To evaluate the effect of EGF in primary culture of ulcer patients and N87 cell line on expressions of apoptotic genes. Ulcer patients who applied to Gastroenterology Clinic of Mersin University Medical Faculty were included in this study. EGF application to the primary culture of biopsy samples with ulcer patients and gastric cancer cell-line (ATCC-NCI-N87) has been carried out at three different doses. The expression levels of Bax, Bcl-2 and Fas genes were measured with Quantitative Real-Time PCR (qRT-PCR). ΔΔCT analysis with qRT-PCR revealed no significant change in gene expression of Bax, Bcl-2 or Fas within the ulcer, normal tissue and gastric cancer. No significant change was determined between Bax and Bcl-2 gene expression levels and applied EGF doses when groups were compared for each EGF dose. On the other hand, when 50 ng/μl of EGF was administered, Fas mRNA expression level was significantly lower in gastric cancer cell line compared to patients with ulcer and normal gastroduodenal tissue (p ˂ 0.05). In this study which was done with a restricted patient group, our results revealed that apoptosis induced by Fas expression in gastroduodenal suppressing carcinogenesis process plays an active role in gaining anti-apoptotic properties of cells.
EGF, N87, Primary culture of gastric epithelial cells, Fas, Bcl-2, Bax
The gastric mucosa is normally exposed to a wide range of aggressive factors and has developed efficient mechanisms to repair tissue injury. In the case of superficial damage, the regeneration of the epithelial surface is very rapid. In the case of deeper mucosal damage, such as erosions and ulcers, both epithelial and connective tissue components, including sub-epithelial myofibroblast, smooth muscle cells, vessels, and nerves are destroyed and must be regenerated. Polypeptide growth factors such as Epithelial Growth Factor (EGF) have received much attention in recent years because of their ability to regulate essential cell functions involved in tissue healing including cell proliferation, migration, differentiation, secretion [1]. EGF is synthesized by submandibular glands, Brunner's glands of the duodenum, and Paneth cells of the small intestine, but not in the normal gastric mucosa. The removal of submandibular glands in rats, which are the major source of EGF in the gastric fluid, although not sufficient to cause spontaneous ulcer development, may delay the healing of gastric ulcer, while oral administration of synthetic EGF can accelerate ulcer healing [2]. In humans, salivary levels and gastric fluid concentrations of EGF are reduced during the active phase of peptic ulcer disease, but are restored to normal levels after ulcer healing is complete.
Apoptosis is an essential part of the cycle of cellular turnover in many tissues, including the gastrointestinal tract. Apoptosis has been reported to take place in all regions of the stomach, occurring predominantly in the upper part of the gastric glands and involving 2%-3% of all epithelial cells [3]. On the onset of gastric ulceration, the rate of apoptosis rapidly increases. The 3.9 fold increase in mucosal expression of Caspase-3 activity can be detected as early as 2 hours after experimental ulcer induction [4]. Conceptually, apoptosis is an active and regulated process by which cells self-destruct. Deranged apoptosis has been implicated in carcinogenesis, autoimmune diseases, and various infectious diseases including Helicobacter pylori infection [5]. The regulation of apoptosis is a complex process that includes the activation of various apoptosis-related proteins such as the BCL2-associated X protein (BAX), B-cell CLL/Lymphoma 2 (BCL-2), p53, TNF receptor super family, member 6 (FAS), Fas Ligand (FASL) and Interleukin-1β-related Concerning Enzyme (ICE) family [6]. There are two pathways that lead to apoptosis: the positive induction by ligands binding to plasma membrane receptors and negative induction by the loss of suppressor activity. One pathway involves apoptosis mediated by death receptors, such as CD95 (Fas) and tumor necrosis factor receptors.
The Bcl-2 proteins are the best-studied family of proteins involved in the mechanism of apoptotic cell death. The first member of this family, Bcl-2, was discovered in human B-cell lymphomas at a chromosomal translocation t (14:18) where it was thought to lead to malignant transformation because of its ability to prevent apoptotic cell death [7]. Some members of the Bcl-2 family such as Bax promote apoptosis whereas the other members such as Bcl-2 and Bcl-xL inhibit this process. The susceptibility of a cell to apoptosis depends on the balance between apoptosis-promoting and apoptosis-suppressing factors [8].
Expression of genes related to apoptosis in both mRNA and protein levels have been studied in various types of cancer like gastric cancer; however the effect of epidermal growth factor on gene expression has not been examined on ulcer tissues.
Apoptotic cell death is a highly regulated process, which plays a crucial role in many biological events. The regulation of apoptosis at several levels is essential to maintain the delicate balance between cellular survival and death signaling that is required to prevent disease. Apoptosis can be triggered by signals from within the cell or by extrinsic signals, such as the growth factors. Several mechanisms have been implicated in the pathogenesis of stress ulcers such as increase in gastric acid and pepsin secretion, a decrease in gastric blood flow. EGF exhibits gastroprotective and ulcer healing properties, presumably due to mitogenic and growth promoting actions. It is also known that EGF family members in gastric cancer are elevated in expressions and suppressed apoptosis leading to carcinogenesis [9].
The aim of the present study is to analyze the effect of EGF in ulcer patients on expressions of genes related to apoptosis (Bax, Bcl-2, Fas), and show, if any, the similarities and/or differences among expression patterns in the cell line.
Twenty-six ulcer patients who applied to Gastroenterology Clinic of Mersin University Medical Faculty were included in this study. Gastroduodenal biopsies were obtained from patients undergoing upper endoscopy. Biopsy samples were taken either from the body or the antrum. The gastroduodenal biopsy samples were taken from grossly normal gastric mucosa. These endoscopic biopsy samples contained only mucosa (surface and deep glandular epithelium). The specimens were collected in Leibowitz's L-15 medium (Life Technologies) containing 1% Penicillin/streptomycin (Life Technologies) for transport of tissue biopsy sample from hospital to our laboratory.
Primary culture was formed as described by Smooth DT from the ulcerous and control biopsy specimens [10]. Biopsy specimens were enzymatically disintegrated into Leibowitz L-15 solution consisting of Type II collagenase (Biochrom 0.05% and Dispase (120U). Then, within Ham's F12 medium (10% fetal bovine serum, 1% penicillin-streptomycin, 1% Amphotericin-B), primary culture was incubated into four different 35 mm culture dishes. The culture was incubated at 37 °C in an incubator with 5% CO2.
After epithelial cells were maintained in the culture dishes, Periodic Acid Shift (PAS) staining was carried out in order to characterize stomach epithelium cells and add neutral mucin into them. PAS colors the stomach epithelial cells secreting mucin into lilac, and the nuclei of the cells are seen as light blue. Cultured stomach photographs of epithelial cells were taken both before PAS (Figure 1A) and after it. As a result of PAS, great quantities of PAS positive cells were observed (Figure 1B). To show continuous division of epithelial cells and active DNA synthesis in stomach, immunohistochemistry was done with Proliferative Cell Nuclear Antibody (PCNA) (Figure 1C).
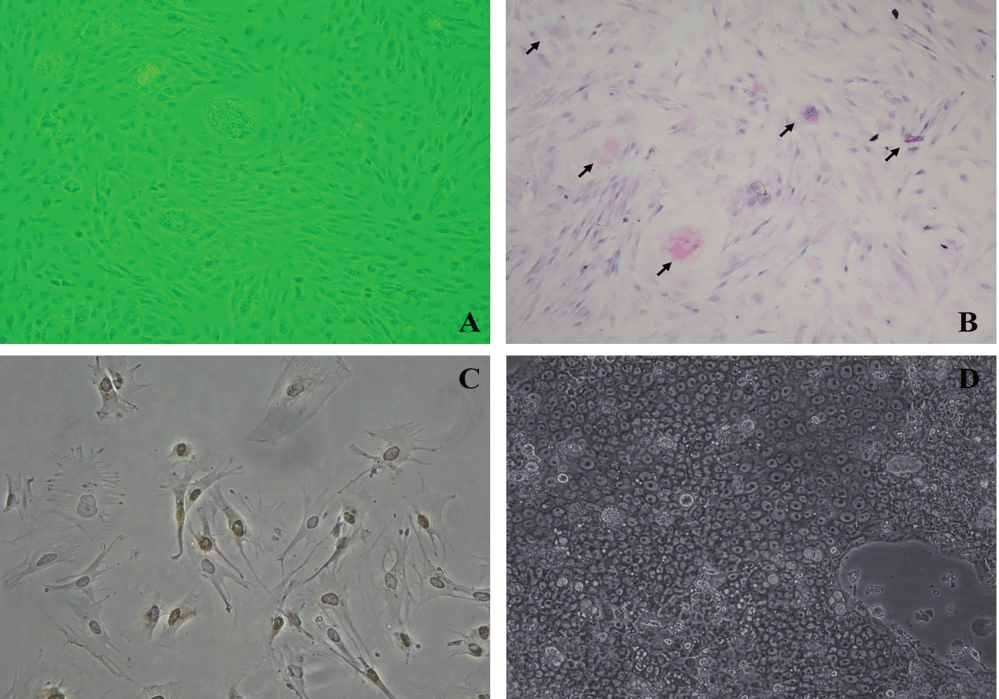 Figure 1: A) Photograph of cultured stomach epithelium cells before PAS; B) Photograph of cultured stomach epithelium cells after PAS. PAS positive cells are characterized by the lilac staining which shows neutral mucin existence. (Nikon Eclipse, TS 100); C) Photograph of active DNA synthesis is determined in stomach epithelium cells after PCNA (Olympus BX50) (100X); D) The inverted microscope (Nikon Eclipse, TS 100) appearance of gastric epithelial cells reproduced from N87 cell line.
View Figure 1
Figure 1: A) Photograph of cultured stomach epithelium cells before PAS; B) Photograph of cultured stomach epithelium cells after PAS. PAS positive cells are characterized by the lilac staining which shows neutral mucin existence. (Nikon Eclipse, TS 100); C) Photograph of active DNA synthesis is determined in stomach epithelium cells after PCNA (Olympus BX50) (100X); D) The inverted microscope (Nikon Eclipse, TS 100) appearance of gastric epithelial cells reproduced from N87 cell line.
View Figure 1
The medium in the culture dishes, which was determined to be confluent as a result of feedings at definite times and microscope observations, was removed and incomplete medium was added, and was kept in this way for 24 hours. After 24 hours, EGF (Sigma-E9644) applications were performed to dishes at three different doses (0-control; 1st dose: 20 ng/μ; 2nd dose: 50 ng/μ; 3rd dose: 100 ng/μ). At the 24th hour following the EGF application, cell count was conducted by removing the cells (Biochrome-Trypsin/EDTA/1.2 × 106 ml).
The gastric carcinoma cell line named N87 was also treated with EGF (ATCC-NCI-N87). The N87 cells resuspended with complete medium (Biochrom-RPMI-1640, 10% FBS, 1% penicillin-streptomycin, 1% Amphotercin-B) and dispensed into culture dishes. The culture was incubated at 37 °C in an incubator with 5% CO2. Flasks were followed regularly with an inverted microscope (Olymphus CK40, 40X). The confluent flasks were passaged with a dilution ratio of 1:3. The cells in the flasks were photographed (Figure 1D). EGF application was similarly held with primary culture.
The extraction of total RNA from primary cell cultures and cell line was carried out with the Acid Guanidinium Thiocyanate-Phenol-Chloroform (AGPC) method [11]. Following precipitation, RNA samples were dissolved in 50 μL DEPC water and stored at -80 °C until analysis.
Reverse transcriptase reactions contained 5 μg of extracted total RNA, 50 nM oligo dT primary, 1 × RT buffer, 0.25 mM each of dNTPs, 50 units of modified M-MuLV Reverse Transcriptase (Thermo Scientific, Vilnius, Lithuania), 25 units of RiboLock RNase inhibitor (Thermo Scientific, Vilnius, Lithuania) and nuclease-free water at a total reaction volume of 15 μl. The reaction was performed on an automated Thermal Cycler (Techne Flexigene, Cambridge, UK). RT-PCR conditions are as follows; keeping for 60 minutes at 37 °C; for 5 minutes at 95 °C; and at 4 °C.
qRT-PCR was performed with the ABI Prism 7500 Real-Time PCR System and SDS 2.0.6 Software for 7500 Real Time PCR Product (Applied Biosystems, Foster City, CA, USA). Having taken into consideration information on the Bax, Bcl-2, Fas and β-actin cDNA sequences, two pairs of gene-specific primaries were designed by Primary Express Program 3.0 (Applied Biosystems). Primaries and probes were created from NM_138761.3 Homo sapiens BCL2-associated X protein (BAX), transcript variant alpha, mRNA of Bax [12], AY220759.1 Homo sapiens B-cell CLL/Lymphoma 2 (BCL2) mRNA of Bcl-2 [13], BC012479.1 Homo sapiens TNF receptor super family, member 6 (FAS) mRNA of Fas [14] and NM_001101.3 Homo sapiens actin, beta (ACTB), mRNA of β-actin (Table 1) [15]. The sequences of primaries and probes were synthesized by Metabion International AG, D-82152 Martinsried/Deutschland.
Table 1: Primary and probe sequences used for qRT-PCR. View Table 1
Bax, Bcl-2, Fas and β-actin expressions were analyzed using the Comparative CT (ΔΔCT) Method [16-18]. The 25 μl PCR reaction included 2.5 μl of cDNA, 12.5 μl TaqMan Gene Expression Master Mix (Applied Biosystems), 900 nmol of gene-specific primaries and 200 nmol of specific labeling probes. The cycling conditions were as follow: one cycle of pre-incubation at 50 °C for 2 minutes, denaturation at 95 °C at 10 minutes, followed by 40 cycles of 95 °C for 15 second, 60 °C for 1 minute. β-actin was used as an endogenous control gene for PCR normalization concerning the amount of RNA added to the reverse transcription reactions. Moreover, the standard RNA (TaqMan Control-Human RNA) was used for CT analysis as internal control. Each real-time PCR reaction was performed in duplicate in order to assess data reproducibility.
The minimum sampling size was calculated using the MedCalc Free Trial statistical software package to be significant of differences between the mean values of the groups (alpha = 0.05 and power of test = 0.80). Expression data were assessed by Kruskal-Wallis test followed by Dunn's test. All statistical analyses were performed using the SPSS software 21 and Statica software 8.0. The P ˂ 0.05 value was defined as being statistically significant.
In this study, the comparative CT quantification (ΔΔCT method) of qRT-PCR for comparing the changes in gene expression of Bax, Bcl-2 and Fas was applied following primary culture of gastroduodenal epithelial cells with ulcer patients and gastric cancer cell line (N87). Relative quantification was performed using β-actin as endogenous control gene. β-actin exhibited the less variability among the samples as determined by the analysis of the standard deviation of the threshold cycles (CT) values (Figure 2).
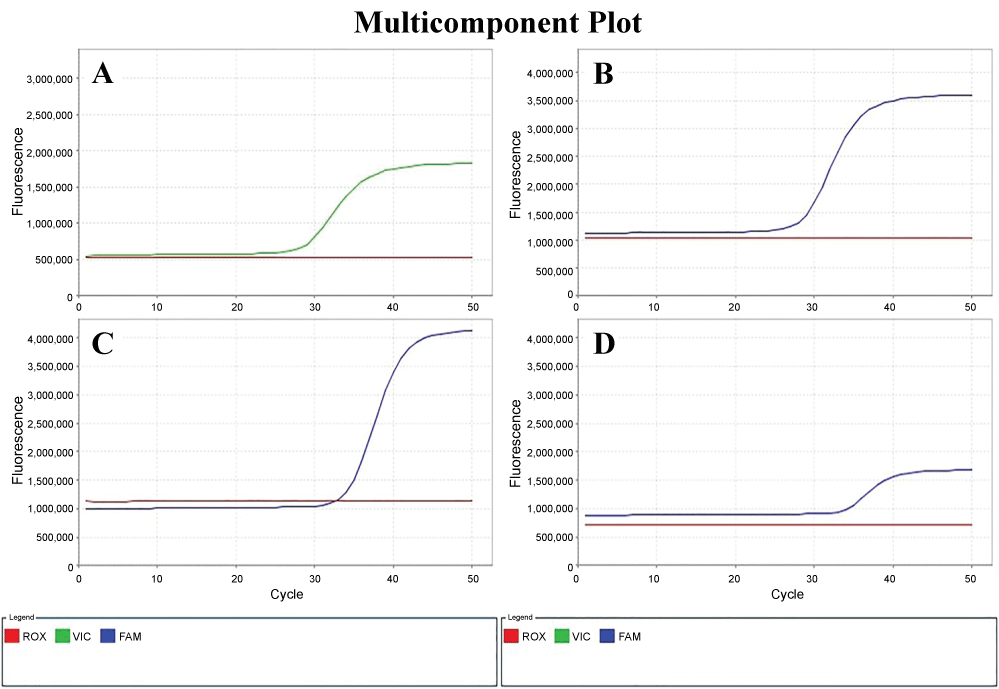 Figure 2: Amplification curves of β-actin labeling Yakima A) Yellow; B) Bax; C) Fas; D) Bcl-2 labeling FAM in the qRT-PCR.
View Figure 2
Figure 2: Amplification curves of β-actin labeling Yakima A) Yellow; B) Bax; C) Fas; D) Bcl-2 labeling FAM in the qRT-PCR.
View Figure 2
The expression level of Fas mRNA did not have a significant difference in the ulcer, gastric cancer and normal gastric tissues. However, a significant difference was identified in Fas gene expression only in tissues to which 50 ng/ml EGF was applied. Moreover, Fas gene expression level was significantly lower in gastric cancer cell line than in duodenal ulcer and normal gastric tissue samples as a result of the application of 50 ng/ml EGF in gastric cancer (p ˂ 0.05) (Table 2).
Table 2: Relationship between Fas mRNA expression levels and EGF doses in the groups. View Table 2
The results of the study revealed that there were no significant differences in Bax mRNA expression levels in the primary culture, normal gastric cells nor N87 cell lines of ulcer patients. Also, expression level of Bax mRNA and applied EGF doses were not significantly different when groups were compared for each EGF dose (Table 3).
Table 3: Relationship between Bax mRNA expression levels and EGF doses in the groups. View Table 3
The expression level of Bcl-2 mRNA did not have a significant difference in the ulcer, gastric cancer and normal gastric tissues. Significant differences were not determined between Bcl-2 gene expression levels and applied EGF doses when groups were compared for each EGF dose (Table 4).
Table 4: Relationship between Bcl-2 mRNA expression levels and EGF doses in the groups. View Table 4
A peptic ulcer is a distinct breach in the mucosal lining of the stomach (gastric ulcer) or in the first part of the small intestine (duodenal ulcer) as a result of caustic effects of acid and pepsin in the lumen.
It is the most common ulcer of the gastrointestinal tract. Various polypeptide growth factors were shown to have an important role on the mucosal cell proliferation and the regulation of its differentiation during the healing process of gastric and duodenal ulcer. One of these growths factors, EGF, was determined in the human normal gastric mucosa [19]. EGF was thought to show activities in gut lumen of EGF as a gastrointestinal growth factor [20].
Expression of genes related to apoptosis has been studied in many cancer types including gastric cancer; however there is not any published study about the effect of EGF on expressions of these genes in gastric or duodenal ulcers. It has been stated that ulceration in the gastrointestinal system induces EGF immune reactivity within the boundaries of an ulcer [21]. These studies show that the ulcer process in the human gastrointestinal system induces to development of new cells from stem cells secreting EGF. On the other hand, it has been observed that there has been no increase in the process of gastric damage in the EGF expression while raising TGF-α mRNA and protein expression in the rat gastric mucosa where acute damage is caused with hydrochloride and taurocholate [22]. These contradictions about EGF expression in the previous studies may be the results of a current antibody depending on the immunohistochemical method that is used, tissue perpetrates or the differences about the present phase of evaluated gastric ulcer.
It has been demonstrated that apoptosis is in every part of stomach and it is induced after acute mucosal damage and during the period of gastric ulcer healing. It was found that Bax and p53 expressions increased in gastric ulcer and Bcl-2 expression was also similar to the controls. In this study, the expression levels of Bax, Fas and Bcl-2 genes in gastric ulcer, duodenal ulcer and gastric cancer were evaluated in the EGF existence. The expression level of Bax, Bcl-2 and Fas mRNA was found to be similar in the ulcer, normal gastric cell and N87 cell line.
No relationship was found between Bax and Bcl-2 expression levels and EGF doses that were performed on primary cell cultures of ulcer patients and gastric cancer cell line. However, a significant difference was identified in Fas gene expression only in tissues to which 50 ng/μl EGF was applied. Also, Fas gene expression levels were significantly lower in the gastric cancer cell line than in the patients with duodenal ulcer and normal gastric tissue samples as a result of the application of 50 ng/μl EGF in gastric cancer cell line. (p ˂ 0.05)
Gastric cancer is one of the most common malignant tumors in the world. Currently, no effective treatment modalities are available for its metastasis and recurrence. Epidemiological studies have implicated that colonization of the stomach by Helicobacter pylori is a risk for the development of gastric diseases including gastric cancer [23]. Abnormal Fas expressions in gastric carcinoma are involved in carcinogenesis and metastasis of gastric cancer. Fas, a cell-surface receptor, activate the apoptosis signal pathway by binding to its ligand, FasL, resulting in cancer cell apoptosis.
By investigating the role of Fas expression in the carcinogenesis and metastasis of gastric carcinoma, several investigators have found that Fas expression in gastric carcinoma is decreased. It is shown that the Fas expression level was lower in gastric carcinoma tissue samples than in normal gastric mucosa tissue samples [24]. It was suggested that the expression of Fas protein was down regulated with the increase in the gastric carcinoma volume and the malignancy degree [25].
The Fas protein encoded by Fas gene is a member of the TNF-receptor superfamily. This receptor contains a death domain. It has been shown to play a central role in the physiological regulation of programmed cell death, and has been implicated in the pathogenesis of various malignancies and diseases of the immune system. The interaction of this receptor with its ligand allows the formation of a death-inducing signaling complex that includes Fas-associated Death Domain protein (FADD), Caspase 8, and Caspase 10. The autoproteolytic processing of the caspases triggers a downstream caspase cascade, and leads to apoptosis. Recently, Fas has also been shown to promote tumor growth, since during tumor progression, it is frequently down-regulated. Cancer cells in general, regardless of their Fas apoptosis sensitivity, depend on the constitutive activity of Fas.
On the other hand, Helicobacter pylori infection is now accepted to be linked to severe gastritis-associated diseases, including peptic ulcer and gastric cancer. The infection remains latent in the majority of infected patients, and only a minority of individuals with H. pylori infection ever develops the disease [26]. It has been reported that gastric cancer develops in approximately 3% of H. pylori-infected patients during the observational period of 10 years, compared to none of the uninfected patients [27].
EGF is known to play a role on regulating mucosal cell proliferation in the process of gastric ulcer healing. It is shown that increasing cell proliferation in the gastric mucosa infected with Helicobacter pylori may correlate with the up-regulation of EGF. In this study, since H. pylori results from patients could not be obtained, the ability to comment on the results of this topic are limited. As a result of this research, expression of Fas mRNA was found to be lower than that of ulcers. Several investigators have also found that Fas expression in gastric carcinoma has decreased. In the case of ulcer patients, each gene has its own proliferative effect in proportion to apoptotic genes and EGF concentrations, but there is no statistical difference between the expression values for each EGF concentration.
Although the incidence and mortality rate have been decreasing in recent decades, gastric cancer is still one of the most common malignancies in the world. Although H. pylori may have no carcinogenesis-promoting effect itself, infection causes inflammation of the gastric mucosa and chronic infection causes mucosal atrophy, resulting in intestinal metaplasia. Apoptosis and oxidative stress are closely interrelated and may play a determinant role in the evolution of ulcer to gastric carcinogenesis. Further progress in epidemiologic research is needed to resolve this issue. In this study, decreased expression of Fas mRNA was observed in gastric cancer while elevated Fas mRNA expression levels were determined in duodenal ulcer compared to healthy gastric epithelial cells with EGF application. The process of gastric carcinogenesis may have a relationship with abnormal expression of Fas mRNA depending on EGF in ulcer patients.
In the case of ulcer patients, each gene has its own proliferative effect in proportion to apoptotic genes and EGF concentrations, but there is no statistical difference between the expression values for each EGF concentration.
All ulcer patients gave written informed consent, and the project was reviewed and accepted by the Ethics Committee of Mersin University, Turkey.
The authors have no conflicts of interest to declare.
This work was supported by The Scientific and Technological Research Council of Turkey (TUBITAK) with 108S108 project number, cofounded by the Scientific Research Projects Unit of Mersin University.