis", "disability", "MS" or "multiple sclerosis", "MDEM" or "multiphasic demyelinating encephalomyelitis". The research was limited to articles published between 01 January 2000 and 01 January 2020. Only articles in English were included. The age of the population of interest was limited to 18 years. No limitation has been established for the type of the articles or the gender.
Patients and inclusion criteria: The patients hospitalized in our institution between 01 January 1998 and 31 October 2019 for coma or encephalitis was identified through the database of the Intensive Care, Neurology and Radiology Units.
The inclusion criteria in the study are based on the diagnostic recommendations for ADEM made in 2013 by the International Study Group on Multiple Sclerosis [11]. The following clinical and radiological elements are needed for the diagnosis of ADEM: A first clinical event of the central nervous system, which is polyfocal and presumed of inflammatory demyelinating origin; encephalopathy which cannot be explained by fever, systemic disorder or by a postictal condition; no new clinical or radiological element occurs more than 3 months after the onset of the disease; brain MRI (magnetic resonance imaging) is abnormal during the acute phase that can last up to three months, with diffuse and poorly demarcated lesions in the white matter and eventually in basal ganglia and thalami.
Among patients meeting these criteria, those who developed later MS or MDEM were excluded.
Data collection: In the patient files, the following data were collected: Sex, date of birth, date of onset, date of admission, duration of hospitalization, duration of follow-up and history (particularly infections and vaccinations).
Particular attention was paid to the following clinical features: Presence of motor or sensory disorders, cerebellar symptoms, cranial nerves deficits, visual disorders, oculomotor disturbances, speech impairments, convulsions, behavioural disorders, impairment of consciousness and coma.
About the treatment, the following elements were collected: The time between the onset of the symptoms and the start of the corticotherapy, the administered dose of corticosteroid, the duration of the corticotherapy and the administration of intravenous immunoglobulin (IVIg).
For each patient, the complementary investigations were reviewed in search of: An inflammatory syndrome on the first blood sample (C-reactive protein greater than 1 mg/dl, leukocytosis greater than 11,000/microl), abnormities in the CSF (red blood cell count greater than 5/microl, white blood cell count greater than 5/microl, protein level greater than 0.45 g/l) and an abnormal electroencephalogram (EEG).
The MRI exams were reviewed. The following elements have been investigated: The relative symmetry in terms of location, size and intensity of the lesions, the bilateral aspect of the affection, the damages in the cerebellum, the cerebellar peduncles, the cerebral peduncles, the brain stem, the basal ganglia, the thalami, the internal capsule, the corpus callosum, the subcortical, periventricular and deep white matter, the spinal cord, the optic nerves, the presence of a restricted diffusion (hyperintensity in diffusion-weighted imaging and hypo intensity in ADC - Apparent Diffusion Coefficient), an enhancement after intravenous gadolinium injection and an hypointense aspect on T1 sequence. The number of lesions was also collected according to the following considerations: The lesion had to have a minimal diameter of 3 mm in the axial, frontal or sagittal plane and the lesions were considered as distinct when they were separated by at least 1 mm of healthy appearing tissue [16].
Prognosis and follow-up: The presence of sequelae after the acute phase has been evaluated on base of the reports of the follow-up consultations conducted by the referent neuropediatrician. The presence of sequelae was determined when the patient had persistent residual deficits after the acute phase of the disease, which can last up to three months. All types of sequelae were considered.
Study design, data treatment and statistical analysis: This is a retrospective observational case-control study. Its aim is to highlight a possible relationship between the appearance of sequelae and the parameters mentioned above.
For the continuous variables, the Shapiro-Wilk test was used to verify the distribution normality. The relationship between the prognosis and the normally distributed variables were evaluated using the Student's t-test for two independent samples. For non-normally distributed variables, the Wilcoxon signed-rank test was used.
The relationship between the prognosis and the discrete variables was assessed using the Pearson's chi-squared test. For the variables having a statistical significance under p = 0.1, a multivariable logistic regression model was realised, and the least significant variables were gradually removed to increase the level of significance of the test.
A difference was considered as statistically significant when the p-value was under 0.05. The statistical analysis was performed using Statistics 9.0 and SSPS Statistics 20.
Among the 139 patients admitted for coma or encephalitis, 29 (21%) met the diagnostic criteria for ADEM. Among these, three (10%) later developed MS and a patient (3%) developed a MDEM. The resulting cohort consisted of 25 patients (Figure 1).
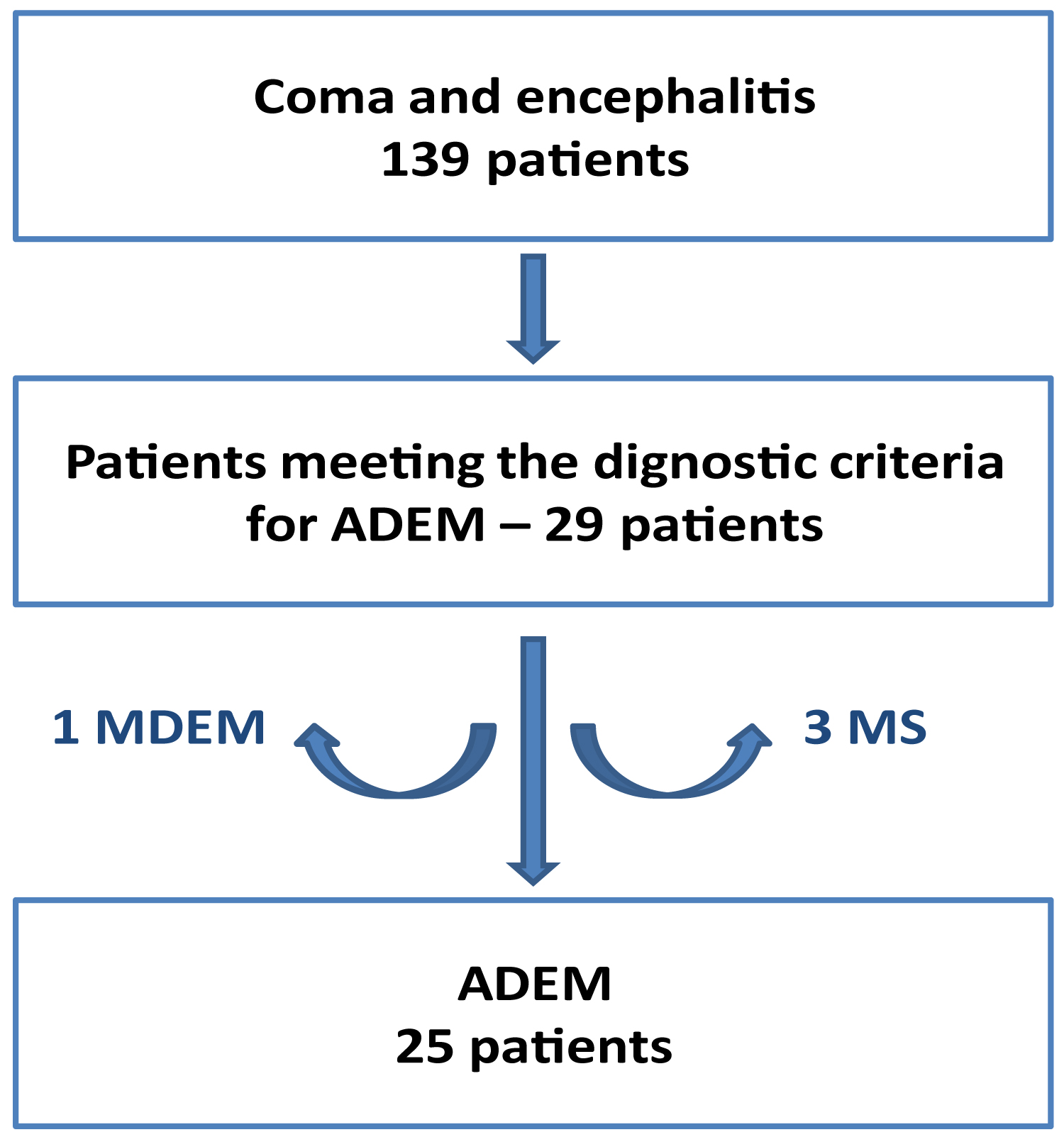 Figure 1: Patient selection.
View Figure 1
Figure 1: Patient selection.
View Figure 1
Among the 25 children, 11 (44%) presented sequelae: 7 patients (28%) developed learning disorders and difficulties at school, 3 patients (12%) developed motor disorders, 2 patients (8%) developed behavioral disorders and 2 patients (8%) developed epilepsy (Table 1). 2 patients (8%) have been admitted in specialized institutions because of the severity of the deficits (motor disorders for 2 patients).
Table 1: Sequelae presented after ADEM. View Table 1
The average follow-up duration was 1 year and a half, with a minimum of 2 months and a maximum of 7 years. Patients who presented sequelae benefited an average follow-up duration of 3 years against 4 months for patients who did not have sequelae.
The average age of the patients at the onset of the disease was 5.8 years. The youngest patient was 8-months-old and the oldest was 13 years. 68% of the patients were younger than 6 years (Figure 2 and Table 2).
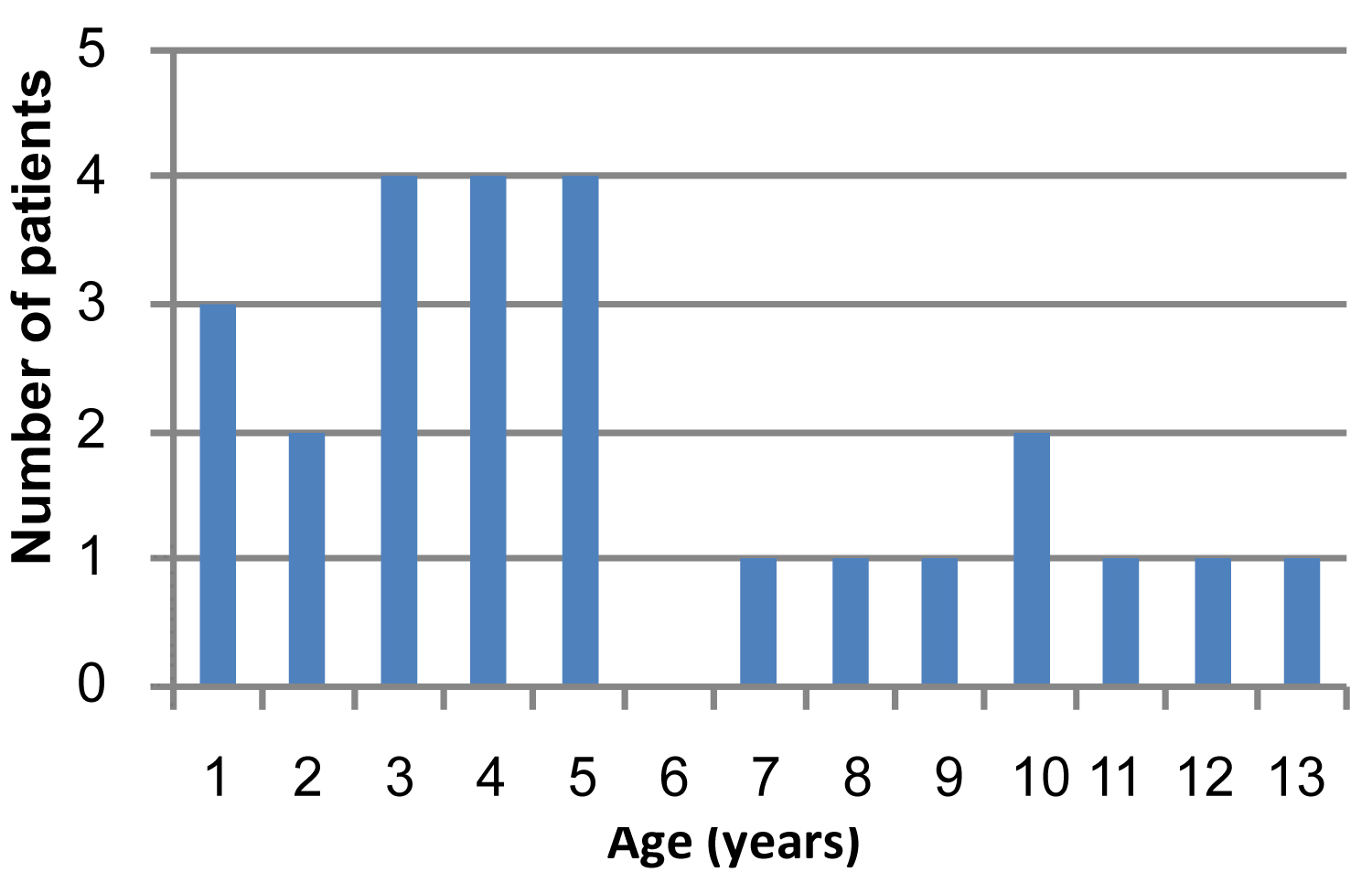 Figure 2: Distribution of the patients by age.
View Figure 2
Figure 2: Distribution of the patients by age.
View Figure 2
Table 2: Epidemiological data, clinical presentation, treatments, and complementary exams - number of patients (%). View Table 2
The cohort was constituted by 8 (32%) female and 17 (68%) male patients, with a sex ratio M:F of 2.1:1 (Table 2).
The number of cases was higher during the spring (10 of 25 patients, or 40%) and the autumn (7 out of 25 patients, 28%) (Figure 3).
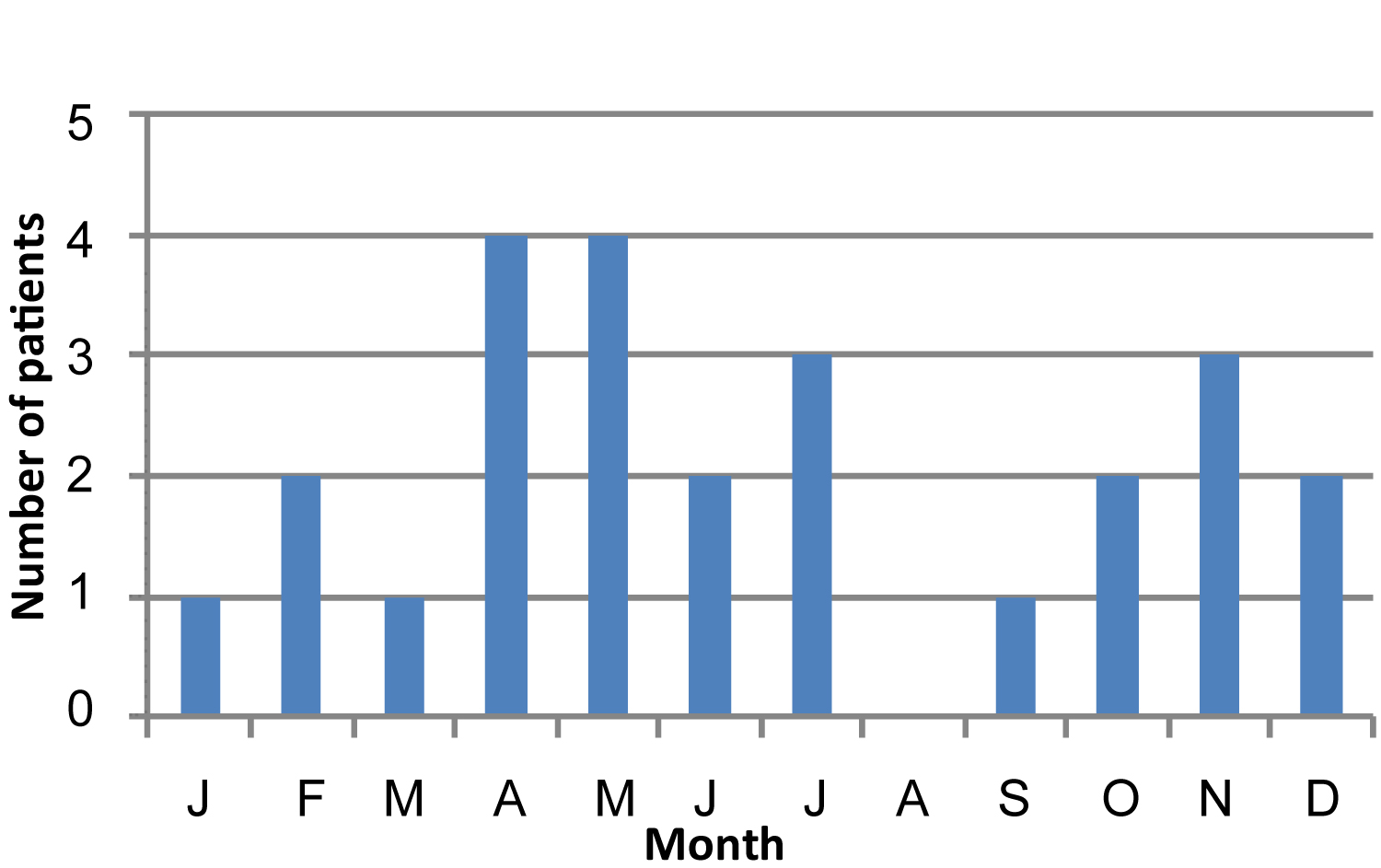 Figure 3: Monthly distribution.
View Figure 3
Figure 3: Monthly distribution.
View Figure 3
6 patients (24%) had an infectious episode in the six weeks preceding the onset: 3 patients (12%) had chickenpox, 2 patients (8%) had an otitis and one patient (4%) presented angina (Table 2). The 3 patients who had chickenpox, all developed sequelae later. No patient had recently received a vaccine before the onset of the disease.
Between the group that developed complications and the one that did not, there is no statistically significant difference for age, sex, the monthly distribution, or the infectious history.
Among the 25 patients, 19 (76%) were polysymptomatic (had more than one symptom), 14 (56%) had an impairment of consciousness and 6 (24%) developed a coma. Language disturbances were present in 10 patients (40%). 9 patients (36%) had seizures. Motor disorders were demonstrated in 8 patients (32%). 4 patients (16%) had oculomotor disturbances: 2 patients (8%) developed a strabismus and 2 patients (8%) developed a nystagmus.
Intravenous corticosteroids were administered to 17 patients (68%). The mean dose was 29.4 mg/kg/day. The average time between the onset of symptoms and the start of the corticotherapy was 5.3 days. The mean duration of the corticosteroid therapy was 4.7 days. 8 patients (32%) received IVIG.
For all these parameters, the comparison between the group with deficits and the one without has not demonstrated statistically significant difference. These data are summarized in Table 2.
An inflammatory syndrome was present on the initial blood sample in 9 patients (36%).
A lumbar puncture was performed in 24 patients (missing data for 1 patient) and the CSF was abnormal in 11 (46%): 10 patients (42%) had an increased white blood cell count, 7 patients (29% ) had an increased protein level in the CSF and 2 patients (8%) showed an increased red blood cell count. No infectious agent has been demonstrated.
An EEG was performed in 20 patients and presented abnormalities in 17 (85%) of them: 11 patients (55%) had a slow diffuse dysrhythmia, 5 patients (25%) had signs of diffuse brain damage, and 1 patient (5%) showed an excess of theta waves. 3 patients (15%) also showed epileptiform discharges.
For these exams, no statistically significant difference was found between the group with sequelae and the one without.
These data are resumed in Table 2.
The images of the first MRI exams have been obtained for 23 patients (92%), those of the second MRI for 21 patients (84%) and those of the third MRI for 13 patients (52%). The average number of MRI exams per patient was 2.5. The radiological file has not been obtained for two patients. On 57 exams, the diffusion weighted sequence was performed for 55 (96.5%) of them.
An intravenous injection of gadolinium was performed for 44 exams (77%). MRI examples are illustrated in Figure 4.
 Figure 4: Radiological presentation of ADEM. (A) Brain MRI of a 3-year-old male patient. FLAIR sequence in coronal plane. Bilateral lesions, symmetrical and poorly demarcated, of the basal ganglia and the subcortical white matter. (B) Brain MRI of a 5-year-old female patient. FLAIR sequence in coronal plane. Bilateral asymmetrical lesions, poorly demarcated, of the profound and subcortical white matter. (C) Brain MRI of a 2-year-old male patient. FLAIR sequence in coronal plane. Atypical presentation with lesions involving only the basal ganglia.
View Figure 4
Figure 4: Radiological presentation of ADEM. (A) Brain MRI of a 3-year-old male patient. FLAIR sequence in coronal plane. Bilateral lesions, symmetrical and poorly demarcated, of the basal ganglia and the subcortical white matter. (B) Brain MRI of a 5-year-old female patient. FLAIR sequence in coronal plane. Bilateral asymmetrical lesions, poorly demarcated, of the profound and subcortical white matter. (C) Brain MRI of a 2-year-old male patient. FLAIR sequence in coronal plane. Atypical presentation with lesions involving only the basal ganglia.
View Figure 4
First MRI: The lesions were bilateral in 21 patients (91%) and relatively asymmetrical in 12 patients (52%). The average number of lesions identified on each exam was 8.4.
Lesions were present in the subcortical white matter in 13 patients (57%) and in the periventricular and deep white matter in 7 (30%). Cortical lesions were observed in 10 patients (43%). The lesions of the cortex and the white matter were localized in the frontal lobe in 11 patients (48%), in the temporal lobe in 9 patients (39%), in the parietal lobe in 9 patients (39%), in the occipital lobe in 6 patients (26%) and in the insular lobe in 6 patients (26%).
Lesions of the thalami were described in 11 patients (48%). 13 patients (57%) had lesions of the basal ganglia: 13 patients (57%) in one or both lenticular nuclei and 9 patients (39%) in one or both caudate nuclei.
Lesions in the corpus callosum were present in 3 patients (13%).
6 patients (26%) had lesions localized in the brainstem, 5 patients in the cerebellum (22%) and 2 patients (9%) presented lesions of the spinal cord.
The optic nerve has not been investigated in an optimal way because the appropriate sequences and slices were not performed. Taking these limitations into account, no patient presented lesions at this level.
A restricted diffusion was observed in 4 patients (17%). 8 patients (35%) presented hyposignal lesions in the T1 sequences.
An intravenous injection of gadolinium was performed for the 1st MRI in 21 patients (91%). Enhancement was objectified in 4 of them (19%).
Statistically significant differences have been demonstrated. The patients with sequelae had more asymmetric lesions (p = 0.006) and more lesions localized in the temporal (p = 0.021), parietal (p = 0.002) and occipital (p = 0.043) lobes.
These results are summarized in Table 3.
Table 3: First MRI - number of patients (%). View Table 3
Second and third MRI: Statistically significant differences between the two groups have also been demonstrated. Concerning the second MRI performed, lesions localized in the frontal and parietal lobe were observed more frequently in patients with sequelae (respectively p = 0.011 and p = 0.031).
The other parameters measured at the second MRI as well as those at the third showed no statistically significant differences between the 2 groups.
The obtained results are summarized in Tables 4 and Table 5.
Table 4: 2nd MRI - number of patients (%). View Table 4
Table 5: 3rd MRI - number of patients (%). View Table 5
This work allows us to highlight that some radiologic elements seem to have an interest in the prognosis of those children. The patients who presented on the first brain MRI asymmetric damages or lesions localized in the temporal, parietal or occipital lobe seem to have a higher risk of developing sequelae or residual deficits. The presence of frontal or parietal lesions on the second MRI also seems to have a pejorative effect on prognosis.
Clinical presentation, blood sample, CSF and EEG abnormalities do not appear to influence the occurrence of sequelae. Treatment with corticosteroids or immunoglobulins does not appear to influence the occurrence of residual deficits. However, only a randomized controlled prospective trial could really evaluate the effectiveness of these treatments, which has so far not been realized yet.
Our study demonstrated a sex ratio M:F of 2.1:1. There is no statistically significant gender difference regarding the occurrence of sequelae. But it is interesting to note that 63% of female patients evolved without sequelae, against 53% for male patients. Sex could thus also play a role in the prognosis of ADEM, as in the prognosis of other inflammatory pathologies [17,18].
The rate of infection and vaccination prior to the onset of the disease was relatively low compared to the numbers reported in the literature [4-6]. This can be explained by the fact that such antecedents were not systematically sought. Moreover, the parents do not always report a former infection, especially if it was relatively mild.
The nature of the infectious agent could also possibly affect the occurrence of sequelae, especially chickenpox [19,20]. The 3 patients who had chickenpox, all developed sequelae later. However, the limited number of patients does not allow us to draw any meaningful conclusions.
The rate of sequelae in our cohort was 44%, which seems to be particularly high compared to the results obtained by some authors [6,10]. This result from the lack of homogeneity in the definition of the sequelae and in the types of residual deficits considered. Murthy had shown a sequelae rate of 17% after an ADEM but did not consider the cognitive and behavioural deficits [10]. Dale, by considering it, highlighted a sequelae rate of 43%, comparable to our results [4].
We have studied the occurrence of sequelae in children with ADEM, but it would also be interesting to study the factors influencing the severity of these effects. 2 patients (8%) were admitted to specialized institutions because they had extremely severe consequences. The limited number of patients has limited us in a deeper study of this subject.
Our study has the advantage of using homogeneous, clear, and reliable inclusion criteria, based on the diagnostic recommendations for ADEM made by the International Study Group on Multiple Sclerosis [11]. We retained all types of sequelae. We included in this work many clinical, biological, and radiological data to assess the relationship between them. Our study also considered the evolution of the lesions on MRI and was not limited to the first MRI performed. Concerning the follow-up, a minimal follow-up duration of three months was required to ensure that the residual deficits persist after the acute phase of the disease [11]. For patients who did not have complications, such a long follow-up was not always necessary and shorter follow-up periods have been tolerated.
The limited number of patients, although comparable to the other studies on the subject, inevitably reduces the quality of the study, preventing among other things the realisation of a multivariable model. The other weaknesses of this study are related to the disadvantages faced by any retrospective study, among other things a difficult control of all the factors and of the matching. There is also a bias since the most severely affected patients have benefited of more additional tests, and MRI.
Our work has shown that asymmetric damages and lesions preferentially localized in the temporal, parietal and occipital lobes on the 1st MRI seem to provide elements concerning the appearance of sequelae in children with ADEM. It also showed that the presence of lesions in frontal and parietal lobe on the 2nd MRI may also play a role. The evaluated clinical and biological elements do not seem to have a prognostic interest.
This study is a preliminary work for future studies about the role of imaging in the understanding of the complex physiopathology of ADEM.
• After ADEM, an important part of the cohort presented sequelae.
• Clinical and biological features do not have predictive interest.
• Some imaging features seem to influence the prognosis.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.