It was noticed that Fundic Gland Polyps (FGP) are more frequently seen. There are reports suggesting that this condition is associated with the use of Proton Pump Inhibition (PPI).
Study the incidence of FGP's and relates this with the use of PPI.
All consecutive patients who underwent endoscopy in the years 2006-2015 in whom FGP's were seen were included. An extensive search was done in order to assess use of PPI's. As a control group all consecutive patients in 2012, in whom no macroscopic abnormalities were seen, were used. In addition, all consecutive endoscopies in almost 25 years, 1992-2015 were searched for the presence of FGP's. In addition, data on the use of PPI's in the Netherlands was collected.
In 270 consecutive patients FGP's were diagnosed. During upper gastrointestinal endoscopy a hiatal hernia was seen in 152 patients (56.3%), reflux oesophagitis in 35 (12.9%), and metaplastic epithelium in the distal oesophagus in 22 (8.2%). Patients with FGP's were significantly more often on PPI therapy compared with controls group, p < 0.0001. The incidence of the condition shows a clear increase in 25 years. The use of PPI's also showed a clear rise.
Incidence of FGP's is rising and probably related to the increasing use of PPI's.
The introduction of Proton Pump Inhibitors (PPI's) was an important breakthrough in the treatment of reflux disease. Shortly after the introduction it was speculated that these drugs would lead to chronic hypergastrinemia finally resulting in the development of gastric carcinoid tumors [1]. Fortunately, this assumption appeared to be incorrect.
In daily practice many patients undergo upper gastrointestinal endoscopy for many different reasons. Recently, it was noticed that Fundic Gland Polyps (FGP's) are more frequently seen. There are reports in the literature stating that this condition is associated with the long-term use of Proton Pump Inhibition (PPI) [2].
For this reason a cross-sectional study was done in all consecutive patients undergoing upper gastrointestinal endoscopy in which FGP's were seen, and relate this with the use of PPI. Also the yearly incidence of this condition was studied.
All consecutive patients who underwent upper gastrointestinal endoscopy in the years 2006-2015 in whom FGP's were seen were included in the present study. From all patients an extensive search was done in endoscopy records and clinical records in order to assess use of PPI's. Patients underwent the procedure because of all kinds of clinical reasons, but mostly reflux complaints and dyspepsia.
As a random control group all consecutive patients undergoing endoscopy in the year 2012, in whom no macroscopic abnormalities were seen, were used. Use of proton pump inhibitors in these patients was also assessed. These study years were chosen because of the availability of electronic files and records.
Since 1992 a prospective consecutive dataset on endoscopy outcomes is collected. All consecutive endoscopies in almost 25 years, 1992-2015 were searched for the presence of FGP's. The incidence of FGP's was noted as the percentage of the total number of endoscopies done each year. Only the first endoscopy, detecting FGP's, in a patient was included. Repeated endoscopies were excluded.
FGP's were detected on basis of the macroscopic appearance, if in doubt biopsy specimens were taken. If patients in the group with FGP are died, the cause of death was determined.
Data on the use of PPI's in the Netherlands was collected via the "Stichting Farmaceutische Kengetallen" [3]. Statistical analysis was done with the chi-square test for contingency tables. A value below 0.05 was judged significant.
In 270 consecutive patients (76 men, 194 women) FGP's were diagnosed. The control group, n = 465, consisted of 151 men and 314 women. During upper gastrointestinal endoscopy a hiatal hernia was seen in 152 patients (56.3%), reflux oesophagitis in 35 (12.9%), and metaplastic epithelium in the distal oesophagus in 22 (8.2%). As shown in Table 1 there was a significant difference in the use of PPI's. Patients with FGP's were significantly more often on PPI therapy compared with the patients in the control group, p < 0.0001. Even if all missing data were judged as either using PPI or not using PPI this significance did not change.
Table 1: Presence of fundic gland polyps and use of proton pump inhibitors. View Table 1
Figure 1 shows the incidence of FGP's in the period of 25 years. As can be clearly seen the incidence of the condition shows a very clear increase.
 Figure 1: The incidence of FGP's in the period of 25 years.
View Figure 1
Figure 1: The incidence of FGP's in the period of 25 years.
View Figure 1
Thirty nine patients with FGP's died. None because of gastric cancer. The cause of death was: cancer in 18 cases, cardiovascular in 4, neurological in 11, miscellaneous in 4, and unknown in 2 cases. Figure 2 shows the actual number of PPI tablets, pills or capsules distributed in the Netherlands in the period 1995-2016. These are incident and prevalent users. As can be seen the use of especially omeprazole and pantoprazole showed a clear rise in the years.
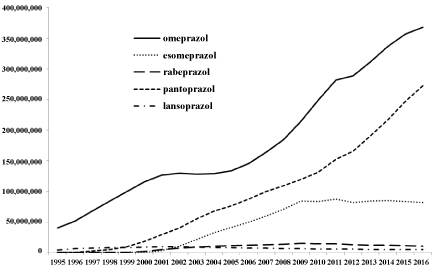 Figure 2: The actual number of PPI tablets, pills or capsules distributed in the Netherlands in the period 1995-2016.
View Figure 2
Figure 2: The actual number of PPI tablets, pills or capsules distributed in the Netherlands in the period 1995-2016.
View Figure 2
Gastric polyps can be seen during inspection of the stomach. A large retrospective study of more than 36000 consecutive endoscopies revealed gastric polypoid lesions in 2.22% of the patients. Hyperplastic polyps were the most common type of polyps (36.2%), followed by FGP's (8.3%), inflammatory fibroid polyps (2.4%) and adenomatous polyps (1.9%). Malignant transformation was seen in 0.42% of hyperplastic polyps and in 23.1% of adenomatous polyps [4]. Changes of the appearance of the gastric mucosa are described in PPI users. Elevated lesions and cobblestone-formation were often noticed in an endoscopic study [5].
In the beginning of this century it was stated that a causal pathogenetic relationship between FGP's and use of PPI was unlikely [6]. However, a recent meta-analysis revealed an increase in FGP's amongst PPI users compared to controls (OR 2.46, 95% CI 1.42-4.27, P = 0.001). Especially if the drugs were taken for a longer period of time [7].
In a study from Huang, et al. long-term use of PPI's also was a risk factor for the presence of FGP's [2]. Zelter, et al. found FGP's in 4.3% of consecutive endoscopies. And they concluded that three variables were associated with the presence of these polyps: PPI intake: P < 0.0001, OR 9.00 (95% CI 5.44-14.89); female gender: P = 0.0001, OR 2.95 (95% CI 1.69-5.15); and age: P = 0.001, OR 1.03 (95% CI 1.01-1.05) [8]. Hongo, et al. showed that FGP's developing in patient with reflux disease on long-term PPI therapy. They also showed that the development was related to high gastrin levels [9].
The best method of studying the relation of FGP's and the use of PPI's would be regular follow-up endoscopy in patients starting with PPI therapy with a group of non-users as control. Of course, such a study is not possible. Hence, the second best will be the present study.
This study also shows that patients with FGP's are significantly more often on PPI therapy compared with patients in whom no abnormalities were seen in their stomach during upper gastrointestinal endoscopy. A possible shortcoming of the study is that no exact data are present on type, dosage and duration of the PPI therapy. In addition, no data on presence of H. pylori are present.
From Figure 1 it will be clear that the incidence of the condition is rising, at least in the Zaanstreek region, in a period of 25 years. In 1992 FGP's were very rare, while in the last five years the condition is much more frequently seen. This rising incidence could be explained by the increasing use of PPI therapy, mostly because of reflux disease, but also as "stomach protection" in patients on NSAID therapy because of rheumatic disorders or patients taking double platelet aggregation inhibitors because of cardiovascular disease.
In the beginning of the introduction of PPI's it was stated that gastric carcinoid tumors would develop because of the high serum gastrin levels. A study showed that long-term use of PPI was associated with an increased risk of gastric cancer [10]. However, this final risk was very low and even uncertain [11]. In the present study none of the patients with FGP's developed gastric cancer. There was one patient with oesophageal cancer. Hence, the risk for cancer seems to be non-existing. However, an adenocarcinoma can be hidden in a FGP [12]. Also adenomas can be associated with FGP's [13].
It is concluded that the presence and/or development of FGP's is associated with the increasing use of PPI's. However, there appears to be no clinical consequence. Patients still may benefit from the therapeutic effects of PPI's.