Recent virological studies have shown the association between digestive diseases and some enteric viruses: adenovirus, rotavirus, enterovirus, .... Our objectives were to show the impact of viral infections of the digestive complications and interactions between enteric viruses and digestive tract cells.
During the period 2010-2011, the tissue samples preserved in paraffin obtained after reduction surgery in children hospitalized for digestive diseases (appendicitis, IIA, BA, peritonitis) in the pediatric surgery department of the CHU Fatouma Bourguiba, Monastir, Tunisia. Enteric viruses were detected by PCR of rotavirus, genotyping by RT-semi-nested multiplex PCR.
During the study period, 64 samples were collected from children with gastrointestinal surgical pathologies. The AdV were detected in 9 cases (14%) and rotavirus in 1 case (1.5%). A male predominance was observed, with a sex ratio of 1.9. The age ranges from 1 day to 8 years. The child found positive in rotavirus was among have 5-months-old and had an array of ileo-IIA Cecile. Children detected positive in adenovirus were aged 2 months to 7 years. All subjects had surgical treatment with systematic appendectomy.
To our knowledge this is the first study carried out in our country etiology. About viral etiology of these diseases in our study, adenovirus was present in 13% of children and involved in 20% of children with acute appendicitis, rotavirus was found in one child invaginated.
Enteric viruses, Intussusception, Association
Surgical digestive diseases are common, especially in developing countries. They are responsible for many deaths and undertake a significant portion of hospital costs [1]. Among these diseases, the intussusception (IIA), biliary atresia (BA) and appendicitis are considered among the more frequent pediatric abdominal emergencies.
IIA is defined by the penetration of a segment of bowel downstream. This le ads to intestinal obstruction with pain, vomiting, and possibly transit stop and peritonitis. Classified into two categories: idiopathic infant IIA and IIA secondary to a local cause. The BA is characterized by a biliary obstruction, occurring in the perinatal period. Two kinds of BA are distinguished: The BA syndromic congenital BA and nonsyndromic BA.
Appendicitis is an inflammation of the appendix located on the cecum [2]. It is the most common abdominal emergency in surgical practice [3]. It could be complicated in some cases to an abscess, peritonitis or systemic infection [4].
Recent virological studies have shown the association between gastrointestinal diseases and some enteric viruses: adenovirus (AdV) and rotavirus (RV) [5,6].
The AdV humans have been associated with a variety of diseases (gastrointestinal infections, respiratory and eye) that are complicated in some cases IIA.
A rotavirus vaccine, Rotashield®, licensed in the United States, was withdrawn from the market after less than a year of use because of its association with intussusception [7]. Group C and Group A RV were detected in patients with BA.
The aims of our study were the optimization of the molecular techniques for detection of enteric virus genomes in paraffin fixed tissue; establishment of the impact of viral infection on gastrointestinal complications and the interaction between enteric viruses and digestive tract cells.
We performed a retrospective review of pathology records during the period 2010-2011, to search for intestinal resections that had been performed to treat intussusceptions in children. The tissue samples preserved in paraffin fixed tissue obtained after resection in children who's aged from 1 day to 10 years hospitalized for digestive diseases (appendicitis, IIA, BA, peritonitis) in the pediatric surgery department of the CHU Fatouma Bourguiba. Rotavirus was detected using RT-PCR and genotyped by RT-semi-nested multiplex PCR. For adenovirus, we used a PCR.
For each tissue biopsy, we performed 5-10 thin sections 10 microns thick. These cuts were made using a microtome under sterile conditions. The deparaffination step was performed by washing twice with xylene (57 ℃ for 5 min each). After each wash, centrifugation 12.000 × g for 10 min was performed to remove all traces of wax.
Deparaffinized tissue was placed in a tube containing 500 µl of the cell lysis buffer supplemented with proteinase K (20 mg/ml). Then the reaction mixture was incubated at 52 ℃ overnight until complete dissolution of tissues [8].
Viral DNA was extracted using the ZR Viral DNA Kit™ (Zymo research) according to the manufactures protocol.
PCR was performed based on the protocol described by Allard, et al. [9]. PCR was carried out using: positive control sample (stool) and a negative control containing the pre-reaction mixture without DNA. The revelation of the amplification products was conducted by electrophoretic migration on 1% agarose gel of 10 µl of sample, at 100V for 30 to 50 min in buffer Tris-Borate EDTA 0.5 x (0.5 x TBE). A molecular size marker is deposited (100 bp DNA Ladder (800 µg/ml), Promega). The bands were visualized by staining with 0.30 µg of ethidium bromide per ml and inspection under UV light.
Viral RNA was extracted using TRI-Reagent® (Sigma). The extraction begins by the addition of 750 µl of Tri-Reagent® according to the manufactures protocol.
RV detection was made by amplifying the gene encoding the VP6 protein by RT-PCR regarding protocol described by Gomara, et al. [10]. To 5 µl of dsRNA extract, 1 µl of each primer sense and antisense VP6F/VP6R (10 microM) was added with 3 µl of DEPC water. The mixture is placed in a thermal cycler at 94 ℃ for 5 min, then immediately cooled in ice to keep the two strands separate primer annealing during reverse transcription.
The reverse transcription reaction was carried out for 45 min at 42 ℃. For each Eppendorf tube we added 40 µl of a reaction mixture comprising: 18.95 µl of DEPC water, 10 µl of the reverse transcriptase buffer (5x), 2 µl of dNTP mix (10 mM), 1 µl of DTT (100 mM), 8 µl of MgCl2 (25 mM), and 0.05 µl (10U) of Moloney Murine Leukemia Virus Reverse Transcriptase (M-MLV RT) (200 U/microl, Promega, USA), added to the last minute. The concentration of MgCl2 chosen after optimization is 0.4 mM.
PCR was performed in the same Eppendorf tube than the RT, by adding to each tube 50 µl of a second reaction mixture containing 26.1 µl of sterile distilled water, 20 µl of buffer for Taq polymerase (10x), 2 µl of dNTP (10 mM), 1.6 µl of MgCl2 (25 mM) and 0.3 µl (1.5 U) of Taq polymerase (5 U/microl, Promega, USA). All tubes were placed in the thermal cycler for 30 cycles of polymerization preceded by an initial denaturation step which lasts 5 minutes at 94 ℃, and a final elongation step lasts 7 min at 72 ℃.
Molecular VP4 genotyping was carried out by RT-PCR semi-nested multiplex. We followed the protocol described by Gentsch, et al. [11].
To 5 µl of extracted dsRNA were added 1 µl (10 microM) of each external primer VP4F/VP4R and 3 µl of DEPC water. The mixture was placed in the thermal cycler at 97 ℃ for 5 min and then immediately cooled in ice to keep the two strands separate primer annealing at the retro-transcription.
. The reverse transcription was conducted at 42 ℃ for 45 min. To each Eppendorf tube we added 40 µl of a reaction mixture comprising: 24 µl of DEPC, 2 µl of dNTP mix (10 mM), 5 µl of reverse transcriptase buffer (10x), 1 µl of DII (100 mM), 8 µl of MgCl2 (25 mM), and 0.05 µl (50U) of M-MLV RI. All these steps must be realized in cold ice.
PCR was performed in the same Eppendorf tube than the reverse transcription, by adding to each tube 50 µl of a reaction mixture containing: 25.7 µl of sterile distilled water, 20 µl of buffer TAQ (10x), 2 µl of DNIP (10 mM), 2 µl of MgCl2 (25 mM), 1 µl of each internal primer (10 microM) and 0.3 µl of TAQ polymerase (5U/microl, Promega, USA). All tubes were placed in the thermocycler. The PCR cycle was the same as that previously used in the amplification of VP6.
In a sterile Eppendorf tube, an appropriate amount of cDNA ranging between 0.5 and 0.4 µl was added, according to the intensity of the observed band since the first amplification with 50 µl of a reaction mixture comprising: 28.2 µl of sterile distilled water, 2 µl of MgCl2 (25 mM), 20 µl of buffer TAQ, 2 µl of DNIP (10 mM), 1 µl of each internal primer (10 microM) and 0.3 µl of TAQ Polymerase (5U/µl, Promega, USA).
Molecular genotyping of RV VP7 was performed by RT- POR semi-nested multiplex. We following the protocol described by Gouvea, et al. [11,12]. This protocol is the same as that used for the VP4 genotyping, using primers specific for VP7 genotyping.
The revelation of the amplification products was conducted by electrophoretic migration on 2% agarose gel of 10 µl of sample, at 100V for 30 to 50 min in buffer Tris-Borate EDTA 0.5 x (0.5 x TBE). A molecular size marker is deposited (100 bp DNA Ladder (800 µg/ml), Promega). The bands were visualized by staining with 0.30 µg of ethidium bromide per ml and inspection under UV light.
The demographic and epidemiological data were collected using a descriptive card (age, gender, reasons for hospitalization ...).
A total of 64 cases of children with gastrointestinal surgical pathologies, a male predominance was observed, with a sex ratio of 1.9. For the population of patients infected with the virus (AdV and RV), the sex ratio is 1.5 (n = 10). The children range in age from 1 day to 8 years, with an average age of 3 years. The child was found positive RV 5 months old. Children detected positive AdV were aged 2 months to 7 years. Among subjects who were positive for AdV in molecular diagnosis, 67% were hospitalized for appendicitis, peritonitis, 22% and 11% for IIA ileocolic. The patient infected with RV had an array of ileo-IIA Cecile. All subjects had surgical treatment with systematic appendectomy (Table 1).
Table 1: Epidemiology and characteristics of samples detected positive for adenovirus or rotavirus. View Table 1
The AdV were detected in 9 cases (14%) of sixty-four samples studied. For the 7 samples showed positive bands of low intensity was used to extraction kit ZR Viral DNA Kit TM to improve the intensity of the band (Figure 1).
 Figure 1: Products of "hex" gene amplification of adenovirus before and after optimization of the extraction revelation with 2% agarose gel.
Figure 1: Products of "hex" gene amplification of adenovirus before and after optimization of the extraction revelation with 2% agarose gel.
Well 1: molecular size marker (100 bp ladder); Wells 2-3: 533/533*; Wells 4-5: 892/892*; Wells 6-7: 2321/2321*; Wells 8-9: 2196/2196*; Well 10-11: 5386/5386*; Wells 12-13: 5960/5960*; Well 14-15: 5150/5150*; Wells 16: negative control.
*Sample extracted Kit.
View Figure 1
We have found 1 positive sample (1.5%) of the 64 samples tested by RT-PCR gene VP6. The sample detected positive is shown in Figure 2.
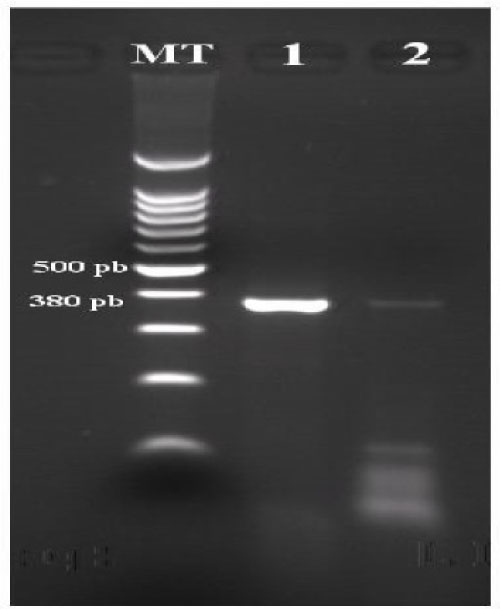 Figure 2: Products of the amplification of gene 6 of rotavirus positive sample detected, revealing agarose gel 2%.
Figure 2: Products of the amplification of gene 6 of rotavirus positive sample detected, revealing agarose gel 2%.
MT: molecular size marker; Well 1: positive control RV; Well 2: positive sample RV.
View Figure 2
The first amplification did not give the band 633 bp expected. The second amplification gave a band of 147 bp thus strain RV genotype P [6].
In young children, the stomach and spleen have not reached their full energy, which explains the frequency of digestive disorders in the young. Suffice it then error dietetics, an emotion or an infection to cause vomiting, diarrhea, and abdominal pain [13]. These symptoms are sometimes the result of digestive disorders which may be complicated in some cases. Among digestive surgical pathologies affecting the child, appendicitis, bowel obstruction, AVB and IIA.
IIA is the penetration of the intestinal segment in the segment below by a reversing mechanism of a glove finger [14]. There are two types IIA: IIA primary or idiopathic form of the infant, which is the most common, and secondary IIA, whose starting point, is a local cause organic [15]. The association between these diseases and certain viral infections (AdV infection, RV infection) has been reported several times [5,6].
Our work consisted in the study of digestive surgical pathologies occurred in children hospitalized in the pediatric surgery department of the CHU Fatouma Bourguiba. In our study, we adopted the techniques of molecular biology research enteric viruses (AdV, RV) may be involved in the digestive surgical pathologies. The samples were in the form of intestinal tissue preserved in paraffin from the laboratory of anatomy and pathological cytology CHU Fattouma Bourguiba.
Biopsies and surgical specimens are usually kept in formalin fixed, paraffin for histological analysis. Although the morphological integrity of the tissue is preserved, this technique can lead to the degradation of nucleic acids RNA and especially because it is more fragile. The development of extraction protocols has not ceased to be the concern of different research teams.
Silva, et al. in 2007, shows that the deparaffinization by d-limonene gives best results in quantity and quality of RNA extracted from the toluene deparaffinization [13]. In a recent study carried in Germany, the deparaffinization tissue was done by incubating the tissue in octane and methanol washes were required to remove traces of octane, after DNA extraction the detection of DNA by PCR was 70% [16]. While toluene was chosen by several teams for its effectiveness in removing paraffin [17,18].
In our work, we opted for the toluene deparaffinization technique that has been continued to warm during cell lysis with proteinase K. This technique has been described by several authors who have shown that its efficiency and profitability [13,17,18]. In our work, we had a good average concentration of DNA/RNA (between 50 and 300 micrograms/ml) measured by spectrophotometer.
For separation of viral genes, we used the technique of using phenol (Trizol®). According to a study by Guarner, et al., the percentage of detection was 100%, this high sensitivity is due to the use of commercially available kits (Colormark Plus, Erie Scientific, Portsmouth, NH and Qiagen, Valencia, CA) for deparaffination tissue and DNA extraction [18]. To improve the quality and quantity of DNA we also used a commercial kit for DNA extraction Kit (ZR Viral DNA Kit TM) which gives an excellent performance.
Several gene amplification techniques can be used for the detection of enteric viruses and most are based on the amplification of conserved sequences.
In our study, research AdV genome was done using PCR with specific primers that detect a region conserved in all serotypes of AdV in the hexon gene [9]. RV detection was made by amplifying the gene encoding the VP6 protein by RT-PCR as it was previously described [9,10]. These results are consistent with those reported in the literature and showed that the PCR technique is recommended for the detection of viruses in paraffin-embedded tissue samples. AdV are involved in 5 to 10% of inflammatory diseases in children. It is very common to develop an immune response against the AdV before the age of 10 years. The AdV are responsible for persistent infections including reactivation in immunocompromised patients and can lead to serious health consequences [19].
The association between IIA and AdV infection has been reported in several studies [5,20,21] and the high frequency of the disease is reported in season between April and June which is when many viral diseases of oto-rhino-laryngeal child are diagnosed, this reinforcing the hypothesis of the involvement of AdV in IIA [22]. In our study, the frequency of association between IIA and AdV infection in the inverted population of children (n = 9) was 14%. This frequency is lower than the detection rate reported in the literature which varies between 30 and 60% [18,23,24]. This difference could be due to the small size of the sample studied (9 cases).
In 2006, Selvaraj, et al. studied 21 cases of IIA in children; the Adv was isolated in 40% of cases, with a predominance of group C AdV infections [5]. Similarly, in a study in 1961, AdV was highlighted by the complement fixation technique in 6 among 10 children stool inverted (60%). AdV detected among these as follow were; 4 AdV type 5, one AdV type 4 and one AdV type 7 [13].
According to a study conducted in Mexico, AdV group C was found in 33% of tissue samples fixed in formalin and paraffin soaked taken from Mexican patient's hospitalized pediatric ward with IIA. Antigens and viral nucleic acids were localized in epithelial cells and mononuclear cells [18].
A new hypothesis has recently been developed on the pathophysiology of IIA associated with AdV infections and assumed that Adv infection could be the cause of inflammation of the mesenteric plexus nerves characterized by an influx of CD3 positive which affects intestinal motility and causes obstruction [25], inflammation of the mesenteric plexus has also been reported in infections by Cytomegalovirus and Epstein Bar Virus [26]. This hypothesis is partly related to the fact that the enteric nervous system is a key element of the control of intestinal function. This particular control intestinal motility by releasing neurotransmitters (acetylcholine, nitric oxide ...); this is the phenomenon of neuromuscular transmission. These chemicals issued by the neurons will induce coordinated activity of intestinal muscle.
According to the literature, the average age of children with IIA is between 4 and 12 months [22,23,27]. Page, et al. report that IIA occurs in 70-80% of cases, before the end of the first year. In our study, the average age of the invaginated child was 3 years. Male predomination was found in many published data with a sex ratio varying between 2 and 3 and was observed in our study a sex ratio equal to 2, despite the small size of the sampling [22,28]. According to a retrospective study, 72.7% of lesions are ileocecal-colic seat [29], as is the case in our patient invaginated.
Appendicitis is abdominal surgical pathology most frequently encountered in the pediatric emergency department [30]. It can be not complicated and some cases atypical can be complicated by peritonitis. The standard treatment for appendicitis is appendectomy [31].
In our study, the population consisted of 40 children with acute appendicitis. The AdV was demonstrated in 20% of cases. Until now, the associations between AdV infections and appendicitis are not searched, but the results and arguments presented by some studies are encouraging and depart in favor of this hypothesis.
In 2000, a study in the United States showed that the incidence of acute appendicitis is high along with outbreaks. Relationship between appendicitis and AdV infection was explained by the increase of Acs anti-AdV [32].
In 2001, this hypothesis was supported by another study in Central Africa (Bangui). In this temperate country, an increase winter and spring appendicitis was highlighted. This seasonal variation is observed especially in children of age school and has been explained by a viral etiology [33].
The detection rate of RV was 12.5% in the child population inverted in our study. This rate can neither affirm nor confirm a causal relationship between RV and IIA due to the small sample, but this result is similar to these found by other teams: Konno, et al. reported that wild RV was detected by electron microscopy in 11 of 30 children inverted (37%) [34].
Mulcahy has carried the epidemiological study in order to investigate a possible relationship between the RV and the IIA showed no association, while research by electron microscopy, immunofluorescence and ELISA feces showed the presence of RV in 2 out of 24 (8%) children inverted [35].
The role of rotavirus has been described mainly in acute enteritis, but its role is also found in other diseases such as Henoch Schonlein syndrome, Reye's syndrome, Crohn's disease and IIA in children [36].
Studies have reported cases of IIA in children following inoculation of tetravalent reassortant rotavirus vaccine human (RRV-TV; RotaShield). The vaccine was withdrawn from the market because of its association with the IIA [37,38]. Two pathological mechanisms are discussed recently, inflammation of the nerve plexus due to lymphocytic infiltration and is associated with AdV infection and hyperplasia of Peyer's patches with the development of a mass pseudo-morality. In fact, Peyer's patches are made of a very dynamic lymphoid tissue that responds to stimuli of various kinds, by the formation of hyperplastic follicles lymphocyte. In children, RV infection intestinal has been identified as the cause of the hyperplasia of lymphoid tissue and the formation of a pseudo-tumor mass [27].
For wild RV which detected in cases of IIA, the pathophysiological mechanism involves was a viral enterotoxin (NSP4) produced by these viruses and the effect depends on the age of the infected individual and the dose of enterotoxin introduced in the digestive tract. In mouse, enterotoxin acts on the enteric nervous system by increasing the secretion of fluids and electrolytes affecting intestinal motility [39].
Children surgical digestive diseases are very common, especially in developing countries. Their origins are diverse and poorly studied in Tunisia which raises the scientific community a challenge. The viral etiology has been suggested in several studies which all can neither confirm nor deny a causal relationship between viral infections and digestive surgical pathologies. Our results are in line with those reported in the literature. The PCR technique used is sensitive and specific for the detection of adenovirus in tissues fixed in formalin and soaked in paraffin. To our knowledge, this is the first study in Tunisia which focuses in etiology of enteric viruses in the digestive surgical pathologies in children.
No.