The neoplasms of the Parpharyngeal Space (PPS) are rare 0.5%-1% of all head and neck tumors [1]. The PPS is an inverted cone-shaped region that extends from the skull base to the hyoid bone. A complex structure called the tensor-vascular-styloid fascia, divides the PPS into the prestyloid and poststyloid compartments. The PPS can be involved by four different types of neoplastic lesions: Primary tumors (benign or malignant), metastatic lymph nodes, lymph node involvement by lymphoproliferative diseases, and tumors arising from adjacent sites that secondarily extend into the PPS [2-4].
Some 50% of the neoplasms arise from the deep lobe of the parotid gland or minor salivary gland. These lesions are typically benign pleomorphic adenoma and involve the prestyloid compartment of the PPS. In the retrostyloid compartment, the most common tumours are neurogenic (schwannoma, paraganglioma, neurofibroma [5].
The goal of this paper is to report two cases of salivary gland tumours in PPS and the importance of imaging studies in the evaluation and differential diagnosis of tumors that involve it.
A 69-year-old man sought medical attention for a swelling of the left tonsil that had been present for 6 months. His history was negative for recent head and neck infections. On hematologic tests were within normal limits. On physical examination, a mild displacement of the left tonsillar fossa and was evident; the overlying mucosa was apparently normal. The rest of the physical examination was unremarkable. To evaluate the lesion that clinically appeared to take origin from the left PPS, CT imaging was realized (Figure 1).
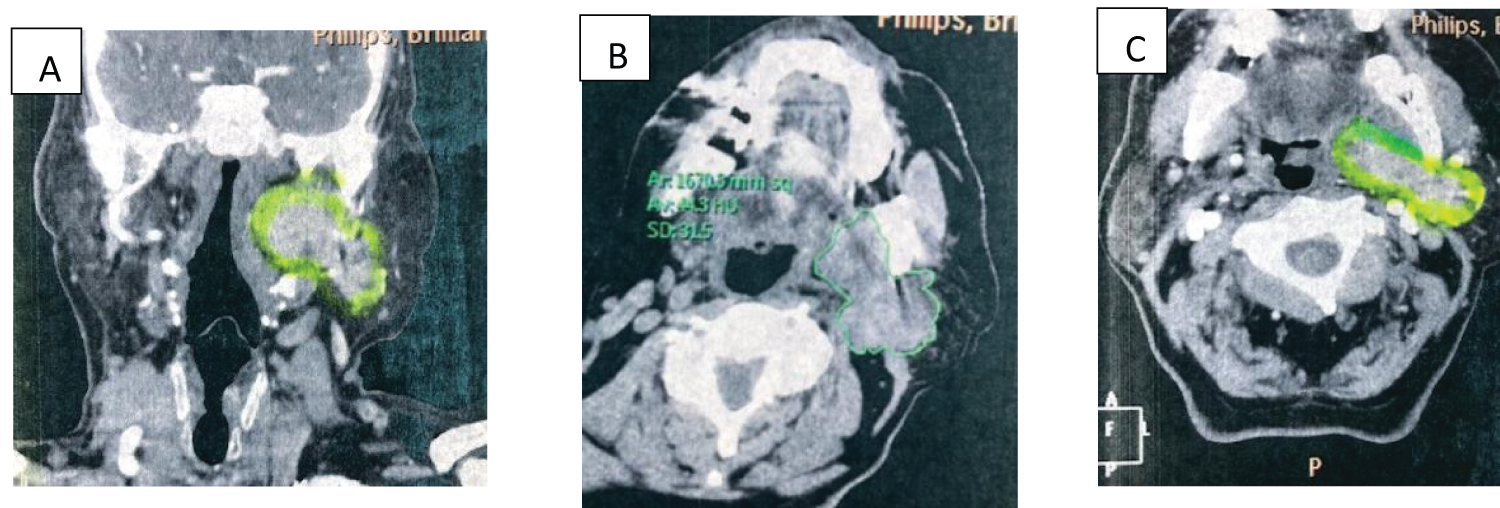 Figure 1: (a) Coronal reconstruction of a contrast CT showing a left, well defines, enhancement in an intraparotid lobulated mass; (b & c) Axial simple and contrast CT image of neck showing an intraparotid mass.
View Figure 1
Figure 1: (a) Coronal reconstruction of a contrast CT showing a left, well defines, enhancement in an intraparotid lobulated mass; (b & c) Axial simple and contrast CT image of neck showing an intraparotid mass.
View Figure 1
An ultrasonography-guided fine needle aspiration cytology (FNAC) allowed diagnosis: Benign, Warthin tumor. The patient underwent excision of the parapharyngeal mass through a transcervical approach. At definitive histologic study Warthin tumor. The postoperative course was uneventful, and the patient was discharged from the hospital 3 days after surgery.
A 69-year-old man was referred to head and neck surgeon with a gradually increasing, painless swelling in the rigth tonsillar area of 6 months duration. There was clinical evidence of dizziness and was diagnosed as Parkinson disease as relevant medical history. Physical examination showed a 6 cm smooth swelling in the right tonsillar area extending superiorly to the nasopharynx and inferiorly to the base of tongue.
There was no associated dysphagia or dysphonia. The swelling was of varying consistency and was not tender on palpation. A MRI imagine was realized (Figure 2).
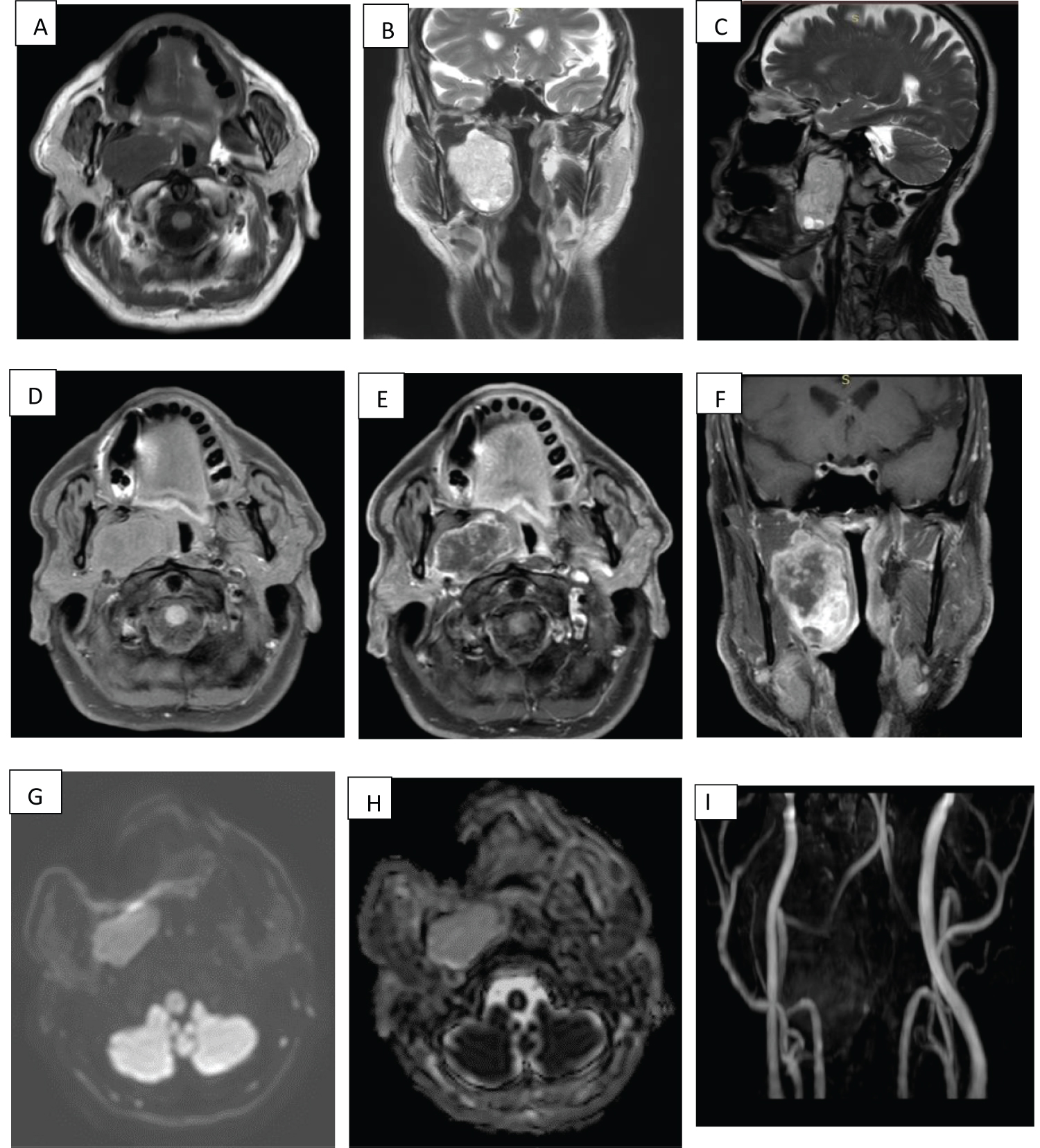 Figure 2: MRI- (a) Axial image on TSE-weighted T1 sequence shows a hypointense, homogeneous, oval lesion with well-defined borders located in the right PS which decreases the caliber of the lumen of the oropharynx and hypopharynx; (b and c) Coronal image on TSE-weighted T2 sequence show a hyperintense mass with some cystic areas; (d, e and f) Precontrast SPIR T1-weighted axial image and postcontrast axial and coronal images showing heterogeneous enhancement of the lesion; (g and h) DWI images and ADC map showing restriction of the lesion; (i) V3DI MC 3D showing that the cervical vessels are intact.
View Figure 2
Figure 2: MRI- (a) Axial image on TSE-weighted T1 sequence shows a hypointense, homogeneous, oval lesion with well-defined borders located in the right PS which decreases the caliber of the lumen of the oropharynx and hypopharynx; (b and c) Coronal image on TSE-weighted T2 sequence show a hyperintense mass with some cystic areas; (d, e and f) Precontrast SPIR T1-weighted axial image and postcontrast axial and coronal images showing heterogeneous enhancement of the lesion; (g and h) DWI images and ADC map showing restriction of the lesion; (i) V3DI MC 3D showing that the cervical vessels are intact.
View Figure 2
The patient had an excision of the parapharyngeal mass through a transcervical approach without complication. Histological examination of the section from the parapharyngeal mass 4.5 × 6.3 cms showed Pleomorphic adenoma (Figure 3).
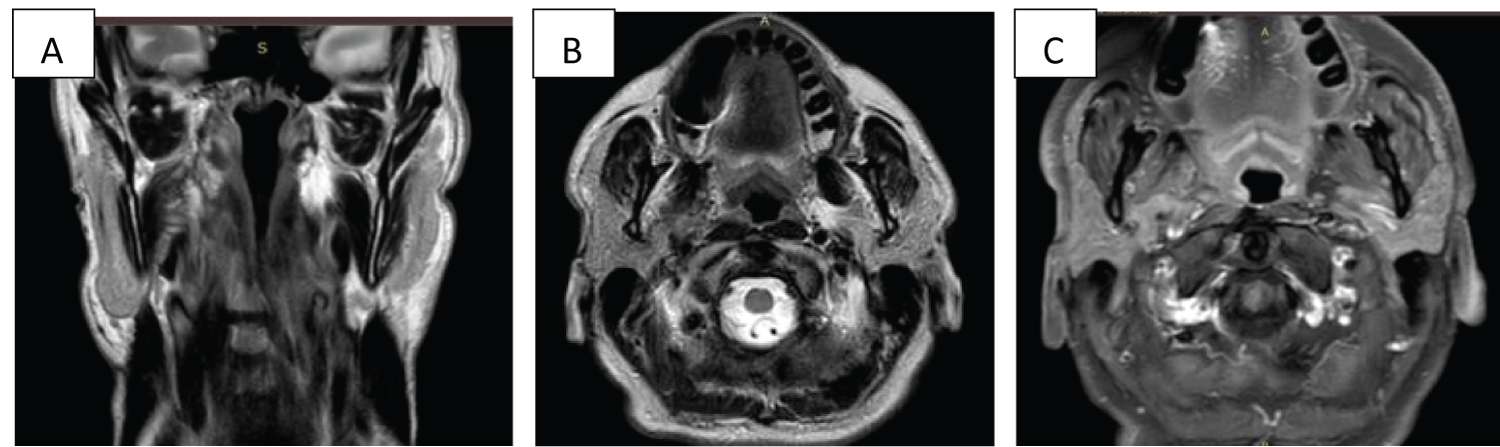 Figure 3: Postoperative study: Coronal (a) and axial (b) images on T2-weighted pulse sequence and axial T1 SPIR image with gadolinium (c) showing postoperative changes in the right PS, without evidence of residual tumor 3 months after surgery.
View Figure 3
Figure 3: Postoperative study: Coronal (a) and axial (b) images on T2-weighted pulse sequence and axial T1 SPIR image with gadolinium (c) showing postoperative changes in the right PS, without evidence of residual tumor 3 months after surgery.
View Figure 3
Of all head and neck masses, lesion of the PPS is rare. The literature on this topic is limited to single-center, case reports or case series [6]. As we had presented our series of cases [7] now this report of two cases of great tumors in PPS.
Neoplasms of the PPS pose a challenge to clinicians with regard to both preoperative diagnosis and treatment. For almost all the lesions, diagnosis is achieved only by biopsy, which may be hazardous in this area [8].
Fine needle aspiration cytology (FNAC) can help clinicians to be reasonably sure about the nature of the lesion and avoids an open biopsy. FNAC is a simple, speedy, relatively painless and cost-effective procedure that can be used for the diagnosis of a tumour anywhere in the body that is accessible by a needle [9].
It is of utmost importance in the diagnosis of a metastasis from an unknown primary site as it gives a clue to where the primary is and narrows down the number of sites to be investigated. The PPS can be involved by metastatic lymph nodes, lymph node involvement by lympho proliferative diseases.
Sonography is useful tool if needed for guidance of submandibular or superficial lobe parotid gland biopsy [10].
FNAC is recommended by multiple authors for the diagnosis of parapharyngeal tumors with accuracy 100%. Even cystic lesions arising in the poststyloid compartment of the PPS [11].
However, results with FNAC are inconsistent among different centers as reported Luna [12].
In ours first case the FNAC was great utility guide by ultrasound and had correlation with histologic biospy.
The imaging studies are essential to establish the diagnostic approach and for pre-surgical planning, the correct characterization determines preoperative treatment planning and is important in optimizing the individualized surgical plan.
The findings on a computed tomography of a Warthin tumor like our first case are: Classic appearance is a well defined heterogeneous solid cystic lesion within the superficial lobe of parotid, parotid tail or deep lobe. Well defined lesion without calcification. Cystic changes appear as intralesional lower attenuation or moderate enhancement and presence of a mural nodule is strongly suggestive or Warthin tumor [13].
They may be bilateral or multifocal in up to 20% of cases and are the most common neoplastic cause of multiple solid parotid masses. In the first case, this characteristic was not detected in the biological behavior of Warthin's tumor.
An entity that should be considered in the differential diagnosis is intraparotide nodular lesions of metastatic origin, which could have cystic changes similar to those of Warthin's tumor [14].
When evaluating a very bulky mass in the head and neck, the no Hodgkin Lymphoma of nodal and extranodal origen should also be consider, as reported in our research. The no Hodgkin Lymphoma could compromise the PPS [15].
Magnetic resonance imaging (MRI) is a modality of choice for evaluation and differentiation between benign and malignant lesions, on account of it better definition of soft tissue, and provides precise information concerning tumor margins as well as the relationship with the surrounding structures. Very useful in case two. The imaging of salivary gland tumors is a major challenge for radiologists due to the great variety of differential diagnoses, their appearance usually depend on tumours size [16,17].
For its evaluation by resonance some signs have been established as algorithm for the MRI diagnosis of Pleomorphic adenoma as in the study by Zagui, et al. where was developed on the basis of five “high probability” criteria that all must be fulfilled for the MRI to qualify as a positive test result: Bright T2-signal, sharp margins, heterogeneous nodular enhancement, lobulated contours, T2-dark rim, found that of 103 parotidectomy, 41 cases corresponded to pleomorphic adenoma eighteen of 21 cases that met all five “high probability” MRI criteria were consistent with PA on final histopathology; they concluded what a “high probability” MRI is about 95% specific for pleomorphic adenoma as our case [18].
The Mucoepidermoid carcinoma is other tumor to consider, the MRI findings seem to have a tendency to be related to the histological grade. Low to intermediate signal intensities on T2 weighted images reflect high-cellularity, and the ill-defined margin reflects the invasive growth of tumour cells. MRI features of intermediate-grade Mucoepidermoid Carcinoma show a combination of those of low- and high-grade MECs Among low-grade Mucoepidermoid Carcinoma tumors usually had a hyperintense area on T2 weighted images, reflecting a cystic architectural pattern because of the existence of abundant mucin-secreting cells [19].
The cervical paragangliomas are highly vascularized neoplasms. The carotid paraganglioma in the carotid bifurcation, the most frequent, It has been reported that Carotid Body Tumors can cause splaying of the carotid bifurcation, contrary to vagal paragangliomas, which cause displacement of the internal carotid artery (ICA) anteriorly with no increase of the distance between the internal and external arteries, which can be appreciated at MRI [20] obtaining angiographic images are very important to distinguish these entities. In MRI show low signal on T1 sequences and high signal on T2 sequences, enhancement after homogeneous and intense contrast. The 'salt and pepper' pattern representing flow voids is characteristic, but not diagnostic. In vessels and areas of hypersignal due to slow flow or hemorrhage, it is more common in paragangliomas large (> 1 cm). Angiography shows hypervascular mass with large afferent vessels and early venous drainage. The ascending pharyngeal artery is the “paraganglioma artery”, since its branches can supply tympanic, jugular, vagal and carotid paragangliomas [21]. The Figure 2 obtained by MRI allowed us to rule out this diagnostic possibility in our patient.
The Schwannomas have been called solitary nerve sheath tumors and perineural fibroblast tumors, may arise in the PPS [22]. Schwannomas grow longitudinally along the length of the nerve assuming a fusiform shape. Imaging findings have been described to differentiate the pattern of vessel displacement observed in vagal Schwannomas vs. those originating in the sympathetic chain [23,24]. In MRI show a circumscribed lobulated ovoid heterogeneously enhancing soft tissue mass lesion in the PPS, heterogeneously, hyperintense on STIR sequences, hypointense on T1 and reveals thin T2 hypointense capsule. Changes cystic degeneration and hemorrhage can also be observed. The Figure 2B and Figure 2C by RMI although show a hyperintense pattern, it lacks the much delimited fusiform shape, with displacement of the vessels, typical in Schwannomas allowed us to rule out this differential diagnosis in our case.
Tumors of PPS are very rare, the Pleomorphic adenomas of the deep lobe of the parotid being the most frequent. Imaging studies such as ultrasound, axial tomography and MRI are very useful in planning therapy for each patient. The multidisciplinary management of these cases allows the correct diagnostic and therapeutic approach as in our cases, which are not different from those reported in the international literature.