Capnocytophaga is a rare bacteria found in the saliva of canines. C. canimorsus is present in the oral cavity of 67-86% of dogs. In vitro growth requires specialized media. Patients who are immunocompromised are at greatest risk for infection. There are no clinical trials examining the duration of antibiotics.
Capnocytophaga species is a very rare fastidious, slow growing gram negative rod found in the saliva of canines and felines [1]. It belongs to the genus flavobacteriacea, a capnophilic facultative anaerobe [1]. In vitro growth requires a specialized media containing, high iron and carbon dioxide. The exact number of cases is unknown because this is not a reportable disease. In one study the incidence rate was estimated at 0.67 per million [2]. Humans contract the infection after receiving an animal bite, scratch, or an open wound being licked. Of the nine species within the genus C. canimorsus is the most common cause of infection in humans. In the Netherlands a retrospective study found 32 cases of C. canimorsus isolated over a three year period. Of those, 28 were diagnosed by blood culture [2]. This particular species is associated with a high morbidity and mortality. A large scale retrospective study from California reviewed 56 human isolates during a 32 year period with a 33% mortality rate [2]. One of the reasons for the high morbidity and mortality is because the bacteria contains lipo-proteins which inhibit hemolysis causing disseminated intravascular coagulation (DIC) [1]. Researchers have also discovered that C. canimorsus possesses a mechanism of passive evasion. The passive mechanism involves the escape of TLR4 detection through the unique LPS structure. Also, it has been discovered the bacteria does not activate signals leading to the release of proinflammatory cytokines, chemokines, and nitric oxide [3]. Patients who are immunocompromised, asplenic, alcoholic, or have cirrhosis are at the greatest risk for infection [2].
Capnocytophaga, Dog scratch, Risk factors
We present a case of a 64-year-old Caucasian male, with no past medical problems or alcohol abuse who presented to the emergency department with acute severe generalized weakness, fever, chills, nausea, vomiting, diarrhea, arthralgias and visual hallucinations. Two weeks prior his dog scratched him on the abdomen. It was not uncommon for his dog to scratch him, but this time the scratch did not heal and progressively turned black. Vital signs on admission, core temperature 100.4, BP 95/71, HR 115, RR 33, Oxygen saturation 93% on room air. On physical exam the tip of his nose and lips were cyanotic (Figure 1A). Skin rash was noted on his face, abdomen, and on all extremities (Figure 1A and Figure 1B). There was mottling of the hands and feet with centripetal spread pattern. Purpuric lesions with a lacy reticular rash pattern were present on both legs. An area of well demarcated linear necrosis measuring 8 cm in length and ½ cm wide, was seen on the epigastric region extending into the left upper quadrant (Figure 1B). Labs on arrival: white blood cells (WBC) 20.6 × 1000 mm3 (4.0-11.00 mm3), sodium 132 meq/L (135-1450 meq/L), potassium 5.0 meq/L (3.5-4.5), blood urea nitrogen 57 mg/dL (9-21 mg/dL), creatinine 3.6 mg/dL (64-108 mmol/L), bilirubin 1.3 mol/L (< 4 Umol/L), alkaline phosphate 171 u/L (56-119 u/L), aspartate aminotransferase 915 u/L (< 35 u/L), ALT 704 u/L (< 45 u/L), creatine phosphokinase (CPK) 6,242 mcg/L (10-120 mcg/L) lactic acid 2.59 mmol/L ( 0.5-1.0 mmol/L), myoglobin > 2000 ng/ml (0-85 ng/ml), platelets 56,000 u/L (150,000-450,000 u/L). Coagulation panel: Protime 26.6 seconds (11.0-13.5 seconds), international normalized ratio (INR) 2.5 (1.1 or below), activated partial thromboplastin time 54 seconds (30-40 seconds), Fibrinogen level 183 mg/dL (150-400 mg/dL), D-Dimer > 35.2 mg/L (< 0.5 mg/L). Arterial Blood Gas: 7.340 pH (7.35-7.45), Arterial PaCO2 < 15.0 (38-42), Arterial PaO2 83.9 (75-100), oxygen saturation 96% (94-100%).
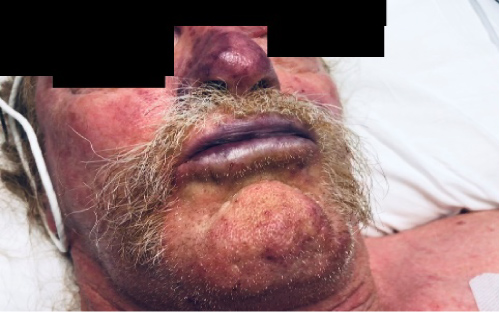 Figure 1A: On physical exam the tip of his nose and lips were cyanotic.
View Figure 1
Figure 1A: On physical exam the tip of his nose and lips were cyanotic.
View Figure 1
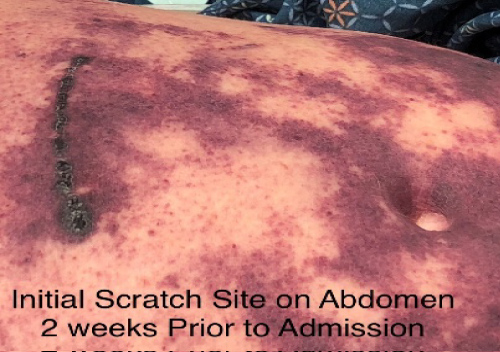 Figure 1B: Initial scratch site on abdomen 2 week prior to admission.
View Figure 2
Figure 1B: Initial scratch site on abdomen 2 week prior to admission.
View Figure 2
Patient was admitted to ICU and started on broad spectrum IV antibiotics for suspected toxic shock syndrome and DIC secondary to possible Streptococcus, Clostridium, Capnocytophaga, or anaerobic infection. Clindamycin was added for the inhibition of toxin production. Vasopressors were not required as his blood pressure remained normotensive. Excisional wound debridement of the abdomen was performed beyond the lines of demarcation, to ensure removal of devitalized tissue. Tissue culture was sent to microbiology. On hospital day #2, repeat labs declined with a platelet count of 12,000/uL, at that time patient was treated with intravenous immunoglobulin and Cryoprecipitate 100 units. Continued CPK elevation > 13,000 was indicative of Clostridium vs. Capnocytophaga. CPK levels reflected rhabdomyolysis which indicated bacterial invasion into the muscle and toxin production. After hospital day #3 all labs started to improve. Blood cultures grew gram negative rods at 72 hours. Lab samples were sent out for DNA identification and antibiotic sensitivity testing. The lack of organism growth after one week may have been secondary to antibiotic effect or slow growing facultative bacteria. The patient continued to improve and remained hemodynamically stable. Large bullae were present the hands bilaterally (Figure 1C). He was transferred out to a tertiary facility due to worsening severe necrosis of his hands and feet bilaterally, to the point of mummification (Figure 1D, Figure 1E). On day 10 there was laboratory confirmation of Capnocytophaga species on blood culture. The CDC was notified and tissue samples were collected from the hospital. Subsequently, the CDC report did not show any further species identification. We can conclude this is C. canimorsus based on the nature of the acquired infection. At the receiving facility, the patient was started on IV levofloxacin and continued Clindamycin and meropenem. Additionally patient received amputation of the first and second digits of the right hand. Digits 3-5 of the right were amputated at the level of the distal metacarpals. The left hand had amputation of digits 2-5 at the level of distal metacarpals. Twice daily dressing changes were performed on bilateral hands and feet. The patient continued IV antibiotics through a PICC line for a total of eight weeks. Patient was discharged from the hospital with weekly wound care which consisted of minor debridement and bathing eschar in iodine baths. Ultimately our patient had bilateral transtibial amputations.
 Figure 1C: Large bullae were present the hands bilaterally.
View Figure 1c
Figure 1C: Large bullae were present the hands bilaterally.
View Figure 1c
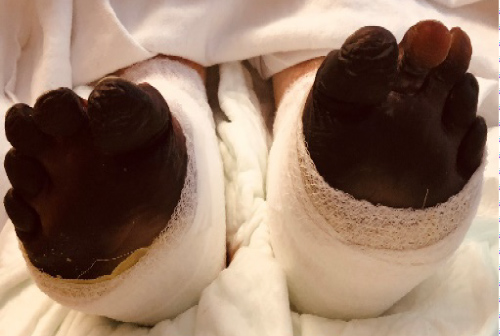 Figure 1D: Worsening severe necrosis of feet bilaterally.
View Figure 1d
Figure 1D: Worsening severe necrosis of feet bilaterally.
View Figure 1d
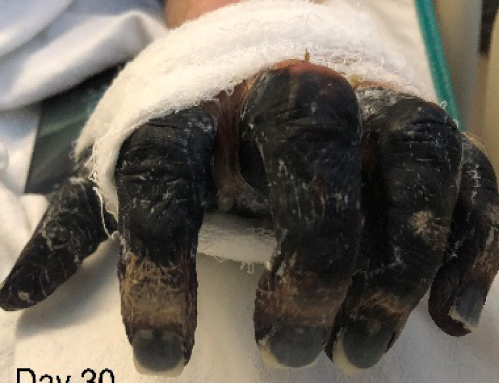 Figure 1E: Worsening severe necrosis of hands bilaterally.
View Figure 1e
Figure 1E: Worsening severe necrosis of hands bilaterally.
View Figure 1e
To date he is stable and continues to follow with his primary care physician on an outpatient basis and physical rehabilitation.
Capnocytophaga is a rare infection associated with high morbidity and mortality. Clinicians should have a high level of suspicion for Capnocytophaga species if patients report any wound from an animal, even if it is only a scratch. If this organism is in your differential it is important to notify the lab of specialized media requirements when obtaining cultures. Due to the slow growth rate, cultures may not be positive for 14 days. In our patient, blood cultures were positive at day 10. With that in mind, empiric antibiotic coverage should be adjusted accordingly. Besides aggressive antibiotic therapy, our patient required surgical debridement, IVIG, and cryoprecipitate due disseminated intravascular coagulation. To date there are no clinical trials examining the duration of antibiotic therapy. Our patient received a total of eight weeks of IV antibiotics. This case summarizes the importance of early identification and aggressive treatment of Capnocytophaga.
This research was supported (in whole or in part) by HCA and/or an HCA affiliated entity. The views expressed in this publication represent those of the author(s) do not necessarily represent the official views of HCA or any of its affiliated entities.