Acute Coronary Syndromes include a wide variety of clinical scenarios. Coronary Angiography (CA) is a low-resolution imaging technique and its inability to provide a satisfactory tissue characterization of culprit lesions could be a concern in some cases. Optical Coherence Tomography (OCT) is an emergent intravascular high-resolution imaging modality which could be complementary to CA in challenging cases. We report a case of a young man in whom OCT had been determinant to understand the pathology of culprit lesion and to formulate a tailored therapy.
We report a case of a 28-years-old man, presenting to the emergency department and complaining of acute chest pain lasting about 30 minutes. Cardiovascular risk factors were cigarette smoking and familial history of Coronary Artery Disease. At admission both EKG and echocardiogram were unremarkable; a mild positivization of high sensitive troponine T was detected. The next day CA was performed showing a "minus" image in the proximal Left Anterior Descendent Artery (LAD), highly suspicious for a pedunculated, non-stenosing, adherent wall thrombus which prompted adequate antithrombotic and anticoagulant therapy. At control angiography we performed an OCT study which showed a thin-capped eccentric vulnerable plaque. We therefore decided to implant a bioresorbable scaffold on the proximal LAD. Subsequent thrombophilia screening showed severe hyperhomocysteinemia and homozygous C677T Methylenetetrahydrofolate Reductase mutation. The patient was discharged home 2 days later on aspirin, ticagrelor, folic acid and vitamins B6 and B12. At 6-month follow-up, a new OCT image acquisition showed satisfactory apposition of the bioresorbable scaffold.
OCT is a promising high-resolution intravascular imaging modality able to provide a comprehensive tissue characterization of unstable coronary plaques. OCT should be used as a complementary technique in challenging clinical scenarios.
Acute Coronary Syndromes, Optical Coherence Tomography, Atherosclerosis
ACS: Acute Coronary Syndrome; CA: Coronary Angiography; CAD: Coronary Artery Disease; ICU: Intensive Care Unit; IVUS: Intravascular Ultrasound; LAD: Left Anterior Descending Artery; LVEF: Left Ventricular Ejection Fraction; OCT: Optic Coherence Tomography; IVUS: Intravascular Ultrasound; CA: Coronary Angiography; MTHFR: Methylenetetrahydrofolate Reductase
Acute Coronary Syndromes (ACS) include a wide variety of clinical scenarios. Coronary Angiography (CA) is the most used imaging technique in this setting; however CA entails some limitations that could be of concern in particularly challenging cases; indeed CA has limited ability to provide a satisfactory tissue characterization of culprit lesions, beyond the detection of calcium and grossly ulcerated plaques or dissections. New high-resolution imaging techniques have been developed, such as Optical Coherence Tomography (OCT) and Intravascular Ultrasound (IVUS). OCT is an intravascular imaging modality with 10-fold higher resolution than IVUS which could offer further insights in those patients in whom CA meets its limitations; of note tissue characterization of culprit lesions in ACS, guidance of intracoronary interventions as well as evaluation of post implantation stent apposition are amongst the main fields of application of OCT [1]. We report a case of a young man in whom OCT had been determinant in understanding the characteristics of the culprit lesion and in formulating a tailored therapy.
The patient is a 28-year-old man. He was a heavy cigarette smoker (about 50 cigarettes per day) and had familial history of Coronary Artery Disease (CAD). He performed regular aerobic physical activity and had a balanced diet.
On the day of admission, he had experienced intense, oppressive chest pain, radiating to the left harm, lasting about 30 minutes. At arrival at the emergency room he was asymptomatic; no dynamic ST-segment modification was evident on seriated EKGs. A mild positivization of highly sensitive troponine T was detected after serial testing (maximum value 63.8 pg/ml, normal range < 14 pg/ml). At admission to Intensive Care Unit (ICU) resting heart rate was 101 bpm and blood pressure was 130/85 mmHg. Patient's body mass index was 24.7 kg/m2. Thoracic and heart auscultation were unremarkable, no signs of pulmonary or systemic congestion were detected; EKG pattern was within normal range (Figure 1). Trans-thoracic echocardiography showed no signs of segmental wall motion abnormalities nor signs of global left ventricular systolic dysfunction (left ventricular ejection fraction, LVEF = 65%), normal dimensions of cardiac chambers and aorta, no pulmonary hypertension. Blood test findings were within normal range except for a mild increase in high sensitive troponine T and low HDL-cholesterol levels (0.87 mmol/L); LDL-cholesterol was 2.69 mmol/L. A diagnosis of ACS was made; the patient was treated with acetylsalicylic acid(500 mg iv), enoxaparin, beta-blockers, high-dose statin and a CA was scheduled for the next day.
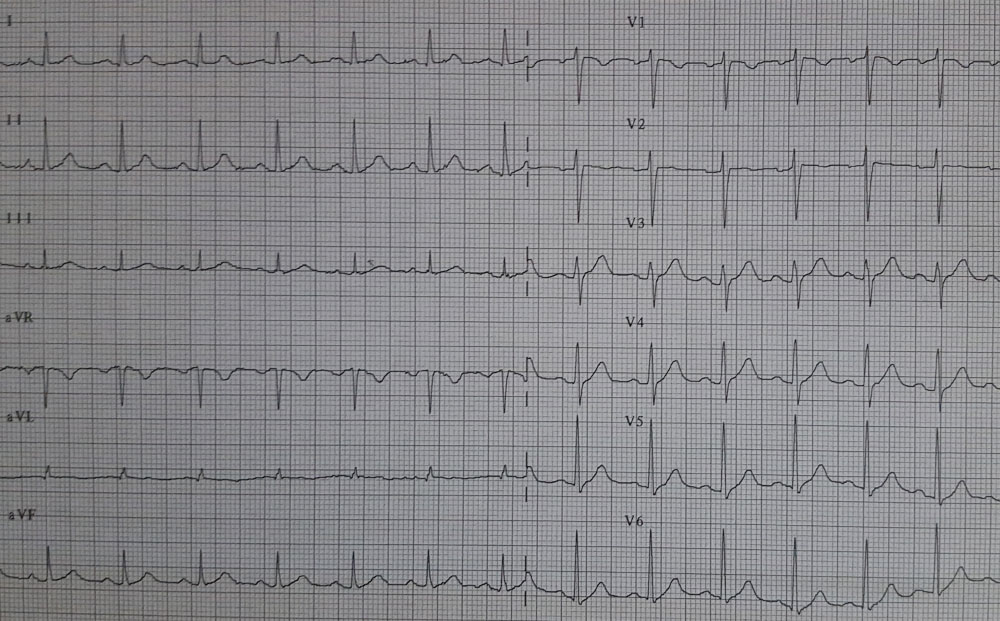 Figure 1: 12-lead EKG at admission.
View Figure 1
Figure 1: 12-lead EKG at admission.
View Figure 1
CA showed left dominant coronary circulation, no abnormalities on the left main and circumflex artery and an hypoplasic right coronary artery; a "minus" image was noticed in the proximal segment of the Left Anterior Descending artery (LAD), resembling a mobile and pedunculated adherent wall thrombus, with dimensions of 3.0 × 2.0 millimeters (Figure 2A). Intracoronary bolus followed by intravenous infusion of abciximab were given, followed by intravenous infusion of sodium heparin; angiographic control after 24 hours showed no significant changes of the "minus" image in the LAD; therefore we decided to perform OCT using a Dragonfly (St. Jude Medical) on the LAD. OCT highlighted a thin-capped, eccentric vulnerable plaque (Figure 2B). A 3.5 × 18 mm bioresorbable scaffold was implanted with satisfactory angiographic result and regular post-procedural course (LAD (Figure 2C and Figure 2D). Ticagrelor was added on therapy. Thrombophilia screening was subsequently performed, showing severe hyperhomocysteinemia (> 50 mmol/l, semiquantitative assay) and homozygous C677T methylenetetrahydrofolate reductase (MTHFR) mutation, which prompted initiation of folic acid supplementation. Afterwards the patient remained asymptomatic and was discharged two days later. At 6-month follow-up, we performed a new OCT image acquisition which showed satisfactory apposition of the bioresorbable scaffold (Figure 3).
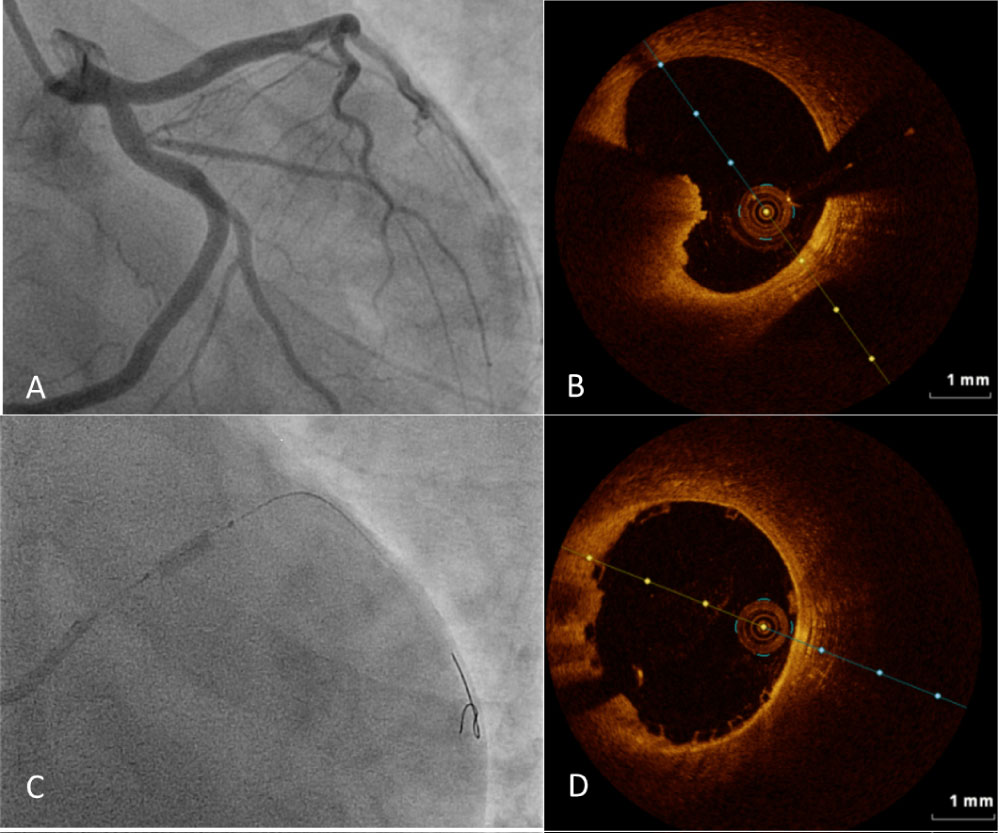 Figure 2: Panel A, Evidence of angiographic "minus" image on LAD; Panel B, First OCT study of LAD; Panel C, bioresorbable scaffold implantation; Panel D, OCT control post bioresorbable scaffold implantation. View Figure 2
Figure 2: Panel A, Evidence of angiographic "minus" image on LAD; Panel B, First OCT study of LAD; Panel C, bioresorbable scaffold implantation; Panel D, OCT control post bioresorbable scaffold implantation. View Figure 2
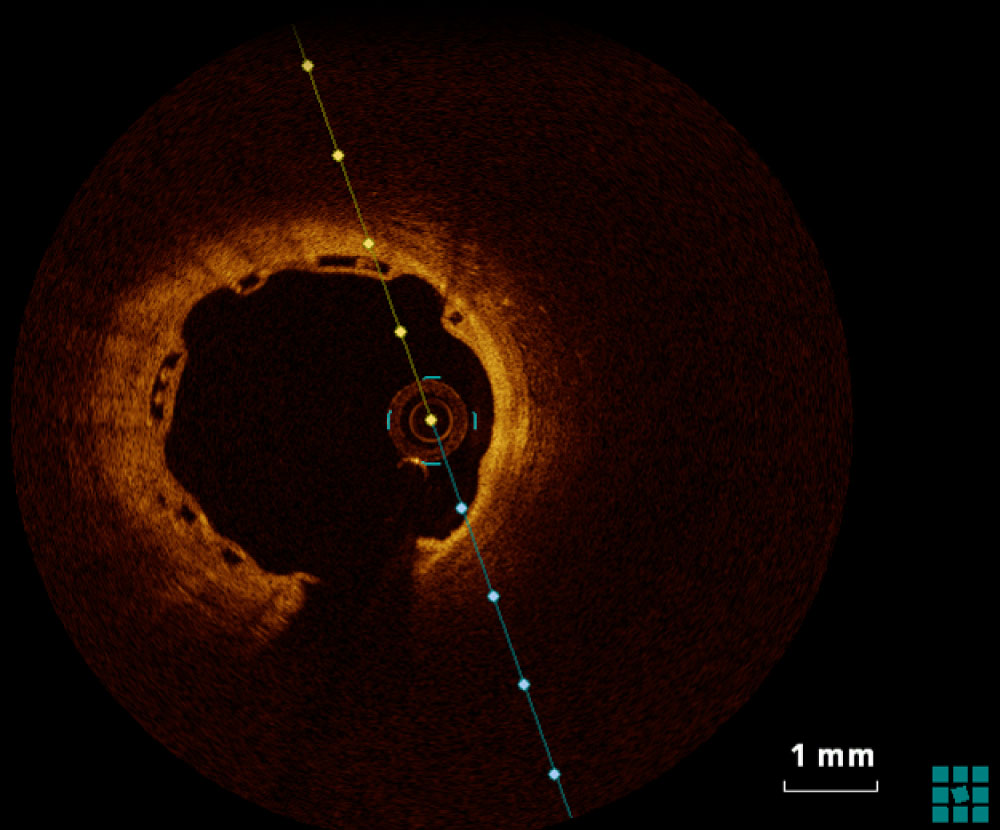 Figure 3: 6-month OCT control of bioresorbable scaffold apposition.
View Figure 3
Figure 3: 6-month OCT control of bioresorbable scaffold apposition.
View Figure 3
Destabilization of a vulnerable atherosclerotic coronary plaque is the main cause of ACS; CA is the most commonly employed imaging technique, used both for diagnostic and interventional coronary procedures; however, the main drawback of CA is represented by its limited ability to characterize tissue and atherosclerotic plaques, especially in some conditions such as overlapping vessels, foreshortening, or calcium in the vessel wall. Nowadays, new high-resolution intravascular imaging techniques are available to help interventional cardiologists, such as IVUS and OCT. While IVUS technique is based on ultrasounds, OCT uses near-infrared light which allows to obtain images with 10-fold higher resolution (10 to 20 µm) than IVUS, at the expense of lower tissue penetration (1 to 3 mm vs. 4 to 8 mm); OCT can discriminate the three layers of the coronary artery wall and provides a better tissue characterization, defining the type of coronary plaque (fibrous, fibrocalcific and lipid-rich) [2,3]; These features make OCT the technique of choice (over CA and IVUS) to assess plaque vulnerability.
Main clinical applications of OCT [1] are:
- Diagnostic assessment of coronary atherosclerosis: particularly in the context of ACS, OCT helps to identify and characterize culprit lesions (plaque rupture, intracoronary thrombus, erosion).
- OCT-guided coronary intervention: OCT is especially valuable in assessing the interface between vessel lumen and stent's struts, helping in early identification of malapposition and coronary intervention-related complications.
We described a challenging case of a young man with ACS in which the sole evaluation by CA would have been inadequate to characterize patient's culprit lesion. Both CA examinations showed a "minus" image highly suspect for intracoronary thrombus with no significant changes despite adequate antithrombotic treatment. OCT study on the LAD allowed individuation of the presence of a thin-capped, eccentric vulnerable plaque, so that we had enough anatomic data to set an interventional therapeutic strategy (bioresorbable scaffold implantation). We preferred OCT over IVUS for the its added value in studying vulnerable plaques; in consideration of the young age of the patient, a bioresorbable scaffold was preferred over conventional stents. ACS in young patients occurs rarely; nevertheless, it represents a remarkable concern because of the years of potential life lost; Schoenenberger, et al. [4] described an incidence of 0.7% of ACS in patients < 35-years-old. Risk profile of young patients is slightly different with higher prevalence of some risk factors such as cigarette smoking, dyslipidemia, familial history of CAD, obesity [5]. Our patient was a heavy smoker, had familial history of CAD and low HDL-cholesterol levels. Moreover, we detected severe hyperhomocysteinemia and homozygous C677T MTHFR mutation. MTHFR C677T mutation is the most common mutation responsible for hyperhomocysteinemia; homozygous MTHFR C677T mutation together with significant hyperhomocysteinemia seems to increase the risk of arterial and venous thromboembolism [6]. As showed by several reports elevated levels of homocysteine suggest an increased risk for cardiovascular events, including coronary artery disease and stroke [7-9]. However, it is not conclusive whether homocystein itself increases the risk of cardiovascular disease and there is no definitive study to date demonstrating hyperhomocysteinemia as an independent risk factor for cardiovascular disease. Mild elevations in homocysteinemia can be due to aging and lifestyle [10] (smoking, sedentary lifestyle, dietary factors like alcohol use, coffee or tea consumption, decreased intake of folic acid and vitamins B6 and B12). It is not clear whether lowering homocysteine levels actually decreases the risk for atherosclerosis and thrombosis but, while awaiting definitive studies, many clinicians recommend treating elevated homocysteine with folic acid and vitamins B6 and B12 supplementation. Following data published in literature data we decided to treat our patient with folic acid and vitamins B6 and B12 supplementation.
ACS includes a wide variety of clinical and anatomical scenarios. CA and IVUS could be inadequate to characterize the physiopathology of coronary culprit lesions in selected cases. OCT is a promising high-resolution intravascular imaging modality able to provide a comprehensive tissue characterization of unstable coronary plaques. OCT should be used as a complementary technique in challenging clinical scenarios.
The authors declare that they have no competing interests.
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor of this journal.
GV, SV, PP and CLV made the conception and design of the paper, drafted the manuscript, revised it critically for important intellectual content.
None declared.