Anti-glomerular basement membrane disease is one of the infrequently encountered nephritic disorders, but one which can lead to poor renal and patient survival. Recent reports have noted the occurrence of anti-GBM disease with atypical histological features on kidney biopsy. The presentation of these patients also follows a differing clinical course as compared to classic cases. We present a case of anti-GBM disease with atypical histological features with an entirely unique presentation among atypical cases and suggest a new paradigm for management of these patients.
Goodpasture's disease, Nephritic syndrome, Rapidly progressive glomerulonephritis, Acute kidney injury
Since the term "Goodpasture's Syndrome" was first coined by Stanton and Tange in 1958, our understanding of this disorder has significantly increased. We now know much more about the pathogenesis and treatment of this disorder. Antibodies directed towards NC1 domain of the alpha-3 chain of type IV collagen are the principal cause of anti-GBM (anti-glomerular basement membrane) disease [1]. Long-term follow up of 71 patients shows 1 year renal survival of 95% in those who present with a serum creatinine of less than 5.7 mg/dL. Renal survival is poor among those who need dialysis on presentation. The chance of renal survival in this group at 1 year is 8% [2]. Recently, a subset of anti-GBM patients have been noted to have atypical histological findings [3]. These patients also follow a more indolent course. The patient described below presents with anti-GBM, atypical histological findings as well as pulmonary hemorrhage - a feature not previously described in atypical anti-GBM disease.
We report a case of atypical anti-GBM disease. A 57-year-old Caucasian woman, with no history of diabetes mellitus and no prior renal disease, presented with 5 days of dyspnea and 1 day of hemoptysis. Chest x-ray revealed diffuse bilateral opacities. Serum creatinine increased from 0.9 mg/dl on admission to 2.7 mg/dl over seven days. Urine sediment showed dysmorphic RBCs. Serologies were negative for Anti-Neutrophil Antibody (ANA) and anti-neutrophil cytoplasmic antibody (ANCA). Complement levels were normal. Anti-GBM serology was positive at 31 units/ml (reference range 0-20 units/ml). Renal biopsy contained 20 glomeruli, 7 of which were globally sclerotic. Light microscopy showed acute tubular injury and was notable for nodular glomerulosclerosis (figure 1). Similar renal biopsy findings have been described previously in 3 patients [4]. Toluidine blue stained section for electron microscopy showed 2 glomeruli with segmental cellular crescent and focal fibrin out of the 5 examined glomeruli. No crescents were identified on other light microscopy stains. Immunofluorescence revealed diffuse linear 3+ staining for IgG, kappa and lambda along the glomerular basement membrane (Figure 2). There was trace segmental linear staining for C3 in occasional glomeruli. IgG subtype staining showed 3+ diffuse linear staining for IgG1 and IgG4, 1+ for IgG2 and negative for IgG3. No mesangial deposits were seen on immunofluorescence. Electron microscopy showed no deposits (Figure 3). Congo red stain for amyloid was negative. Treatment with steroids, cyclophosphamide and plasmapheresis (PLEX), resulted in resolution of hemoptysis and improvement in serum creatinine to 1.8 mg/dl.
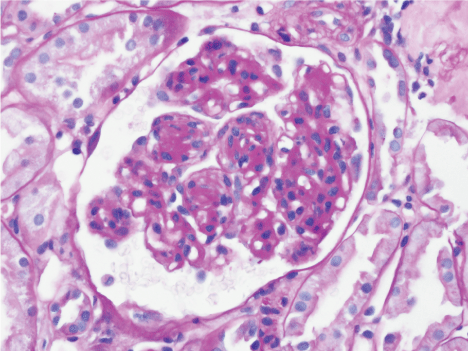 Figure 1: Glomerulus showing nodular expansion of the mesangial matrix (PAS stain, 400X). View Figure 1
Figure 1: Glomerulus showing nodular expansion of the mesangial matrix (PAS stain, 400X). View Figure 1
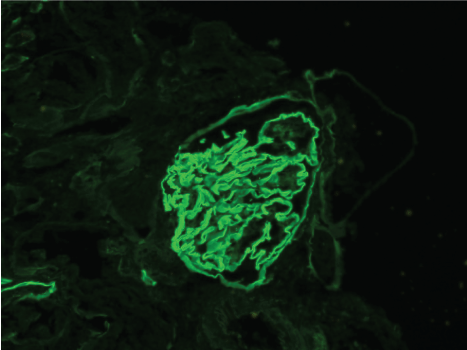 Figure 2: Glomerulus with linear IgG staining along the glomerular basement membrane (Immunofuorescence, 400X). View Figure 2
Figure 2: Glomerulus with linear IgG staining along the glomerular basement membrane (Immunofuorescence, 400X). View Figure 2
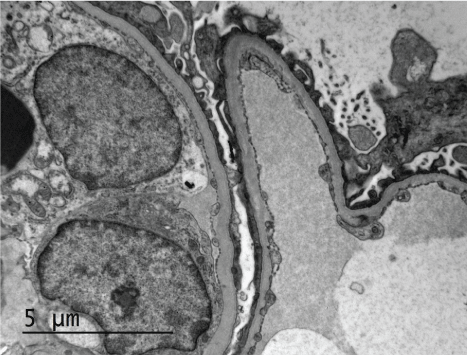 Figure 3: Glomerular basement membrane with no electron dense deposits on ultrastructural examination. View Figure 3
Figure 3: Glomerular basement membrane with no electron dense deposits on ultrastructural examination. View Figure 3
Although infrequently encountered, these atypical anti-GBM cases may not be as rare as previously thought. A recent publication examined the clinical course of 20 anti-GBM patients with atypical histological findings [3]. These patients notably displayed a more indolent course. They presented without pulmonary hemorrhage with lower serum creatinine, but higher levels of proteinuria as compared to classic presentations of anti-GBM disease. Among the reported atypical cases with nodular glomerulosclerosis [4], ours is the only reported case with both positive anti-GBM antibody serology as well as response to standard therapy for anti-GBM disease.
Atypical anti-GBM glomerulonephritis simply implies that the morphology seen on the biopsy is not typical. It may not mean that the disease is not severe and it may require more than conservative treatment. The patients in the aforementioned case series displayed negative anti-GBM serology, a finding that may sway clinicians to follow a conservative treatment strategy. Discordant results between serology and glomerular morphology are frequently encountered in other glomerular disorders. For example, serology is negative for ANCA and M-type Phospholipase A2 Receptor (PLA2R) antibodies in approximately 20% of respective cases of suspected ANCA-related renal disease and primary membranous nephropathy [5,6], however patients are treated on the basis of clinical symptoms rather than serology alone. We suggest the same management approach for atypical anti-GBM disease by treating as the closest clinical picture. Those that present with minimal clinical features could be managed by close follow ups or minimal immunosuppression, while those with nephrotic range proteinuria are treated like membranous nephropathy. Those with crescents and rapidly advancing disease, like the typical anti-GBM glomerulonephritis, could be treated with PLEX and aggressive immunosuppression.
We conclude that much more research is needed to be able to design a universally acceptable and appropriate therapy for these atypical cases.