Articular manifestations are the most common extraintestinal manifestations in inflammatory bowel diseases and are classified into axial spondyloarthritis (SpA) and peripheral SpA. EIM may occur before or after IBD diagnosis, being the proof that IBD is not only limited to gut. Here we present a case series of three patients with Crohn's disease and extraintestinal manifestations admitted to our hospital. Patients had in common young age, Crohn's disease with moderate or severe clinic and endoscopic activity, axial spondyloarthropathy and some particularities: One case associated type 1 peripheral arthritis, another case type II peripheral arthritis with enthesitis and oral aphtous ulcers and the last case associated uveitis and erythema nodosum. After failure of conventional therapy, treatment with TNF-inhibitors was initiated and remission was achieved. These case reports highlight the importance of TNF inhibitors, this therapy being currently the only effective treatment for patients with aggressive disease, associated SpA or who failed to respond to conventional therapy.
Inflammatory bowel diseases, Spondyloarthritis, Biologic therapy, Remission
Articular manifestations are the most common extraintestinal manifestations (EIM) in inflammatory bowel diseases (IBD) and are classified into axial spondyloarthritis (SpA) and peripheral SpA [1]. The prevalence of axial and peripheral involvement in IBD varies between 5%-20% and 3%-25%, respectively [2,3]. EIM may occur before or after IBD diagnosis, being the proof that IBD is not only limited to gut.
The goals of treatment for both conditions (IBD and IBD-related SpA) include reducing the inflammation and preventing any disability or deformity, and require a multidisciplinary approach between the gastroenterologist and the rheumatologist. The strategy should, from the very beginning, identify the patients at high-risk for complications or severe disease course.
The prevalence of CD and UC in 2017 in Constanţa County was 11.7/100,000 inhabitants and 6.7/100,000 inhabitants, respectively; EIM were reported in 9% of the CD cases [4]. Here we present a case series of three patients with CD and EIM admitted to our hospital consecutively, during a period of two months.
A 39-year-old man with therapeutically neglected SpA since childhood was admitted for diarrhea (> 40 watery stools/day), abdominal pain, fever, weight loss, low back pain, hips pain and ankles joint pain with swelling. Based on these clinical features, biological results, imagistic findings and histopathological confirmation, the diagnosis of CD (A2L2B1) with severe clinic activity (CDAI = 480) and severe endoscopic activity (CDEIS = 29.2, SES-CD = 38) (Figure 1) associated with EIM (axial SpA and type 1 peripheral arthritis) was established. Conventional therapy with intravenous hydrocortisone hemisuccinate (400 mg/day) and oral sulfasalazine (3 g/day) was started, but no improvement of the symptoms was obtained. Therapy with adalimumab was initiated and remission was achieved.
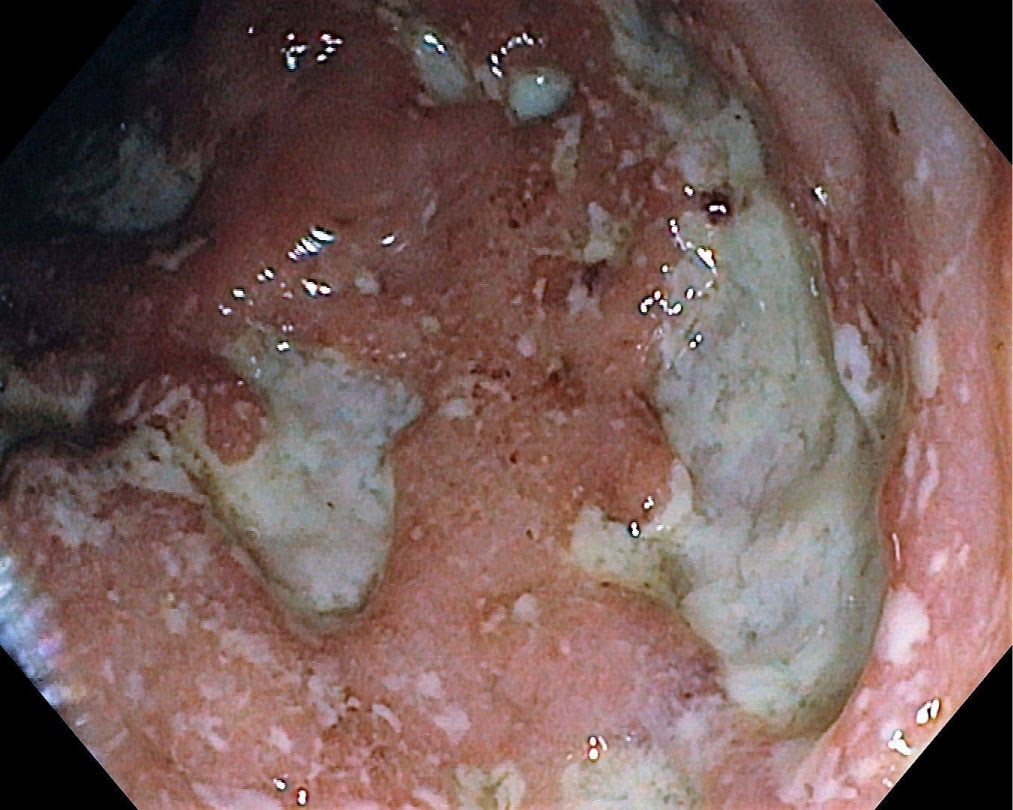 Figure 1: CD with severe endoscopic activity. View Figure 1
Figure 1: CD with severe endoscopic activity. View Figure 1
A 28-year-old female was admitted for diarrhea (15 watery stools/day), abdominal pain, fever, low back pain and joints pain (ankles, knees and elbows) with swelling and tenderness and oral aphtous ulcers. Based on clinical, biological, imagistic and histopathological findings, the diagnosis of CD (A2L2B1) with moderate-severe clinical activity (CDAI = 320) and moderate endoscopic activity (CDEIS = 11, SES-CD = 11) (Figure 2) complicated with EIM (SpA, type II peripheral arthritis with enthesitis, oral aphtous ulcers) was established. SpA was properly diagnosed by clinical features and specific imagistic criteria at MRI. Conventional therapy with intravenous hydrocortisone hemisuccinate and oral sulfasalazine was started and the therapeutic response was assessed: Improvement of the intestinal symptoms but no improvement of the inflammatory articular disease, therefore Azathioprine (AZA) (1 g/day) was added to treatment. When the withdrawal of the steroids was initiated, worsening of the joints pain was noted. AZA was replaced with Methotrexate (10 mg/week) but still no improvement of the symptoms was observed. Biologic therapy with adalimumab was initiated and remission was achieved.
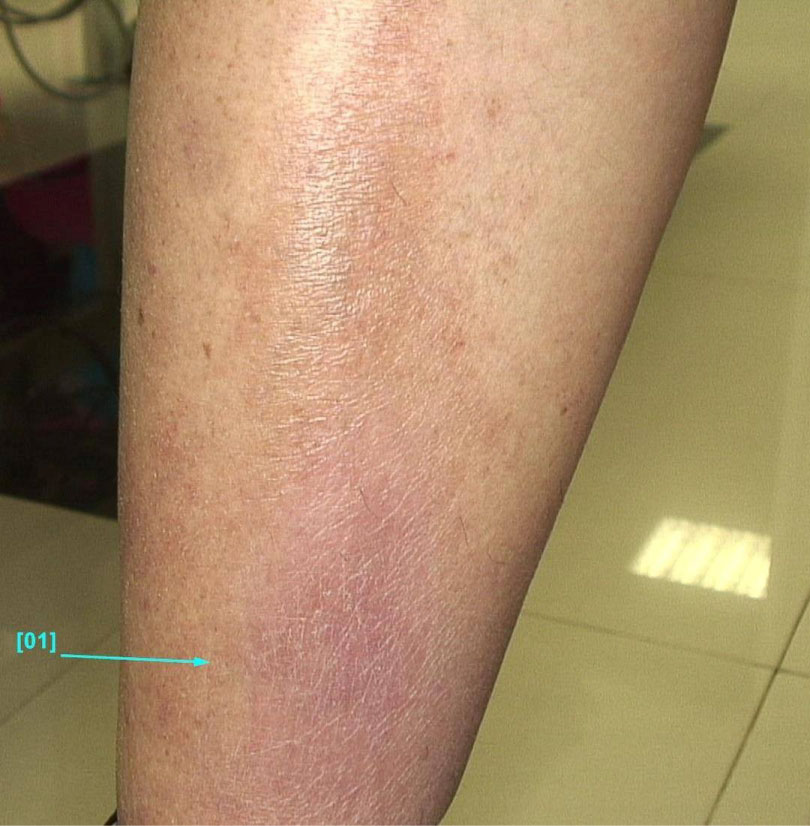 Figure 2: CD with moderate endoscopic activity. View Figure 2
Figure 2: CD with moderate endoscopic activity. View Figure 2
A 33-year-old male with a medical history of axial SpA HLA B-27 positive, uveitis and erythema nodosum since 2009, and CD since 2012 (in remission in the last years), treated with Sulfasalazine and NSAIDs, was admitted for diarrhea (7 watery stools/day), moderate abdominal pain, low back pain, and tender, violet subcutaneous nodules on the trunk and upper extremities. CDAI at admission was 190. Based on biologic and imagistic findings, the diagnosis of CD (A2L2B1) with moderate endoscopic activity (CDEIS = 10, SES-CD = 12) complicated with EIM: SpA and erythema nodosum (Figure 3) was established. Conventional therapy with intravenous hydrocortisone hemisuccinate and then oral methylprednisolone was started concomitant with NSAIDS withdrawal, but no improvement of the symptoms was observed. Therapy with adalimumab was initiated and remission was achieved.
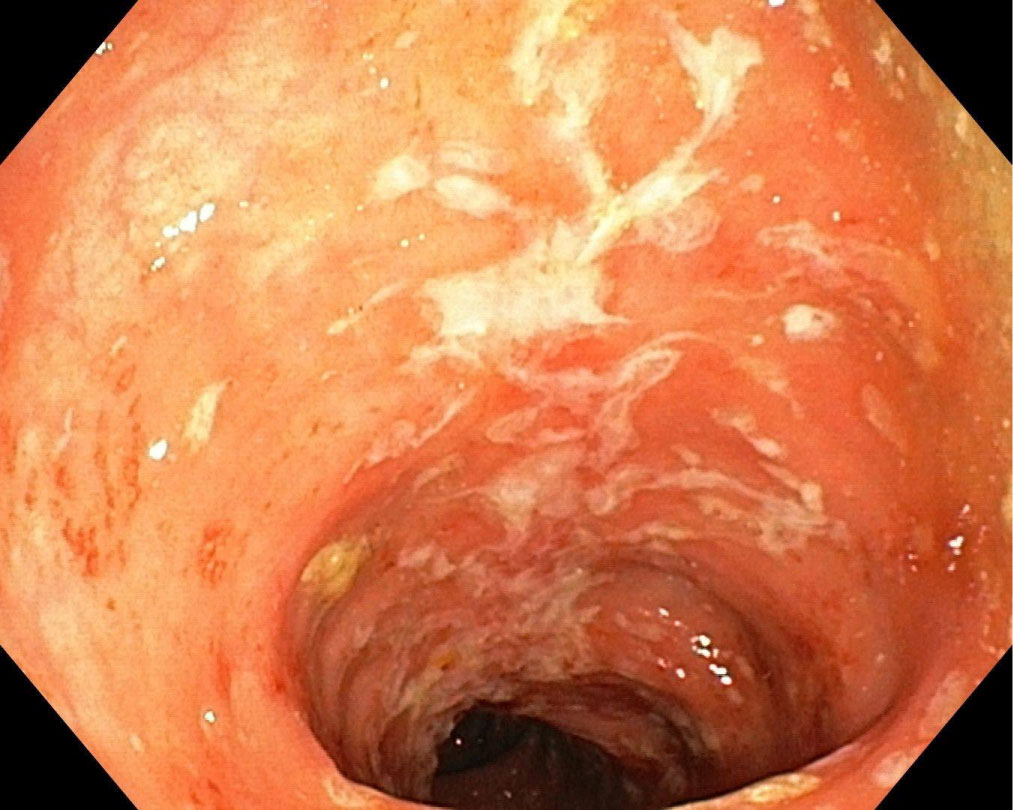 Figure 3: Erythema nodosum in a patient with CD. View Figure 3
Figure 3: Erythema nodosum in a patient with CD. View Figure 3
The association between SpA and IBD suggests a shared pathophysiology, evidences indicating the involvement of IL-23/IL-17 axis in the pathogenesis [5] and HLA B-27 association [6]. Usually, intestinal inflammation precedes or coincides with SpA, the so-called gut-joint axis hypothesis, but in some cases (as in case 1 and in case 3) SpA has been shown to precede IBD suggesting other pathopshysiological mechanisms that should be studied.
Several studies demonstrated various risk factors for a poor outcome in IBD [7-10]. All three cases had in common the following: Disease onset < 40 y, the need for early steroids, deep and extensive colonic ulcerations and the presence of EIM. So, according to literature, the cases presented above meet the criteria for "aggressive disease course" [11].
It is important to identify which factors are associated with different outcomes as this may influence important therapeutic decisions, so stratifying patients into low-risk or high-risk groups should be the first step to establish the treatment targets. Our cases suggested a more aggressive luminal symptoms in IBD patients with SpA and the need for early anti-TNF introduction. TNF inhibitors have been shown to be successful not only against IBD patients with aggressive disease course, refractory to conventional treatment or steroid-dependent but also for the SpA component. According to literature, infliximab is highly effective in moderate to severe IBD [12] and also in SpA [13,14] and ADA had the same excellent results in IBD and SpA [15,16].
Case 1 had a severe onset and evolution of CD with controlled symptoms of SpA, so intestinal inflammation was the main indication for the early aggressive management with TNF inhibitors. In contrast, in Case 2, articular disease was the main indication for biologic therapy.
According to ECCO Guideline [17], patients with axial SpA should be treated with physiotherapy, short term NSAIDs, sulfasalazine and MTX but these have limited efficacy (as we also experienced in our cases) and TNF inhibitors is the preferred treatment. Furthermore, long term NSAIDS therapy should be avoided due to their negative effects on the intestinal lesions, as in case 3 [17].
It is also important to identify high-risk patients both at diagnosis (as we proceeded in case 1 and case 2) and throughout disease course (as we proceeded in case 3). Applying the STRIDE concepts [18], clinical remission was achieved in all 3 cases after the introduction of biologic therapy.
These case reports highlight the importance of TNF inhibitors, this therapy being currently the only effective treatment for patients with aggressive disease, associated SpA or who failed to respond to conventional therapy.
None to report.
None to report.
Cristina Tocia wrote the manuscript, Victoria Cristina Șuța edited the manuscript, Eugen Dumitru and Luana Alexandrescu revised the manuscript. Informed consent was obtained for this case report.