Alterations in immune reconstitution have been implicated with the development of chronic graft-versus-host disease (cGVHD) in allogeneic bone marrow transplantation (BMT) recipients. Since T cells are important mediators in this disease, the aim of this study was to search for a biologic marker for cGVHD moderate and severe forms. T cell subpopulations were studied in cGVHD patients according to the proposal of the NIH consensus criteria at diagnosis, thus, in the absence of immune suppression. We present a one year follow up in a cohort of 42 BMT patients in a prospective analysis of peripheral blood and tissue biopsies collected for diagnostic purposes.
Clinical characteristics of patients with (n = 19) or without (n = 23) cGVHD were not statistically different, as well as reconstitution kinetics in peripheral blood of CD4+, CD8+ and regulatory T cells in both groups. However, immunohistochemistry from four different organs/tissues analyzed at cGVHD onset showed similar characteristics. There was a predominance of CD8 T cell infiltration in close contact with lesion areas, with poor and distant CD4 T cell infiltration and a paucity or absence of Tregs. Our data suggest the pattern of T cell infiltration as a diagnostic marker for cGVHD.
cGVHD diagnosis, Prospective study, CD4 T cell, CD8 T, Treg, Immunohistochemistry
Chronic graft-versus-host disease (cGVHD) is a frequent complication of allogeneic hematopoietic stem cell transplantation (HSCT) with high morbidity and mortality rates. The prevalence varies from 25%-80% in long term survivors [1,2]. The disease resembles auto-immune disorders such as Sjogren's syndrome, scleroderma and systemic lupus erythemathosus [3]. However, the pleomorphic nature of cGVHD as well as the paucity of animal models has brought difficulties on the understanding of the mechanisms underlying physiopathology and clinical strategy management [4] such that little substantive progress has been made over the last decades.
Several retrospective and large prospective studies have validated many aspects of the 2005 NIH consensus and staging criteria including organ score, overall severity, and categories of GVHD. In 2014 the cGVHD Diagnostic and Staging Task Force of the NIH Consensus updated the diagnostic and staging criteria for cGVHD [5-8] improving the disease classification and thus facilitating information exchange among health professionals.
Although several attempts at standardization in clinical and histopathological diagnosis have been published in the literature, little is known about the pathophysiological mechanisms that lead to such commonly encountered events [9].
Several immune cell populations have been shown to be altered in cGVHD. Besides alterations in T and B cell responses, abnormalities in T regulatory cell (Treg) number and function had been reported [10]. The latter is a functionally distinct subset of mature T cells with broad suppressive activity, which play key role in the maintenance of peripheral tolerance [11].
In the setting of experimental allogeneic HSCT, Tregs have been shown to play a role in the establishment of tolerance between recipient tissues/organs and donor derived immunity [12,13]. These studies had shown that depletion of Tregs from the donor inoculum resulted in increased acute GVHD (aGVHD) severity. In accordance, high numbers of donor Treg suppressed aGVHD. In humans, paucity of Tregs in the incoming graft correlates with increased GVHD incidence and severity [14]. Some authors have shown no significant difference in the number of Foxp3-expressing CD4+CD25high T cells in patients with or without cGVHD [15] and others have shown that patients with cGVHD had elevated numbers of CD4+CD25high T cells in comparison with patients without disease when peripheral blood was analyzed [16]. Using Foxp3 mRNA expression to address Treg quantities, other authors have demonstrated that patients with cGVHD had significantly decreased Foxp3 message and this was corroborated by decreased numbers of CD4+CD25+Foxp3+T cells [17]. Two recent studies draw different conclusions using CD4+CD25high CD127low as phenotypic markers for Tregs regarding the extensive form of disease but not the limited form. Li, et al. [18] found a significant decrease on levels of Treg present in the peripheral blood of patients suffering from cGVHD when compared to healthy controls, while this difference was not found by Ukena, et al. [19].
The ability to comparatively analyze the different results regarding particular Tcell subsets as biomarkers at the diagnosis of cGVHD is hindered by different study end points, use of old classification of cGVHD (limited versus extensive disease) and concomitant use of systemic immunosuppression which can alter Treg quantification. Moreover, blood and tissue analysis might reveal different results and enlighten possible mechanisms of the disease [20-22].
With these limitations in mind we decided to use the NIH Consensus criteria to study a prospective cohort of patients with cGVHD. We asked if there were different infiltrate patterns regarding T cell subtypes between moderate and severe cGVHD and if there was a correlation with peripheral blood. We showed a paucity or even absence of Tregs in the target organs analyzed, while we observed a large fluctuation in the peripheral blood. Moreover, T cell infiltration showed a distinctive and typical pattern in all organs analyzed (mucosa, salivary gland, skin and liver) where CD8 T cells were predominant and in close contact with lesioned epithelial cells whereas CD4 T cells were found at distant sites from the lesion area. Altogether, these results suggest that CD8 T cells are the major effectors in cGVHD. We believe our findings can shed light in the use of CD8 T cell infiltration as a distinctive feature understanding the mechanisms underlying cGVHD.
Fifty-two consecutive patients transplanted at the Federal University of Rio de Janeiro (UFRJ) and Brazilian National Cancer Institute (INCA) between November/2006 and February/2010 were included. Samples from patients with untreated infections, who died before D+100 or from those that failed more than 70% of the scheduled visits of the protocol, were excluded, accounting for 42 included patients. Written informed consent was obtained from all subjects prior to blood and tissue sampling in accordance with the Declaration of Helsinki. The protocol was approved by the institutional review boards of both participating centers.
Chronic graft versus host disease was defined and classified in accordance with the NIH Consensus [23]. Peripheral blood samples were obtained from transplanted patients on days 30, 45, 60, 100, 180, and 360 after transplantation, at the onset of clinical cGVHD before systemic immunesuppression (in moderate and severe forms). Tissue biopsies, totaling 15, were indicated to confirm cGVHD diagnosis according to the NIH Consensus Criteria. As controls, we used buccal mucosal lesions samples from non-transplanted patients diagnosed with lichen planus de novo. Control blood samples were obtained from age-matched normal healthy volunteers with a median age of over 31 yrs (range 20-57).
In order to evaluate the percentage of total T and B cells in peripheral blood, 50 μL of blood samples were incubated for 30 minutes on ice with murine anti-human FITC, PE, PeRCP or APC direct conjugated CD45 (clone H130), CD3 (clone 1F4), CD4 (clone SK3), CD8 (clone SK1), all from BD Biosciences (CA, USA), and CD19 (clone SJ25-C1; Caltag Lab, Carlsbad, CA, USA). Treg quantification was performed in mononuclear blood cells obtained by Ficoll-Hypaque PlusTM (GE Healthcare Life Sciences, São Paulo, SP, Brazil) density gradient centrifugation. Samples were either frozen or incubated with the antibodies indicated above and Human Regulatory T Cell Cocktail CD25/CD127/CD4 (BD Biosciences, CA, USA). After surface labeling, cells were incubated with the lysing buffer (BD Biosciences) according to manufacturer's instruction and washed with buffered saline. Flow cytometry acquisition was performed with at least 50000 cells per sample using a FACSCalibur with Cell Quest Pro software (BD).
All samples were fixed in buffered 10% formaldehyde and routinely processed for paraffin inclusion. Paraffin slices were rehydrated and stained with Hematoxilin-Eosin (HE). Immunohistochemical staining was performed on 5 μm-thick serial sections using NovolinkTM Polymer Detection System (Novocastra Laboratories, New Castle, UK). After block of endogenous peroxidase, and recovery of antigen epitope with heated water bath, sections were incubated with the primary antibody overnight at 4 °C. Sections were then washed and incubated with the Novolink TM, according to manufacturer's instructions. The reaction was developed using a solution of 3-3-diaminobenzidine (DAB) with hydrogen peroxide. Nuclei were counterstained with hematoxylin. The following primary antibodies were used: anti-CD3 (CD3 polyclonalantibody), anti-CD4 (CD4 monoclonal antibody [mAb], clone SP35), anti-CD8 (CD8mAb, clone SP16) all from Cell Marque (Rocklin, CA, USA). For foxp3 staining mAb anti-FOXP3 (clone 236A/E7, Abcam, Cambridge, UK) was used.
Statistical analyses were performed using SPSS14.0 for Windows software (SPSS Inc.). Non-parametric statistical comparisons were performed using the Mann-Whitney U test. The evolution of subpopulations was compared using Two-way ANOVA. Differences were considered statistically significant when p values were less than 0.05.
A total of forty-two patients (42) receiving T cell replete grafts were enrolled in the study. They received bone marrow (n = 20, 46%), mobilized peripheral blood (n = 19, 45%) or umbilical cord blood (n = 3, 7%) transplants from related (n = 37, 88%) or unrelated donor (n = 5, 12%) (Table 1). Hematologic malignancies accounted for 36 (85%) and the majority received myeloablative conditioning (n = 37, 88%). All donors were HLA identical (10/10) except one (cGVHD negative), which had a single locus mismatch, and had a one-year follow up for the purposes of this study.
Table 1: Patient and transplant characteristics. View Table 1
In all related grafts, GVHD prophylaxis consisted of a calcineurin inhibitor and either methotrexate or mycophenolate mofetil. Sirolimus was not used in any patient. The only patient receiving the unrelated graft, used ATG as conditioning regimen. Chimerism on D+30 was ≥ 95% in all patients. Details of the subjects are summarized in Table 1. cGVHD was classified according to the last NIH consensus for GVHD [5-8].
From the 42 patients, 19 (45%) presented with moderate or severe form of cGVHD. Diagnosis median time was 7 months (3-16 months). Patients from both groups, with or without GVHD, had similar clinical and transplant characteristics. The only two significant differences observed between the groups was age and relapse rate. Patients with cGVHD were older than patients without cGVHD (median of 19 and 23 respectively, p = 0.005) and had fewer relapses from malignancy (Table 1) in accordance to the literature [24-27].
The predominant cGVHD type was the Classic Chronic accounting for 79% of all cases. Affected organs were, in order of incidence: oral cavity, skin, eyes and liver (Table 2), similar to what has been previously reported by other groups [6,26].
Table 2: Chronic GVHD characteristics at enrollment (n = 19). View Table 2
A total of fifteen biopsies were performed in different organs or systems, for diagnostic purposes, in lesions considered distinctive or diagnostic upon the NIH consensus classification for GVHD.
Leukocyte, as well as T and B lymphocyte reconstitution was not different from the results previously reported by other centers [27,28]. However, a tendency towards an increase in CD4 absolute number around D+100 was observed in patients with cGVHD that was followed by a decrease below levels observed in non-cGVHD patients (Figure S1A and Figure S1B). These variations, although not statistically significant, preceded cGVHD onset and might be related to an over activation of T cells, which then migrate to the target organs, leading to the observed variation in the number of peripheral blood CD4 T cells. Regarding circulating CD8 T cells, no differences between patients with or without cGVHD was observed (Figure S1A and Figure S1C). T regulatory cells (CD4+/CD25high/CD127low) were quantified in the peripheral blood in 24 allo-transplanted patients. The reconstitution pattern of Treg cells in the blood was extremely variable, with no differences between groups (Figure S1B and Figure S1C). We did not find any possible correlation between Tregs in the peripheral blood and the clinical event of moderate or severe cGVHD.
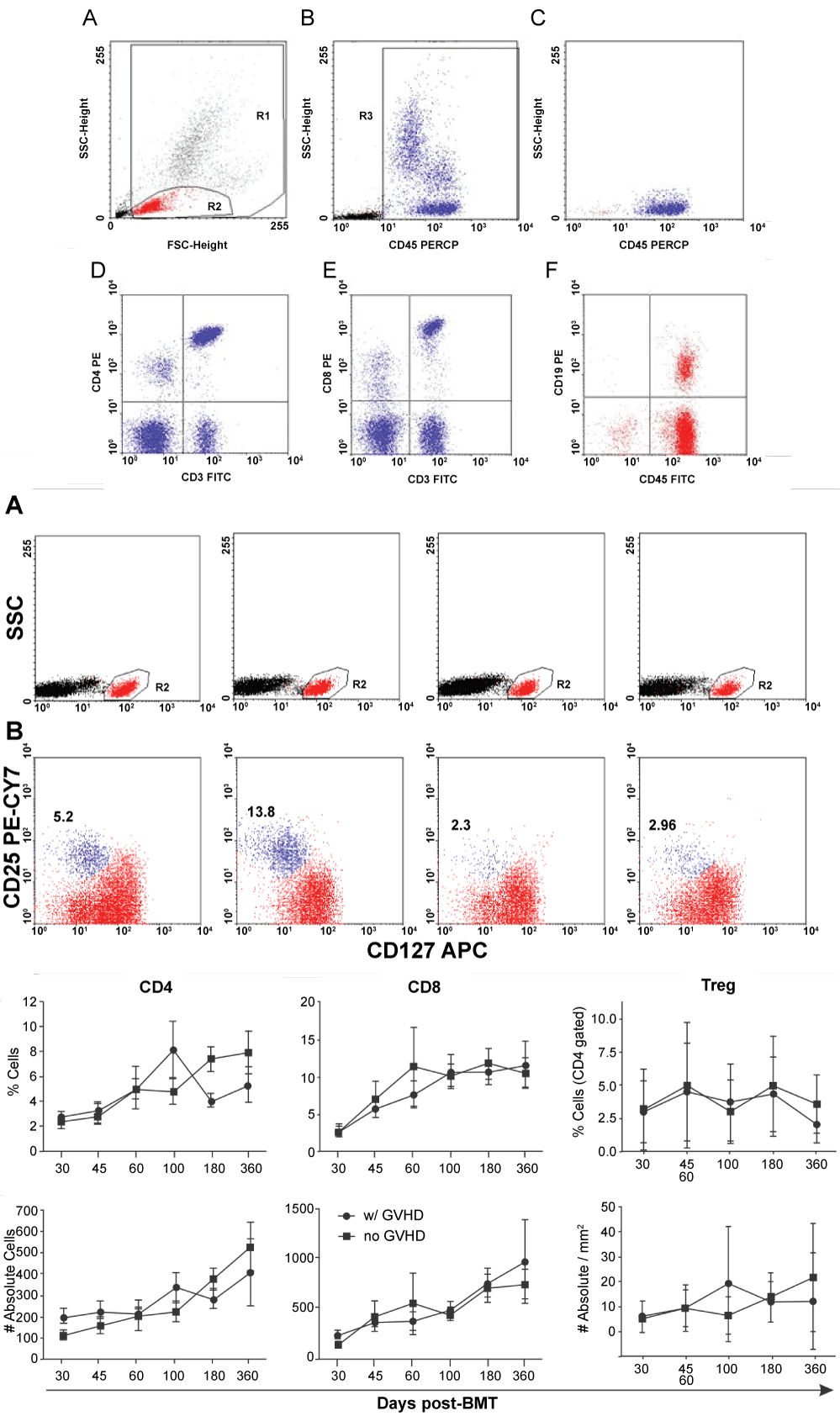 Figure S1: Evolution of CD4, CD8 and Treg subpopulations in peripheral blood of allogeneic hematopoietic stem cell transplanted patients. (A and B) Gate strategy for CD4 and CD8 and Treg analysis respectively. For Treg, plots of four different patients are shown; (C) Percent (top panels) and absolute (bottom panels) number of cells in patients with or without chronic GVHD are indicated. Data represent mean and error bars of 12 patients with and 12 patients without cGVHD. There was no statistical difference between the two groups for the populations studied.
View Figure S1
Figure S1: Evolution of CD4, CD8 and Treg subpopulations in peripheral blood of allogeneic hematopoietic stem cell transplanted patients. (A and B) Gate strategy for CD4 and CD8 and Treg analysis respectively. For Treg, plots of four different patients are shown; (C) Percent (top panels) and absolute (bottom panels) number of cells in patients with or without chronic GVHD are indicated. Data represent mean and error bars of 12 patients with and 12 patients without cGVHD. There was no statistical difference between the two groups for the populations studied.
View Figure S1
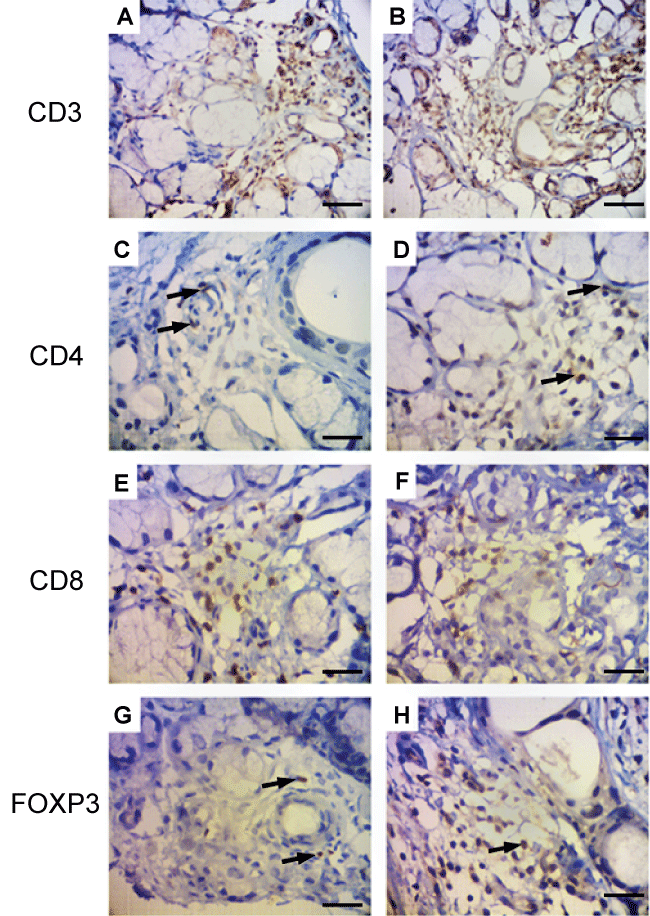 Figure S2: Salivary gland immunohistochemistry stainings in two patients with cGVHD. Samples from two individual patients are shown (A, C, E, G and B, D, F, H). Note intense CD3 infiltrate (A,B) with variable, but poor infiltration of CD4+ cells (C,D arrows). Inumerous CD8+ cells are seen in damaged areas (E,F). A paucity of FOXP3+ cells can be observed (G,H arrows). Bars = 100 μm (A,B), 50 μm (C,H).
View Figure S2
Figure S2: Salivary gland immunohistochemistry stainings in two patients with cGVHD. Samples from two individual patients are shown (A, C, E, G and B, D, F, H). Note intense CD3 infiltrate (A,B) with variable, but poor infiltration of CD4+ cells (C,D arrows). Inumerous CD8+ cells are seen in damaged areas (E,F). A paucity of FOXP3+ cells can be observed (G,H arrows). Bars = 100 μm (A,B), 50 μm (C,H).
View Figure S2
Since there was no difference in T cells from peripheral blood between patients who developed GVHD or not, skin, oral cavity or liver biopsies were performed in 14 patients. One patient with overlap syndrome was submitted to both, cutaneous and oral cavity biopsies.
Oral Cavity histopathological analysis of lip mucosa and salivary glands were performed in patients with unique oral complains. Lip mucosa pathology showed parakeratinized epithelia with dissolution of the cellular basal layer associated with acanthosis. Mononuclear infiltration and fibrosis of the lamina propria were observed. Immunohistochemistry of the oral mucosa showed cGVHD lesion with CD3+ T cells infiltration of the submucosa with some CD3+ cells within the epithelial layer (Figure 1A). Curiously, CD4+ cells were restricted to the submucosa (Figure 1B), while CD8+ cells were seen in the submucosa as well as within epithelial layer, in close contact with vacuolated keratinocytes (Figure 1C and Figure 1D). Foxp3+ cells were rare and present in the submucosa. (Figure 1E, Figure 1F and Figure 1G). As control samples we used biopsies samples from de novo Lichen planus patients as this is not associated with GVHD. In oral biopsies from Lichen planus CD3+ cells were seen in the submucosa and intraepithelial layer (Figure 1H). However, in contrast to cGVHD associated lesions, CD8+ cells (Figure 1I) as well as CD4+ cells (Figure 1J and Figure 1K) were seen in the sub mucosa and within epithelial layer. Foxp3+ cells were observed within lymphocytic infiltration and were more numerous than the same population observed in lichenoid lesions from transplanted patients (Figure 1L and Figure 1M).
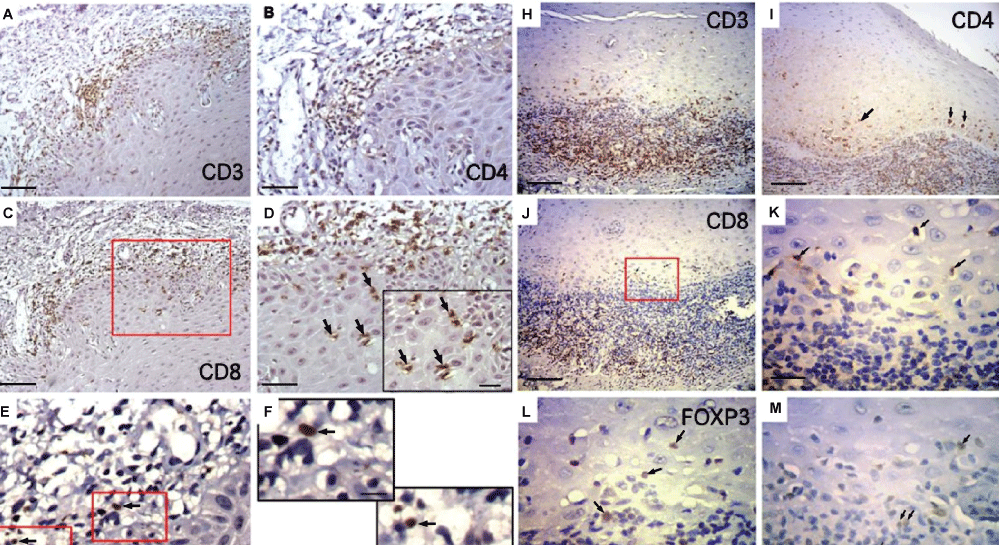 Figure 1: Immunohistochemistry for CD3, CD4, CD8, and foxp3 of representative oral mucosa biopsies from patients with cGVHD or control. (A) (AG) cGVHD lesion showing CD3+ T cells infiltration of the submucosa with some CD3+ cells within epithelial layer; (B) CD4+ cells were restrict to the submucosa; (C-D) While CD8+ cells were seen in the submucosa as well as within the epithelial layer (arrows in D that shows detail of area inside the red square); (E-G) Insert shows detail of CD8 intraepithelial infiltration (arrows); (F-G) Rare foxp3+ cells (arrows) were seen in the submucosa; (H-M) Details of the areas inside the red squares; (H) Control Lichen planus de novo, not associated with GVHD, show CD3+ cells in the submucosa and in epithelial layer; (I) CD8+ cells; (J,K) As well as CD4+ cells; (L-M) Were seen in the submucosa and within epithelial cells. Some foxp3+ cells were present within lymphocytic infiltration (A,C,H,I,J) Bars = 100 μm, (B,D-E) 50 μm, (D,K,L,M) 25 μm (insert in), (F-G) 10 μm.
View Figure 1
Figure 1: Immunohistochemistry for CD3, CD4, CD8, and foxp3 of representative oral mucosa biopsies from patients with cGVHD or control. (A) (AG) cGVHD lesion showing CD3+ T cells infiltration of the submucosa with some CD3+ cells within epithelial layer; (B) CD4+ cells were restrict to the submucosa; (C-D) While CD8+ cells were seen in the submucosa as well as within the epithelial layer (arrows in D that shows detail of area inside the red square); (E-G) Insert shows detail of CD8 intraepithelial infiltration (arrows); (F-G) Rare foxp3+ cells (arrows) were seen in the submucosa; (H-M) Details of the areas inside the red squares; (H) Control Lichen planus de novo, not associated with GVHD, show CD3+ cells in the submucosa and in epithelial layer; (I) CD8+ cells; (J,K) As well as CD4+ cells; (L-M) Were seen in the submucosa and within epithelial cells. Some foxp3+ cells were present within lymphocytic infiltration (A,C,H,I,J) Bars = 100 μm, (B,D-E) 50 μm, (D,K,L,M) 25 μm (insert in), (F-G) 10 μm.
View Figure 1
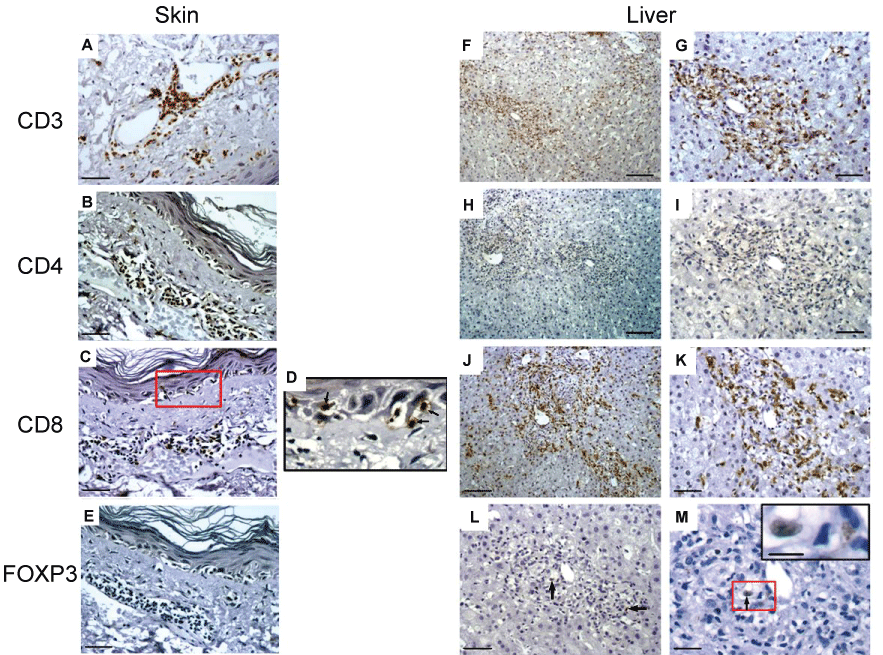 Figure 2: Immunohistochemistry for CD3, CD4, CD8, and foxp3 of representative skin and liver biopsies from a patient with cGVHD. (A) CD3+ cells in the dermis, around blood vessels and nearby the epithelium; (B) CD4+ cells were located exclusively in perivascular areas; (C-D) CD8+ cells were seen in perivascular regions (arrowheads) and with n acantholyctic keratinocytes (arrows); (E) Foxp3+ cells were almost absent. Bars = 100 μm (B-C, E), 50 μm (A), 25 μm (D); (F-G) Massive infiltration of portal space by CD3+ cells; (H-I) Few CD4+ cells within mononuclear infiltrate of portal space; (J-K) CD8+ cells infiltrate perilobular regions and portal space; (L-M) Rare foxp3+ cells (arrow) were observed within mononuclear infiltrate. Insert in G showed detailed of area insides the red square. Bars = 100 μm (F, H, J), 50 μm (G,I,K,L), 25 μm (M), 10 μm (insert in M).
View Figure 2
Figure 2: Immunohistochemistry for CD3, CD4, CD8, and foxp3 of representative skin and liver biopsies from a patient with cGVHD. (A) CD3+ cells in the dermis, around blood vessels and nearby the epithelium; (B) CD4+ cells were located exclusively in perivascular areas; (C-D) CD8+ cells were seen in perivascular regions (arrowheads) and with n acantholyctic keratinocytes (arrows); (E) Foxp3+ cells were almost absent. Bars = 100 μm (B-C, E), 50 μm (A), 25 μm (D); (F-G) Massive infiltration of portal space by CD3+ cells; (H-I) Few CD4+ cells within mononuclear infiltrate of portal space; (J-K) CD8+ cells infiltrate perilobular regions and portal space; (L-M) Rare foxp3+ cells (arrow) were observed within mononuclear infiltrate. Insert in G showed detailed of area insides the red square. Bars = 100 μm (F, H, J), 50 μm (G,I,K,L), 25 μm (M), 10 μm (insert in M).
View Figure 2
Regarding salivary gland, acinar atrophy was associated with mononuclear infiltration. Infiltrating cells followed the same pattern as to the oral mucosa, with a predominance of CD8 cells close to the damaged area and variable, but less, CD4+ cells within the gland. Again, Foxp3+ cells were rare. (Figure S2).
Sclerodermoid lesions (n = 3) showed epidermis atrophy with interpapillary cone rectification and moderate hyperkeratosis. Papillar dermis was thickened, with increased collagenous deposition and poor, or virtually absent, cell infiltrate. Immunohistochemistry analysis was not performed in these samples.
GVHD-associated Lichen planus-like lesions (n = 4) in skin showed moderate to intense perivascular lymphocyte infiltration in the superficial dermis with variable tropism for the basal layer. Atrophy of the epidermis was accompanied by hiperkeratosis, acanthosis and variables areas of vacuolization within the basal layer. Immunostaining for CD3 revealed association with blood vessels and nests in the epidermis (Figure 2A). Regarding CD4+ cells, they were found preferentially in the perivascular areas and superficial dermis, with rare events in the epidermis (Figure 2B). On the other hand, CD8+ cells were found infiltrating the epidermal layer and associated with areas of vacuolated keratinocytes (Figure 2C and Figure 2D), resembling the pattern observed in the oral cavity GVHD-associated lesions. Rare Foxp3+ cells were observed, and when present they were restricted to the superficial dermis (Figure 2E).
Liver specimens were obtained from two patients with cGVHD in order to rule out other possible causes of hepatic dysfunction. Serological tests for viral hepatitis were negative and the biopsies were performed 7-10 days after immunosuppressive therapy was started. Periportal and ductal CD3+ lymphocyte infiltration was observed (Figure 2F and Figure 2G), with the predominance of CD8+ cells and almost no CD4 (Figure 2HK).
In one of those patients, fulminant hepatic failure was correlated with the intense infiltrate. Again, only rare Foxp3+ cells were observed (Figure 2L and Figure 2M).
Altogether, our data describe a T cell infiltration pattern in cGVHD lesions, common to all organs studied, where CD8+ cells infiltrate the target tissue and are positioned juxtaposed to damaged areas, whereas CD4 cells diffusely infiltrates the subjacent connective tissue or are observed around blood vessels. Moreover, Treg infiltration is basically absent in the cGVHD tissues analyzed and this is different from, at least, one lichen planus analyzed here, which was not associated with cGVHD.
In this prospective study, CD4+, CD8+ and Treg cells were evaluated in both, peripheral blood and target tissues in patients with moderate and severe forms of cGVHD classified according to the recent NIH consensus [5-9]. Although our study is very limited due the number of patients and samples, it raises interesting points, which can be helpful for further studies regarding cGVHD characterization. First, except for one patient, which had been under immunesuppression in the last ten days before the biopsies were performed, all samples were collected for diagnosis, in patients not under immune-suppressive drugs. Second, all tissues analyzed show a characteristic pattern of infiltration, which is different from the same clinical entity in one of the cases, derived from non-GVHD pathologies. Our study did not find differences in blood levels of Treg between patients without or with moderate to severe cGVHD at diagnosis. Also, a long lasting reduction in CD4+ cell reconstitution, affecting similarly regulatory and conventional cells, was present. Those results are in accordance with previously published reports [13-15].
Previous retrospective studies had shown quantitative differences in Treg numbers in tissue lesions in cGVHD. In one of them chronic GVHD was solely based on day of onset (after D+100) different from the new NIH criteria for GVHD diagnosis. Low TGI samples showed decreased numbers of Treg when compared to non-GVHD transplanted patients and most patients were under immunosuppressive agents [29]. Another study analyzed skin samples and showed a decreased number of Tregs in 9 cGVHD patients when compared to skin samples from healthy subjects [30]. Ukena, et al. compared Treg transcriptomes from patients with and without GVHD and uncovered regulated gene transcripts highly relevant for Treg function [31].
Their results pointed to a reduced suppressive function of Tregs from GVHD patients with diminished migration capacity to the target organs, suggesting qualitative deficits in Tregs in GVHD, which could explain the paucity of Treg found in the tissues analyzed here.
Regarding CD4 T cell infiltration in the skin Fondi, et al. [30] found it to be basically restricted to the superficial dermis and dermoepithelial junction in accordance to our results. On the other hand, Grogan, et al. [32] observed a sustained activation profile of CD8+ cells in the peripheral blood in patients with active cGVHD consistent with the hypothesis that this subpopulation plays a role in mediating the immunopathology of cGVHD in humans. In fact, our data point to a role of CD8 subpopulation as effector cells in cGVHD physiopathology as these cells were found juxtaposed to damaged areas in all tissues analyzed from cGVHD patients. However, in samples derived from other inflammatory diseases used as controls, as lichen-planus unrelated to cGVHD, CD4+ cells were present within the epithelial layer, whereas if the lesion was derived from cGVHD patients, the CD4 infiltration is restricted to the dermal layer. Importantly this same distinctive pattern present in the oral mucosa was found in the skin and liver, ie. Exclusive juxtaposition of CD8 T cells, and not CD4, to the lesioned area suggests a distinctive cGVHD histopathological sign. Our data, obtained from biopsies taken at the time of the appearance of diagnostic clinical lesions, in patients not under immunosuppression and classified in accordance to the recent NIH consensus, show a uniform pattern throughout the different tissues analyzed. Although we are aware of the small number of patients analyzed, the consistent clinical and histopathological findings described above, should be further studied to check if this pattern can indeed be used as distinctive marker for cGVHD.
M.C.R.M, M.I.D.R and A.B. designed the study, performed research, analyzed data and wrote the paper. R.G.G. wrote the paper collected and analyzed data. D.O.P, S.A.P.S, collected and analyzed data. E.A. and A.C.P. analyzed data.
We thank Ana Paula Fontão and Gabriela Lemos for excellent technical assistance, Heitor Barros for help with the statistics. We are in indebted with Professor Angelo Maiolino and Professor Sandra Torres from Federal University of Rio de Janeiro and Dr. Luiz Fernando Bouzas, Head of CEMO/INCA for supporting the present study. This work was supported by CNPq (# 30.8961/2007-2, # 47.1108/2007-3, # 306624/2010-9), FAPERJ (# E-26/102.898/2011, # E-26/110.323/2010).
The authors declare no financial disclosure.