Introduction: Pancreas graft thrombosis following simultaneous pancreas-kidney transplantation is the leading non-immunologic cause of graft loss. Although essential to prevention of thrombotic failure of the new graft, systemic anticoagulation is controversial as it may lead to an increased risk of postoperative life threatening bleeding.
Methods: A total of 35 simultaneous pancreas-kidney transplants were performed in our Transplant Centre between 2018 and 2020. Patients were divided in two retrospective cohorts according to the anticoagulation protocol. Group 1 received low-molecular-weight-heparin (N = 20) and Group 2 received intravenous unfractionated heparin infusion for 5 days (N = 15). Acetylsalicylic acid 100 mg/day was administered to all patients. Data concerning anticoagulation protocol, demographic and clinical patient's characteristics, surgical and medical complications and mortality were recorded and analyzed.
Results: We documented a relevant reduction in pancreas graft survival rate (70% vs. 86.7%; P-value = 0.228) and a higher incidence of hemorrhagic complications (40% vs. 6.7%; P-value < 0.05) in Group 1. No significant differences were found in biochemical graft variables. Hospital mortality between groups was similar.
Conclusions: To conclude, using a heparin infusion protocol in the postoperative period of SPK transplantation may reduce pancreas graft loss and significantly reduces the risk of bleeding.
Simultaneous pancreas-kidney transplantation, Pancreas transplantation, Graft survival, Thrombosis, Anticoagulation, Heparin
Simultaneous pancreas-kidney (SPK) transplantation is the preferred treatment option for selected patients with end stage renal disease (ESRD) and type 1 diabetes mellitus (T1DM). Among all pancreas recipients, which includes those submitted to pancreas after kidney (PAK) or pancreas alone transplantation, those with a SPK transplantation have the best graft survival rate (85% at 1 year and 50% at 10 years for SPK vs. 80% and 30% to the former two) [1]. Moreover, there is only one surgical intervention and one source of foreign human leukocyte antigen (HLA) to which the patient is exposed [1]. A successful SPK transplantation restores optimal glycemic control, and therefore stabilizes other T1DM secondary complications such as diabetic retinopathy and neuropathy, cardiovascular disease and life-threatening hypoglycemic episodes. It consequently improves patient survival and quality of life [1,2].
Despite great advances, either from surgical procedures, organ preservation, perioperative care or immunosuppression, SPK transplantation is still a complex procedure, associated with significant graft loss, morbidity and mortality rates [2,3]. Surgical complications like hemorrhage and vascular graft thrombosis (VGT) are detrimental to pancreatic graft survival, occurring early and frequently (usually during the first week), and up to 35% of all transplants receptors [1,4]. Furthermore, as pancreas is a relatively low-blood-flow organ, pancreatic VGT is usually irreversible, representing the most common nonimmunologic cause of pancreatic graft failure [1,3,5,6]. The appropriate use of anticoagulants such as heparin aims to reduce the rate of VGT [4-6]. However, in order to prevent thrombosis with the least risk of bleeding, an effective and safe immediate postoperative thromboprophylaxis protocol for SPK transplantation recipients still lacks [5]. We present a retrospective study of SPK transplantation patients, comparing the effectiveness and safety of two anticoagulation heparin based protocols within the first 30 days of SPK transplant.
We included all SPK transplant patients carried out at Hospital Curry Cabral, Centro Hospitalar Universitário de Lisboa Central from January 2018 to February 2020 and divided them in two groups. Group 1 (N = 20) included SPK transplants from January to December 2018 who received low-molecular-weight-heparin (LMWH). Group 2 (N = 15) included SPK transplant from January to December 2019 who received intravenous unfractionated heparin infusion (UFH). Acetylsalicylic acid 100 mg/day (ASA) was initiated to all candidates before transplantation, at the moment they entered on the SPK transplant active waiting list.
A retrospective observational study was performed in order to evaluate clinical effects of our new SPK transplants anticoagulation protocol. All patients started ASA preoperatively and continued it after transplantation. Before the end of 2018, all patients additionally received prophylactic anticoagulation with LMWH right after surgery. Since January 2019, we implemented a new anticoagulation protocol: UFH was started immediately after surgery (500 units per hour) and continued for 5 days. Perfusion dose was adjusted every four hours in order to achieve a partial thromboplastin time (PTT) ratio of 1.5. Perfusion dose was incremented or reduced 1 unit per kg per hour in order to keep PTT in the defined range. In groups, demographic data, donor characteristics, graft function variables, incidence of complications and survival data were collected. After surgery, all patients were admitted to the intensive care unit (ICU) for surveillance and tight grafts function monitoring. Pancreas vascular graft function was defined as the primary outcome.
Infection was defined as the presence of fever (tympanic temperature higher than 38.5 °C) in at least two consecutive days with organ associated symptoms that could not be attributed to other cause. Moreover, these findings had to be associated with an increase in acute phase biochemical parameters and result in the initiation of antibiotics or antifungal treatment. Hemorrhagic complication was defined by a decrease of 1 gr in hemoglobin in less than 24 hours which led to a temporary suspension in the anticoagulation therapy.
Local Ethical Committee approved this study and its publication; written informed consent was waived by the ethics committee.
After a midline incision, the right iliac vein and artery were totally clamped and an appropriate vein-to-vein and artery-to-artery anastomosis was performed. Donors' duodenum was anastomosed to an intestinal loop of the receptor. The pancreas was placed horizontally.
All patients received induction with 500 mg intravenous (IV) methylprednisolone and 1.5 mg/kg IV thymoglobulin before surgical incision. Methylprednisolone was continued on day 1 and 2 and then switched to oral 20 mg prednisolone daily. Mycophenolic acid was started on day 1 250 mg IV twice daily and then switched to oral 180 mg twice daily at day 5. Tacrolimus IV 0.01 mg/kg/day perfusion was started during the first 12 hours after ICU admission and titrated to 10 ng/mL and then switched to per os at day 5.
Blood test samples were collected twice daily during the first 2 days and then once daily in order to assess graft function. These included full-blood count, activated partial thromboplastin time, prothrombin time, glucose, C peptide, kidney and liver function parameters, and C-reactive protein. Doppler ultrasound was carried out on postoperative day 1 and repeated as needed. If clinical and radiological imaging suspected abnormal blood flow, computed tomography with intravenous contrast was performed.
Continuous and categorical variables were described as median (interquartile range [IQR]) and frequency (percentage [%]), respectively. Mann-Whitney test was used for comparison between continuous variables of the two groups. Chi-square was used in categorical variables. The survival rate for pancreas graft was calculated using the Kaplan-Meier method. Statistical significance was set at P < 0.05. Statistical analysis was performed using SPSS version 20.0 (IBM Corp. Armonk, NY, USA).
Thirty-five patients were included in this study. Twenty received postoperative low-weight molecular heparin therapy (Group 1) and were compared with fifteen patients who received unfractionated heparin infusion (Group 2). Donor and recipient characteristics were recorded in Table 1. No significant differences between groups were found concerning recipient's age, gender, body mass index or number of years of diabetes T1DM vintage (difference in years since diagnosis and present day). None of the patients had been previously submitted to transplant. Donor characteristics were similar in both groups and no significant statistical differences were found. All patients received grafts from neurological death donors.
Table 1: Donor and recipient characteristics. View Table 1
Postoperative complication rates can be found in Table 2. LMWH Group presented a higher rate of hemorrhagic complications (40% vs. 6.7%) in comparison to UFH Group (P-value < 0.05). The median day of hemorrhage was similar in both groups as well as the median number of overall packed red blood cells transfused during hospital stay, as seen in Table 3 and Figure 1. Both groups did not differ significantly in median hemoglobin or hematocrit levels. The greatest difference was found on day 5, although it was not statistically relevant (median hemoglobin 82 [76-89] vs. 73[71-81] g/L; P-value = 0.062). Moreover, no significant differences were found in serum amylase or lipase, nor estimated glomerular filtration rate (eGFR) - Figure 1. The number of infections during ICU stay was superior in UFH Group (5% vs. 20%). All cases included intra-abdominal infections, mainly due to gram negative bacteria. There was only one death during ICU stay, belonging to a patient from the LMWH group. Nevertheless, hospital mortality rate between groups was quite similar.
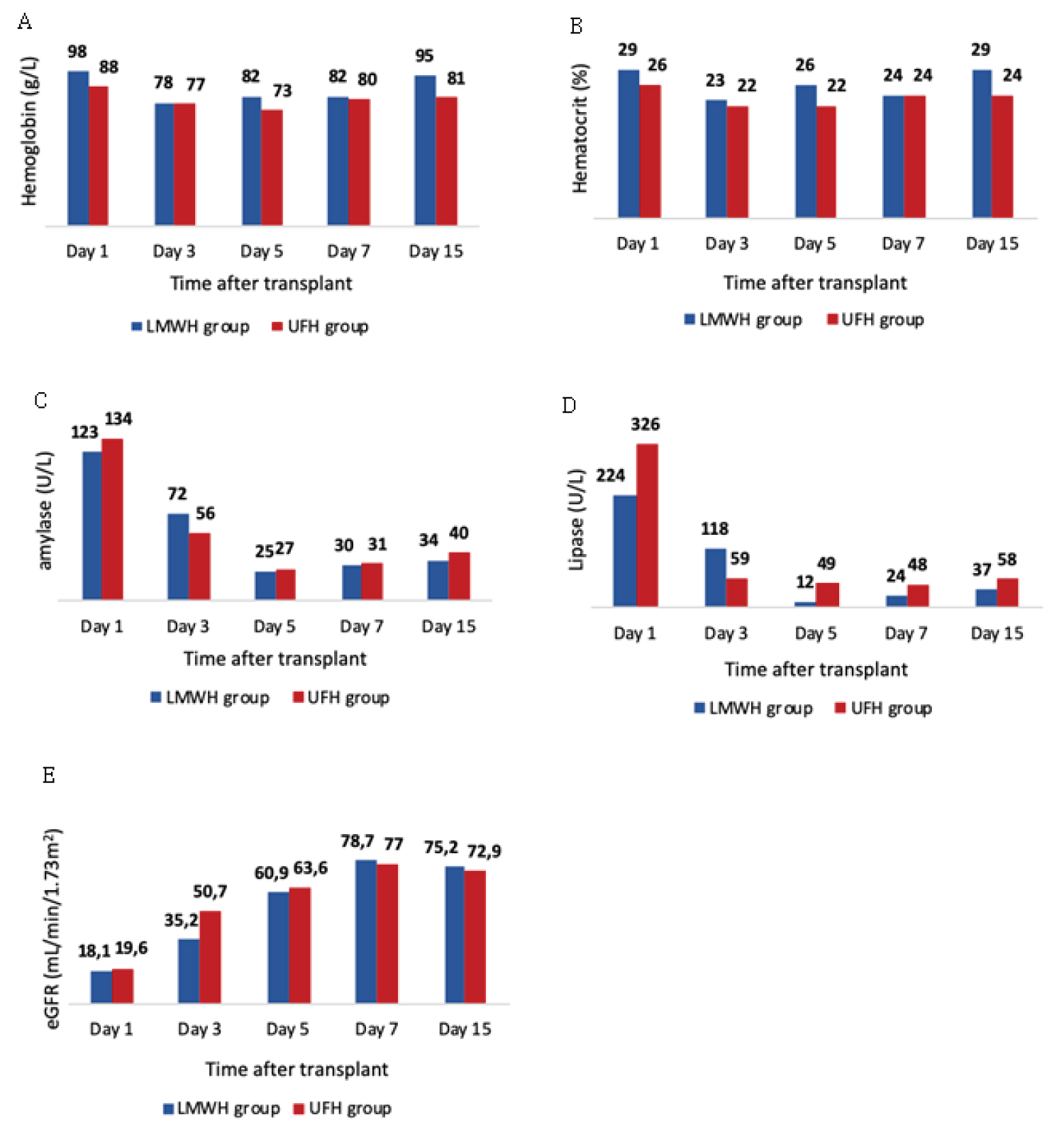 Figure 1: Variation of hemoglobin (A), hematocrit (B), serum amylase (C), serum lipase (D) and estimated glomerular filtration rate (E) during 15 days in patients submitted to simultaneous pancreas-kidney transplantation. Values are expressed as median.
View Figure 1
Figure 1: Variation of hemoglobin (A), hematocrit (B), serum amylase (C), serum lipase (D) and estimated glomerular filtration rate (E) during 15 days in patients submitted to simultaneous pancreas-kidney transplantation. Values are expressed as median.
View Figure 1
Table 2: Post-operative complication rates and mortality. View Table 2
Table 3: Parameters evolution after transplantation. View Table 3
A relevant distinction was found in pancreas graft survival rate. The incidence of pancreas graft thrombosis and consequently graft loss was greater in LMWH Group (6 vs. 2 patients; 30% vs. 13.3%). The median day of graft loss was slightly higher in UFH Group, (day 3 vs. day 5 since SPK transplantation). Using Kaplan-Meier method, UFH Group patients had a functioning pancreas graft of 86.7% up to 30-days after hospital admission, compared with 70.0% pancreas graft surviving in LMWH Group (P-value = 0.228), as seen in Figure 2.
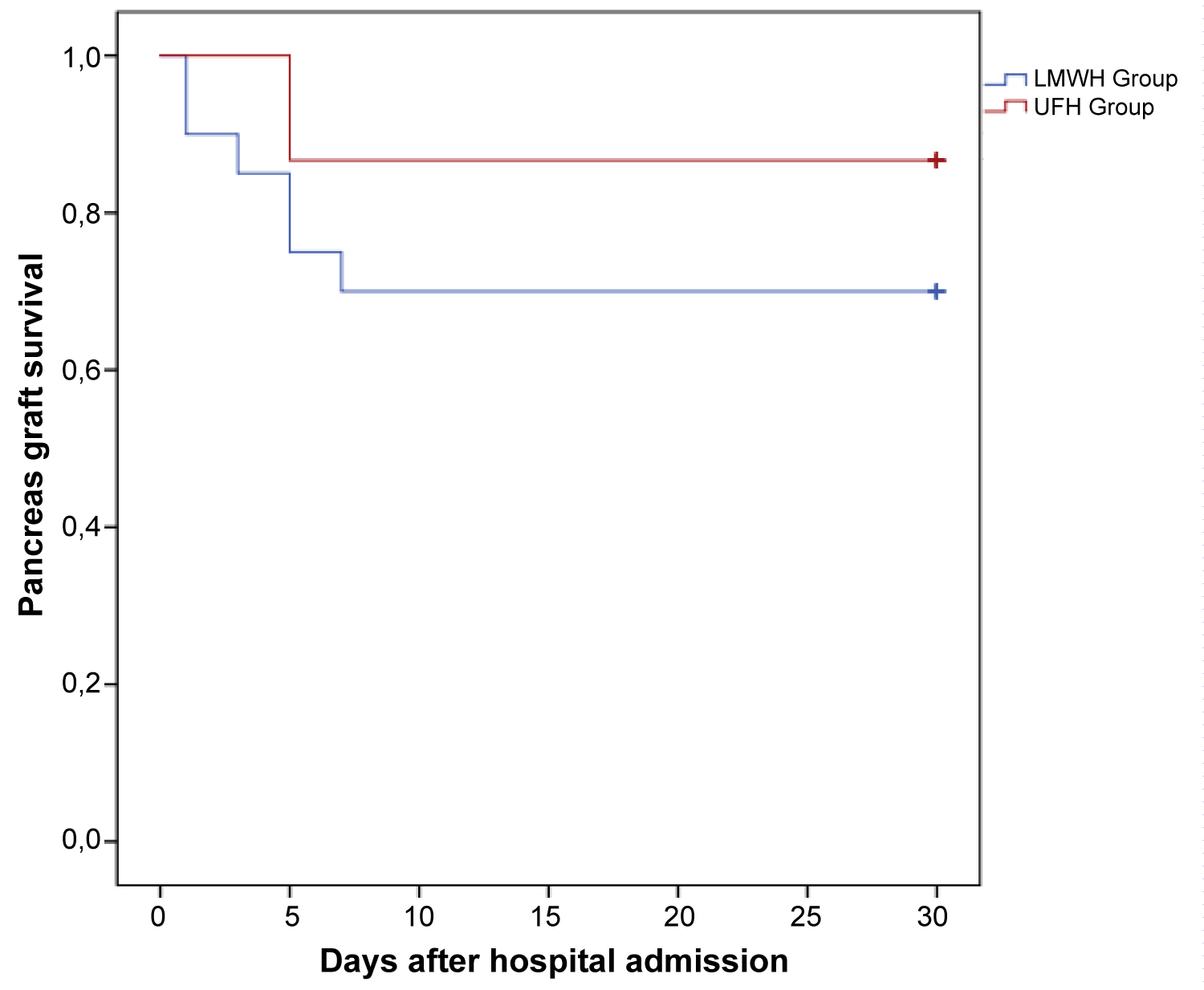 Figure 2: Pancreas graft survival following simultaneous pancreas-kidney transplantation.
View Figure 2
Figure 2: Pancreas graft survival following simultaneous pancreas-kidney transplantation.
View Figure 2
SPK transplantation improves overall survival and quality of life in patients with T1DM and ESRD [2,5,7]. Despite it remains a complex procedure, pancreas graft loss mainly due to thrombosis, may occur in up to one third of the patients [8,9]. Ischemic microvascular injury secondary to donor risk factors, organ preservation and reperfusion injury, inherent hypercoagulability and decreased fibrinolysis of T1DM patients may play an important role as high risk factors for early postoperative graft thrombosis [8,10]. Nevertheless, as there is still no solid recommended anticoagulation protocol for these patients, distinct approaches have been adopted in different centers.
The Japanese group of Okabe, et al. prefer not to anticoagulate SPK transplant recipients due to the postoperative high risk of uncontrollable bleeding. Instead, they use intensive fluid infusion as a way to decrease hematocrit and blood viscosity, [7] both a risk factor for thrombosis. In contrast, Scheffert, et al. performed a retrospective single-center evaluation of patients in postoperative period after pancreas transplantation and received infusion of 500 UI/h UFH for two days. This approach reduced the number of graft thrombosis, despite the absence of statistical significance [9]. Alternatively, Schenker, et al. compared the use of LWMH to therapeutic UFH following all types of pancreas transplants, [6] and concluded that one daily fixed dose of LWMH was not inferior to UFH in preventing pancreas graft thrombosis, without differences in major bleeding requiring relaparotomy [6]. Aboalsamh, et al. performed a similar study using smaller doses of UFH vs. only acetylsalicylic acid 81 mg/day and observed a significant reduction in graft thrombosis and graft loss in patients receiving UFH, with no differences in complication rates [5].
The anticoagulant effect of heparin is related to its binding to antithrombin III which enhances its ability to inhibit thrombin, factor Xa, IXa and XIIa, reducing fibrin formation and thrombin-induced activation of factor V and factor VIII [9]. Moreover, heparin also binds to plasma proteins, endothelial cells and macrophages, interfering with the clotting factor complex adhesion to endothelial cell surface membranes ensuing thrombus formation [11]. According to Tyrell, et al. and Young E, heparin also possesses anti-inflammatory effects mediated by interference with leukocyte recruitment and adhesion to vascular endothelium [12,13]. These may be more significant following ischemia-reperfusion injury post-transplant which induces a pro-inflammatory state with activation of neutrophils, inflammatory cytokines and endothelial cells [9].
We applied a new protocol to SPK transplantation patients, aiming the reduction on pancreas VGF, without increasing hemorrhagic complication rates. In case of bleeding, UFH infusion has a safer profile as it can be stopped immediately and is more reliably reversible with protamine sulfate. Furthermore, we did not use a fixed dose of heparin infusion. Instead we defined a target which takes into count variability between patients and avoids excess anticoagulation. Factors related to lower graft survival such as advanced donor age and high body mass index were similar between groups. Besides all grafts presented with a cold ischemia time lower than 8 hours, UFH group had a slightly higher time (Table 1) which theoretically could increase their risk of thrombosis [5].
Our new protocol reduced pancreas graft thrombosis rate to 13.3% and improved graft survival from 70 to almost 87% in the first 30-days after SPK transplant. Moreover, this benefit was not associated with an increase in the bleeding's rate. Precisely, the number of hemorrhagic complications presented a sharp decrease from 40 to less than 7%, as seen in Table 2 (P-vale < 0.05). Despite the median hemoglobin values being lower in UFH group (Figure 1), the median number of administered packed red blood cells was similar in both groups. These findings can be explained by the fact that using anticoagulation with UFH infusion gives medical team more confidence and control in the anticoagulation protocol, avoids its suspension and, consequently, reduces the risk of graft thrombosis.
Among the limitations of our study, it was a retrospective study with a small cohort of patients. Furthermore, both groups were compared historically (UFH group was more recent than LWMH group). Surgical team remained the same during both periods, and none changed their surgical technique over time. Although it was not described in this study, intraoperative management must be included in future studies as it may influence grafts survival.
To conclude, using an intravenous unfractionated heparin infusion protocol in SPK transplantation may reduce pancreas graft loss as well as the risk of bleeding.
Tiago Isidoro Duarte and Joana Marques designed the study, collected and analyzed data and wrote the manuscript; Nuno Germano designed the study, read and revised the manuscript; Franscico Remedio, Jorge Paulino, Américo Martins, Aníbal Ferreira e Fernando Nolasco read and revised the manuscript.
All authors do not have any conflicts to declare.