Background: COVID-19 has certain presenting features, laboratory biomarkers, imaging findings that help identify patients with poor prognosis.
Methods: Clinical features, laboratory biomarkers, CXRs, and other radiological findings of 804 patients were reviewed with all-cause mortality and ICU requirement. Lung zones at admission were categorized into ≤ 3 zones or > 3 zones and correlated with mortality and ICU requirement after 1 week.
Findings: Logistic regression analysis of presenting complaints with mortality revealed that shortness of breath [OR: 3.1(CI: 2-4.7), p < 0.001] and cough [OR: 1.7(CI: 1-2.8), p 0.05] posed a higher risk of mortality. Fever and gastrointestinal symptoms did not correlate with adverse outcomes. Risk of increased mortality was higher in patients with a greater number of lung zones involved on CXR [OR: 4.7 (CI: 2.9-7.6); p < 0.001]. ICU stay was greater in the same group [OR: 4.4 (CI: 2.9-6.5); p < 0.001]. The findings after 1 week were concordant with the category of > 3 zones, revealing higher odds of mortality [OR 11.3(CI: 5.8-22.0); p < 0.001], and ICU stay [OR: 7.9 (CI: 4.9-12.8); p < 0.001].
COVID-19, SARS-CoV-2, Biomarkers, Radiological manifestations, Clinical manifestations, COVID-19 prognosis
December 2019 heralded the onset of the 2019 novel coronavirus (SARS-CoV-2) outbreak in the Hubei province in China. Ever since the disease has spread across continents resulting in rising magnitudes of cases and fatalities. SARS-CoV-2 is a beta coronavirus that belongs to the family Coronaviridae and order Nidovirales [1]. Six coronavirus species have been identified to infect humans and cause mild disease so far; SARS-CoV-2 is the seventh member of the coronaviruses that infect humans. Fatal illness is mostly observed in severe acute respiratory syndrome virus (SARS-CoV) and Middle East respiratory syndrome Coronavirus (MERS-CoV) [2].
Symptoms of COVID-19 are comparable to those seen in SARS-CoV and MERS-CoV infections [3]. COVID-19 patients present with a full spectrum of disease, ranging from the asymptomatic presentation and primary respiratory involvement to neurological features, gastrointestinal symptoms, and thromboembolic phenomena. Most patients with lower respiratory infections present with fever, cough, dyspnea, and myalgia. Additionally, 17% to 29% of patients have acute respiratory distress syndrome [4,5]. An increased risk of severe COVID-19 has been reported in patients with medical comorbidities, such as diabetes mellitus, hypertension, and cardiovascular disease [6,7]. The predominant CT findings of COVID-19 infection are bilateral, peripheral, and basal predominant ground-glass opacity, consolidation, or both [8,9]. The most common chest X-ray findings that are seen are airspace opacities, which may be consolidations or, less frequently, ground-glass opacities [10].
COVID-19 may also predispose to both venous and arterial thromboembolic disease due to excessive inflammation, hypoxia, immobilization, and disseminated intravascular coagulation (DIC) [11-14]. This multicentric retrospective study aimed to evaluate and summarize the clinical, laboratory, and radiological features in patients admitted to three tertiary care hospitals in Dubai, United Arab Emirates (U.A.E.); and to assess the correlation of these clinical, laboratory, and radiological features with the outcome, measured by all-cause mortality and requirement of ICU stay.
The study was approved by the Research and Ethical Approval Committee with reference number DOH/CVDC/2020/2313. A multi-center retrospective descriptive study was performed on patients admitted to three tertiary care centers, namely Rashid Hospital, Dubai Hospital, and Latifa Hospital.
Medical records of 1709 patients with RT-PCR-confirmed COVID-19, who were admitted to these hospitals during the period 01/06/2020 to 31/11/2020, were retrospectively reviewed (Figure 1). All patients of the age group 18-80 years admitted in this period were COVID-19 positive based on real-time RT-PCR tests. Those who had laboratory investigations (Complete Blood Count, C-Reactive Protein, Ferritin, LDH, and Dimer) on admission and repeated values after 7 days; radiological imaging with Chest X-Ray (CXR) on admission and results repeated after 7 days were included in the study. The patients who were < 18 or > 80 years of age, with a negative COVID-19 PCR report, no admission or follow-up labs or chest X-ray after a week, and in acute heart failure, with active pulmonary tuberculosis, acute exacerbation of chronic obstructive pulmonary disease, acute asthma exacerbation, and other pre-existing lung conditions, which could obscure the progression of COVID-19 were excluded from the study.
 Figure 1: Process of patient selection.
View Figure 1
Figure 1: Process of patient selection.
View Figure 1
In addition to patient demographic characteristics and comorbidities, data regarding clinical features were retrieved from the electronic medical records. Laboratory workup including ferritin, lactate dehydrogenase (LDH), dimer, C-Reactive Protein (CRP), lymphocytes, and neutrophil counts at admission and after one-week post-admission was recorded. Clinical severity was defined using the WHO clinical progression scale [15].
Results of brain imaging, chest imaging (X-ray and CT), abdominal and large vessel imaging, that had been performed for these patients were collected. Imaging data were confirmed by the two radiologists to reduce the chances of error.
We categorized the findings into 4 specifications in the following manner:
• Normal
• Non-specific findings - including patchy atelectasis, bronchial wall thickening, pleural effusions
• Unilateral findings - including consolidations/ground-glass opacities
• Multilobe or diffuse consolidation
• Ground glass or bilateral infiltrates
The extent of the pulmonary abnormalities was monitored through a conventional semi-quantitative scoring system [16]. The assessment for radiological severity was conducted using the Brixia score, which divided the lung into 3 zones, as illustrated in Figure 2. A total score was calculated at admission and after a week of hospital stay to determine an improvement, disease worsening, or no change.
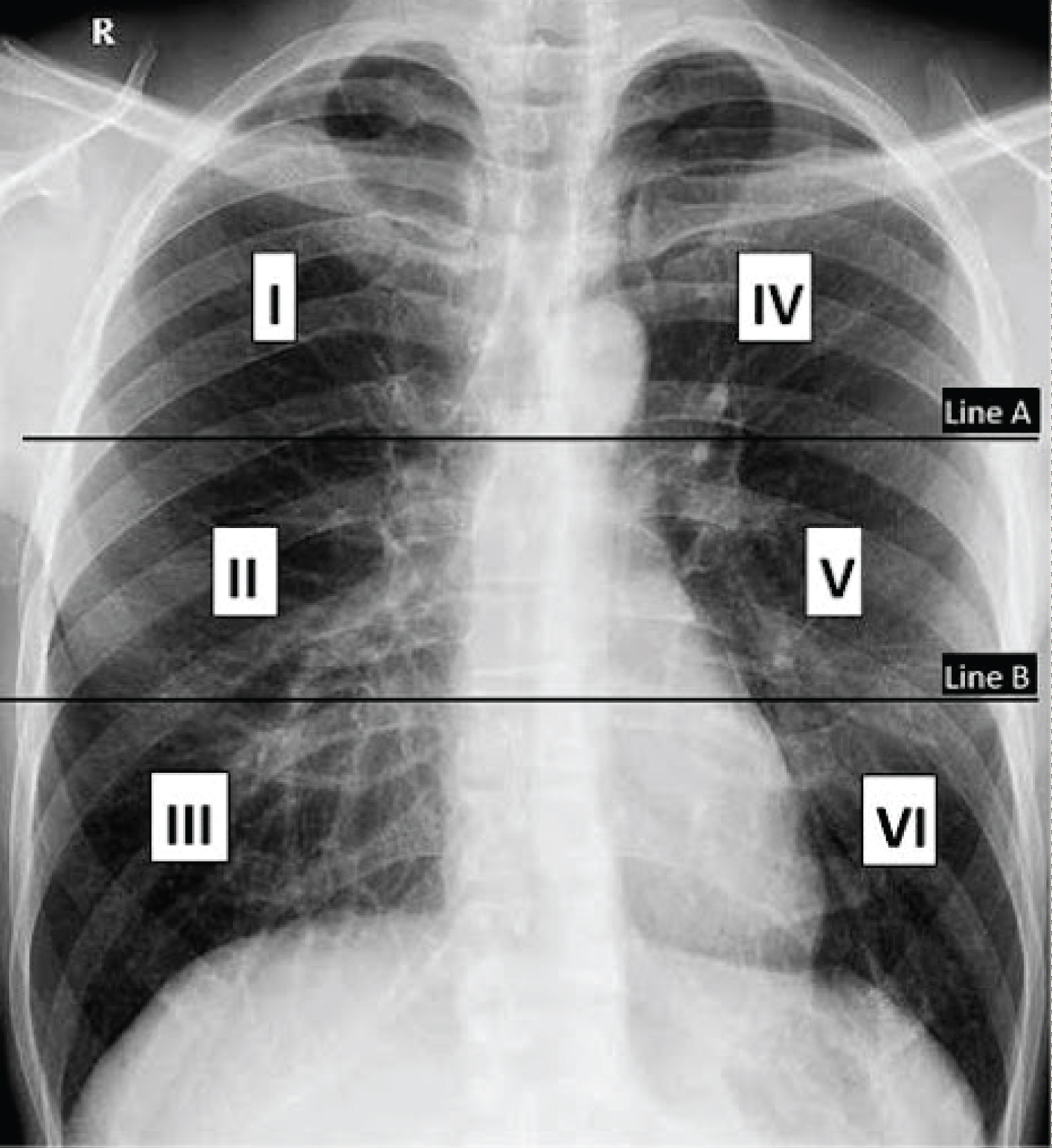 Figure 2: Demonstration of Brixia scoring system - image has been taken from Polish Journal of Radiology.
View Figure 2
Figure 2: Demonstration of Brixia scoring system - image has been taken from Polish Journal of Radiology.
View Figure 2
CT chest findings were studied for the following:
• Ground glass opacities
• Consolidation
• Ground glass opacities + consolidation
• Interlobular septal thickening
• Reticular pattern
• Crazy paving
• Air bronchogram
• Bronchial wall thickening
• Bronchiectasis
• Pleural thickening
• Pleural effusion
• Subpleural line
• Nodule
• Reversed halo sign
• Lymphadenopathy and pericardial effusion [17]
Large vessel imaging was studied for any thrombus presence; findings of clot verse no clots.
Continuous variables were presented as mean ± SD or median (Q1, Q3) as appropriate, and categorical data was organized into frequency and percentages. Logistic regression analysis was performed to assess the presence of symptoms and chest X-ray findings with mortality and ICU stay. Mann-Whitney test is performed to study prognostic markers with mortality. A P-value less than 0.05 indicate statistical significance. Data entry and statistical analysis were done using SPSS v20.0 (IBM Corp, Armonk, NY, US).
804 patients were admitted to multiple centers of Dubai Health Authority with confirmed COVID-19 positive. Table 1 shows the general characteristics of the study population. The mean age of the total population is 45.4 ± 11.6 years and the male population of 737 (91.7%). Assessing for comorbidities, 22.8% (183/804) of the population have hypertension and 32.5% (261/804) have diabetes. There was 11.7% of the population were found to have other comorbidities that included ischemic heart disease (20.2%), dyslipidemia (26.6%), hypothyroidism (9.6%), and the rest had bipolar disorder, depression, peptic ulcer disease, bronchial asthma, and Chronic Obstructive Pulmonary Disease (COPD). The most common symptom was fever (85.9%) followed by cough (72.9%) and shortness of breath (40.2%). Gastrointestinal symptoms were less commonly found in 15% and 3.2% of the patients were asymptomatic. Prognostic markers on admission revealed LDH 312.5 (234-438), ferritin 773 (366-1361), and dimer 0.8 (0.5-1.5) (Table 2).
Table 1: General characteristics of the study population. View Table 1
Table 2: Prognostic markers on admission concerning mortality. View Table 2
Chest X-ray on admission most revealed ground-glass opacities/bilateral infiltrates observed in 454 (56.5%) of the cases. The next common finding was multilobe/diffuse consolidation observed in 186 (23.1%). Normal chest X-rays were observed in 117 (14.6%) of the cases. An improvement in repeat chest x-rays was noted in 358 (44.5%) of the cases, while 297 (36.9%) showed worsening conditions.
A high-resolution CT chest was performed only in 27 patients with ground-glass opacities identified as the most common finding. Brain imaging (CT scan or MRI) was performed in 46 cases, among which 13 cases had findings of infarction and 10 cases had evidence of hemorrhage.
A total of 11 patients in the study had positive large-vessel imaging for deep vein thrombosis, pulmonary embolism, myocardial infarction, and carotid thrombosis.
Using logistic regression, presenting complaints were studied with mortality and we noted that presence of shortness of breath [OR: 3.1(CI 2-4.7); p < 0.001] and cough [OR: 1.7(CI: 1-2.8), p 0.05] posed a higher risk of mortality (Table 3).
Table 3: Analysis of symptoms and comorbidities with mortality. View Table 3
Number of lung zones involved on chest X-ray on admission were categorized into ≤ 3 lung zones or > 3 lung zones; these were studied with mortality and requirement of ICU stay after 1 week. We noted that the risk of mortality was higher in those patients with a higher number of lung zones involved on X-ray chest [OR: 4.7 (CI: 2.9-7.6); p < 0.001]. ICU stay was also noted to be higher in the same group [OR: 4.4 (2.9-6.5); p < 0.001]. The findings after 1 week were concordant with a category of > 3 lung zones revealing higher odds of mortality [OR 11.3 (5.8-22.0); p < 0.001] and ICU stay [OR: 7.9(4·9-12.8); p < 0.001] (Table 4).
Table 4: Relationship of mortality and ICU admission to lung zone involvement on CXR. View Table 4
The SARS-CoV-2 outbreak, which occurred at the end of 2019, has claimed more than 5 million lives across the world and still counting, as per the WHO COVID-19 dashboard data.
Almost 2 years later, as clinical knowledge of this disease accrues, outcomes can improve. This can be achieved by identifying certain imaging, lab findings, and elevations of biomarkers, which are known to correlate with disease severity; thereby leading to the early institution of advanced therapies like tocilizumab, IVIG, and other monoclonal antibodies, resulting in better resolution of the disease in many cases.
This is one of the large retrospective descriptive studies of the region, to our knowledge, which comprehensively summarizes clinical characteristics, prognostic markers, radiological findings, and outcomes of consecutive COVID-19 cases. Male patients formed a great majority of 91.7% in our study. There was no correlation between gender and mortality in contrast to the study by Hannah and colleagues, where male subjects were found to have a higher rate of ICU admission and death [18]. Diabetes mellitus type 2 was the most common chronic disease in our study population seen in 32.5% of cases, followed by hypertension in 22.8% of cases. One of the risk factors, hypertension might worsen the COVID-19 outcomes; however, some data suggest that patients admitted to intensive care with COVID-19 have high blood pressure compared to those who do not have COVID-19 [5,19]. In contrast, another study by Sheppard and colleagues found an inverse relationship between recent blood pressure control and COVID-19 related death [20].
Diabetes was not a prevalent comorbidity, according to a study by Guo and colleagues [21], in the total population (7.4%), but it was significantly more prevalent in the patients who experienced primary complex endpoints (intensive care admission, mechanical ventilation, and death) than in patients who do not have these complications (26.9% vs. 6.1%). In a meta-analysis, among 1558 COVID-19 patients, patients were found to have diabetes, hypertension, and chronic obstructive pulmonary disease in positive correlation with exacerbation and admission to intensive care units [22]. Another study from Brazil [23] found a positive association of diabetes mellitus type 2 and hypertension with mortality, resembling our study.
Amongst the various implicated pathophysiological mechanisms that reveal a connotation between diabetes and COVID-19 severity, deficiencies in the innate immune system due to chronic hyperglycemia, pro-inflammatory responses as a result of inappropriate and exaggerated cytokine response, and underlying pro-thrombotic states have been found to contribute towards its development [24].
The mechanism of angiotensin-converting enzyme 2 (ACE2) receptors in hypertension-associated COVID-19 severity has been implicated through the high affinity of these receptors to the SARS-COV-2 virus. This facilitates the viral binding to the targeted epithelial cells of the lung, heart, and other organs [25].
The presence of cough and shortness of breath correlated with adverse outcomes compared with patients who did not have these clinical symptoms. This is further supported by a study performed in Jakarta [26] where, in a comparison between discharged and deceased patients, the latter were more likely to have cough, fever, malaise, shortness of breath, headache, nausea/vomiting, chills, diarrhea, abdominal pain as the presenting symptoms. However, in our study patients with GI symptoms and others (including malaise, headache, dizziness) did not correlate with mortality.
Lung zones’ involvement had a direct correlation with mortality; this was further supported by a study performed by Anas and colleagues [27], where they concluded that the involvement of 4 or more lung zones had a greater degree of clinical deterioration within a given median interval time of 2 days. Another study published in the European Journal of Radiology [28], which consisted of chest X-ray image analysis of 953 patients, established that the highest value reached by the score was significantly showed higher in deceased patients as compared to discharged ones.
Among the chest X-rays analyzed on admission, unilateral/bilateral infiltrates was seen in 56.7% of the cases and was the most common finding; this was similar to a quantitative meta-analysis covering 2847 patients in China and Australia, and to a multi-centered descriptive analysis of 39 case reports summarising 127 patients, which found changes in bilateral chest radiographs (72.9%, 95% confidence interval 58.6 to 87.1) and ground-glass opacity in 68.5% of cases (95% CI 51.8 to 85.2) due to COVID-19 pneumonia [29].
Three studies found the lymphocyte count, neutrophil count, and CRP were associated with the severity of COVID-19, similar to our study, except the neutrophil counts, which did not correlate with adverse outcomes [30-32]. Another study concluded that LDH could be identified as a significant predictive factor for early recognition of lung injury and severe COVID-19 cases, but the study was limited due to the low population [33]. However, our study of 804 patients identified non-correlation between elevated LDH levels at admission and fatal outcomes.
There were 12.7% patients which were reported deceased in our study, due to various causes, such as acute respiratory distress syndrome, acute respiratory failure, acute kidney injury, septic shock, hospital-acquired infections, and multi-organ dysfunction. This was much higher than the death rate of 4.3% seen in a systematic review and meta-analysis of observational studies [34]. The higher number of deaths found in our study could probably be because our sample size was much smaller than them, and we had only moderate and severe cases admitted at our facility, with mild cases sent to other isolation centers dedicated to the care for stable COVID-19 patients.
A high-resolution CT chest of 27 patients was performed; ground-glass opacities were the most common finding which, consistent with the results of a meta-analysis, correlated with poor outcomes [35]. Considering the availability and reliability of the PCR test for COVID-19 diagnosis, CT was not employed as the diagnostic modality of choice for the initial COVID-19 assessment due to limited availability, risk of disease spread, and arduous time-consuming decontamination measures in presence of lack of substantial evidence or guidelines. CT was used to investigate in cases of rapid deterioration, or when another co-existing or alternative diagnostic possibility was suspected.
There were 46 patients with COVID-19 who underwent brain imaging due to their symptomatology, out of which 28.2% showed an ischemic stroke. There are several mechanisms for ischemic stroke in the setting of COVID-19 infection that have been proposed, including the relationship of severe COVID-19 disease occurring more frequently in older patients who are more likely to have multiple comorbidities increasing their risk of stroke [36]. The key proposed mechanisms include cytokine storm and activation of the innate immune system, embolic events propagated by pre-existing or new-onset arrhythmias, hypoxia-induced ischemia secondary to severe pulmonary disease, thrombotic microangiopathy, endotheliopathy, and multifactorial activation of coagulation [37]. A recent review by Khan and colleagues found an unswerving increase in D-dimer levels in patients with severe compared with a non-severe COVID-19 disease, which was alike our report [38].
Furthermore, 21.7% of the patients who underwent brain imaging were found to have intracranial hemorrhage. Case reports on hemorrhagic stroke associated with COVID-19 in Mexico General Hospital illustrated radiological findings, laboratory tests, and prognostic characteristics of four patients, with stroke. The common symptoms in those patients were predominant cerebellar hemorrhage and acute respiratory distress. A significant increase in cardiac enzymes and brain hemorrhage were seen because of the viral neurotropism. This, accompanied by its entry through various routes to cross the blood-brain barrier, including nasal mucosae, transport through olfactory nerve, or via hematogenous route, directly infecting endothelial cells of the blood-brain barrier of the central nervous system (CNS) is among the many mechanisms by which COVID-19 is known to cause intracranial hemorrhage [39].
A study performed by Corrina and colleagues evaluated the incidence of ICH in Acute Respiratory Distress Syndrome (ARDS) patients, in both COVID-19 and non-COVID-19 subgroups and found no significant difference in its incidence, indicating that COVID-19 may not a directly involved in causing ICH, and the mechanisms are related to etiology of ARDS [40]. Another meta-analysis found a low incidence of ICH in patients with COVID-19 (0.38%), but the high mortality rate (54%) indicates ICH to be among the most severe neurological complications of COVID-19 known to occur in the setting of critically severe COVID-19 [41].
Based on our study, we can conclude that deceased COVID-19 patients had a significantly higher LDH, ferritin, dimer, neutropenia, and lymphocytosis. The odds of requiring ICU care and all-cause mortality were considerably greater in patients with bilateral pneumonia. The most common symptom that was associated with mortality was shortness of breath. These findings confirm the risk of mortality associated with bilateral pneumonia and confirm the relationship of high prognostic markers with mortality.
Chest X-ray study can be employed as a useful tool for initial assessment, follow-up, and prognostication of severity, and for assessment of deterioration in patients withCOVID-19 in resource-limited settings.
There are some limitations of this study. Firstly, the follow-up chest X-rays were not repeated at an exact set point in time and were rather done as per the clinical status of the patient, mostly upon clinically deterioration. Similarly, the repeat of lab biomarkers was also not done with the same frequency and used depending on the clinical status of the patient. We had very few chest CTs, and other radiological imaging done; these were performed only when clinically required, so a more accurate correlation with mortality could not be established. Finally, the study does not correlate the comorbidities, apart from diabetes and hypertension, with mortality, and does not account for the fact that worsening of the pre-existing comorbid conditions might contribute to a more significant change in the patients’ clinical and pathological reports in COVID-19 prognosis.
Higher lung zone numbers on CXR, presence of shortness of breath, and cough predicted an increased risk of mortality. Incidence of fever or gastrointestinal symptoms does not show any association but needs further research.
This research did not receive any specific grant from funding agencies in public, commercial, or not-for-profit sectors.
The authors declare no conflict of interest in the research and authorship.
Rommana Mehdi - conceptualization, methodology, project administration, investigation, writing - original draft, writing - reviewing and editing; Arshee Khan - Clinical data collection, Data analysis, Software supervision, writing - original draft; Priyank Gupta - Data analysis, report writing - original draft; Aisha Siddiqui, Anam Ahsan - Clinical data collection, report writing - original draft; Mona Hosseini, Farah Ghayoor, Jawahir Tahir, Samya Javed - Clinical data collection; Saubia Fathima, Zufana Nasir, Hana MohdSughaiyer, Salama Buqaish, Eman Abdelwahed - Radiological data collection, Erum Rehman - Radiological data collection, data analysis; Deema Harb, Beyla Zuberi - report writing - draft; Laila Aldabal - Conceptualization, project administration, writing - original draft.
• Higher LDH, ferritin, dimer and neutropenia is found in deceased COVID-19 cases.
• Cases of bilateral COVID-19 pneumonia have considerably greater odds of ICU stay.
• Chest X-ray can be employed as a useful tool for initial work-up of COVID-19 cases.
• Brixia scoring is an accurate means of determining adverse outcomes and mortality.