Background: Urinary tract infections (UTIs), mostly caused by uropathogenic E. coli (UPEC), are important human infections. Understanding UTI pathogenesis is important for combating the rise in treatment failure and recurrent infections. We studied the distribution of biofilm production by host and non-host factors, among UPEC antibiotic resistant and sensitive cystitis isolates, from reproductive age women.
Methods: Cystitis isolates (n = 534) from women were tested for (i) Biofilm production using a microtitre plate method, (ii) Susceptibility to 14 antibiotics using the CLSI disk method, and finally, (iii) Phylogenetic group status using the improved Clermont method.
Results: A total of 534 UPEC isolates from women were studied. Biofilm production was highest in the 26-30 years age group (73%), and lowest in the 16-20 years age group (35%). A greater proportion of isolates from inpatients (72%) produced biofilms vs. 45% for outpatients. Most of recurrent UTI isolates (92%) were biofilm producers vs. 48% for first time UTI. The majority of the isolates (61%) were resistant to one or more antibiotics. In the overwhelming majority of cases, biofilm production was highest among the resistant isolates vs. susceptible ones, on average by twice as much. A higher proportion of B2 isolates (77%) were biofilm producers compared to any another group.
Conclusion: Our findings suggest a strong association between biofilm production and several host and non-host factors, including age, UTI recurrence, hospitalisation, and phylogenetic groups B2 and D. These factors are associated with multidrug resistance, suggesting a strong link between biofilm production and multidrug resistance, as previously suggested.
Antibiotic resistant, Uropathogenic Escherichia coli
Urinary tract infections (UTIs) are among the commonest human infections, affecting more than 150 million people annually, worldwide [1]. Consequently, healthcare costs for this condition are huge, with an estimated US$ 3 billion spent on UTIs alone per annum [2]. Thus, studies aimed at elucidating UTI pathogenesis, including management strategies, are needed to help in arresting UTI spread. In most UTI cases, E. coli is the causative pathogen, with over 80-90% of uncomplicated UTIs caused by this organism [3].
Uropathogenic E. coli (UPEC) are endowed with several virulence associated factors (VFs) that enable the organism to attach, invade, and injure the host. Among these are antibiotic resistance factors, and the capacity to form biofilms. Management of uncomplicated UTI has become challenging due to rise in antibiotic resistance among UPEC strains, including to traditional first-line agents, such as trimethoprim-sulfamethoxazole (TMP-SMZ) [4]. Although the mechanisms of antibiotic resistance are diverse, previous studies have demonstrated that there is a potential link between antibiotic resistance and biofilm production [5], although other findings are to the contrary [6].
In addition to rising antibiotic resistance amongst UPEC strains, the production of biofilms among these strains is another concern, often leading to recurrent UTIs and chronic infections [6]. Biofilm production has been shown to be an important UPEC virulence factor, as its formation creates an impermeable barrier for antibiotic penetration, and hence minimizing the amount of antibiotic that can reach the site of action, thus contributing to the development of resistance [7]. According to literature, biofilm is estimated to account for 80% of all microbial infections, and over 65% of nosocomial infections [8]. However, despite this staggering statistic, very few studies have investigated the relationship between biofilm production and host factors such age group and hospitalization status, and non-host factors, including recurrence of UTI, phylogenetic group distribution, and antibiotic susceptibility, among urinary E. coli isolates from reproductive age women. Moreover, there seems to be a relationship between phylogenetic background and virulence in UPEC, and how biofilm production potentially fits into this relationship has not been explored in urinary isolates from Australia.
Understanding the relationship between biofilm formation and phylogenetic group distribution may help predict the pathogenic potential of UPEC strains, and may be helpful in establishing novel strategies for controlling UTIs, including minimisation of treatment failure and recurrent UTIs. Some studies have indicated that biofilm forming bacteria are implicated in recurrent UTI pathogenesis and in complicated UTIs which are normally associated with MDR bacteria [9]. Understanding UTI pathogenesis with regards to factors associated with biofilm formation and/or multi-drug resistance, is key to the development of new therapies [10]. Therefore, we studied 534 urinary E. coli isolates from reproductive age women with cystitis, in order to gain insights into (a) The distribution of biofilm production by age group, hospitalisation status and recurrent UTI, (b) The association between antibiotic resistance and biofilm production, and finally, (c) To understand the relationship between biofilm production and phylogenetic group distribution.
The study was carried out in the Central West region of New South Wales (NSW), Australia (population 180,000), and included 11 regional hospitals and 23 outpatient medical centers. The participating doctors were given a standardized urine specimen collection protocol, as well as a strict clinical diagnostic criteria for defining the UTI as cystitis. During history taking and physical examination, the doctors recorded the following; deidentified patient information: Age, clinical UTI syndrome, previous UTI history, and any known underlying host conditions.
A total of 534 urinary E. coli isolates from reproductive age women with cystitis, were studied. The isolates were collected concurrently over a 3-year period (July 2013-July 2015), with only one isolate per subject included in the study. Excluded from the study were patients with known diabetes mellitus, diarrhoea, menstruation, antibiotic therapy in the last month, or urinary tract abnormalities.
Midstream urine specimens were collected from subjects who met the selection criteria. A diagnosis of cystitis required specific clinical manifestations, including frequency of urination, dysuria, and/or suprapubic tenderness, without fever or loin pain, and a urine culture yielding ≥ 108 cfu/L of E. coli. Bacterial isolates were stored in 5% glycerol in trypticase soy broth at -70 °C until further use.
The 534 urinary E. coli isolates were tested for susceptibility to 14 antibiotic drugs according to the Clinical Laboratory Standards Institute (CLSI)-specified disk diffusion method [11], using Neo-Sensitabs discs (Rosco, Taastrup, Denmark). The antibiotics tested included (disk content): amikacin (30 μg), amoxicillin-clavulanate (60 μg), ampicillin (25 μg), ceftazidime (30 μg), ceftriaxone (30 μg), cephalothin (30 μg), ciprofloxacin (10 μg), gentamicin (10 μg), imipenem (10 μg), nalidixic acid (30 μg), nitrofurantoin (300 μg), norfloxacin (10 μg), tetracycline (30 μg), and trimethoprim-sulfamethoxazole (TMP-SMZ) (5 μg). Production of extended-spectrum β-lactamases (ESBLs) was tested by the double-disk diffusion test, as previously described [12]. Antibiotic resistant isolates were those that were resistant to ≥ 1 agent. The resistance score was the number of antibiotic classes for which an isolate exhibited resistance to ≥ 1 representative agent. E. coli strain ATCC 25955 was used as the control strain.
Capacity to produce biofilm was assessed using quantitatively a using a microtiter-plate test assay (Nunc, Roskilde, Denmark) as previously described [13]. Briefly, the bacterial isolates were grown on Brain Heart Infusion (BHI)agar, and colonies were re-suspended in a BHI broth (Oxoid, Basingstoke, UK) to reach the 0.5 suspension of McFarland's standard. Thereafter, a volume of 200 μL of each of these cell suspensions was transferred to the wells of a microtiter plate. The inoculated microtiter plate was incubated for 24 h at 37 °C, and the adherent cells washed three times using a saline solution, and thereafter stained with a 0.1% crystal violet solution (Mikrochem, Pezinok, Slovakia). The adhering dye taken up by the adherent cells was dissolved by 30% acetic acid, and the optical density of the resulting solution was measured at 570 nm in the Synergy HT Multi-Mode Microplate Reader (BioTek, Winooski, VT, USA). For classification of biofilm production among the isolates, we used the average optical density (OD) value and cut-off value (ODc) (defined as three standard deviations (SD) above the mean OD of the negative control). The final OD value of an isolate was expressed as the average OD value of the isolate reduced by the ODc value.
The project was approved by relevant institutional review boards (Charles Sturt University and Sydney West Area Health Service research committees). No clinical information for patients with UTI was provided anonymously by clinicians, patient consent was not obtained.
Comparisons of proportions were tested using Fisher's exact test. Virulence and resistance score comparisons were tested using the Mann-Whitney U-test.
The distribution of the cystitis isolates by age range was generally uniform, ranging from 10% (55 of 534) in the 16-20 years age range, to 19% (101 of 534) in the 26-30 years age range (Figure 1). Based on the rate of biofilm production, the highest rate was observed in the 26-30 years age range at 73% (73 of 101), whilst the lowest rate was in the 16-20 years age range at 35% (19 of 55) (P = 0.001). Generally, the rate of biofilm production followed an ascending gradient from the 16-20 years age range (35%) right through to 52% in the 21-25 years age range, and rising to the highest in the 26-30 years range at 72%. Thereafter, the rate followed a descending gradient, decreasing to 60% in the 31-35 years age range, then to 42% and 41% in the 41-45, and 46-50 years age ranges, respectively. Overall, 349 (65%) of the 534 cystitis isolates studied were biofilm producers.
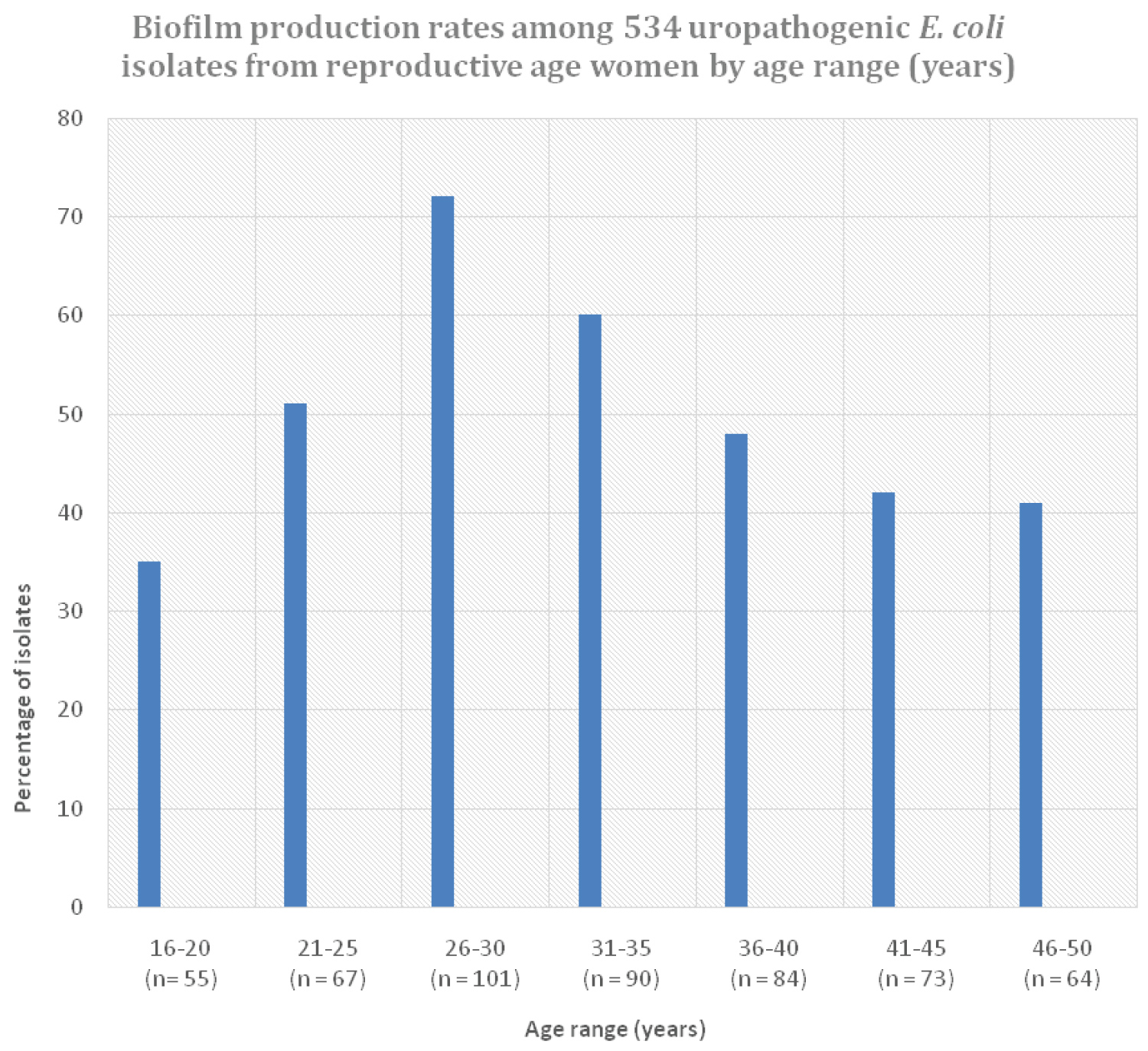 Figure 1: Biofilm production rates amongst 534 uropathogenic E. coli isolates from reproductive-age women by age range (years).
View Figure 1
Figure 1: Biofilm production rates amongst 534 uropathogenic E. coli isolates from reproductive-age women by age range (years).
View Figure 1
Distribution of the cystitis isolates by recurrence of UTI showed that the majority (65%, 347 of 534) of the cystitis isolates were from non-recurrent or first time UTI. And among the recurrent UTI isolates (n = 189), 30% were from women who reported to have had the UTI episode at least 3 times (Figure 2). A big majority of the recurrent UTI isolates (92%) were biofilm producers, compared to only 48% for non-recurrent UTI (P < 0.001). Furthermore, all the UPEC isolates from patients who have had UTI on four or more times were all biofilm producers. In terms of hospitalization, the majority of isolates (364, 68%) were from the outpatient setting. Of these isolates, 45% were biofilm producers, compared to 72% for inpatient isolates (n = 170) (P < 0.001).
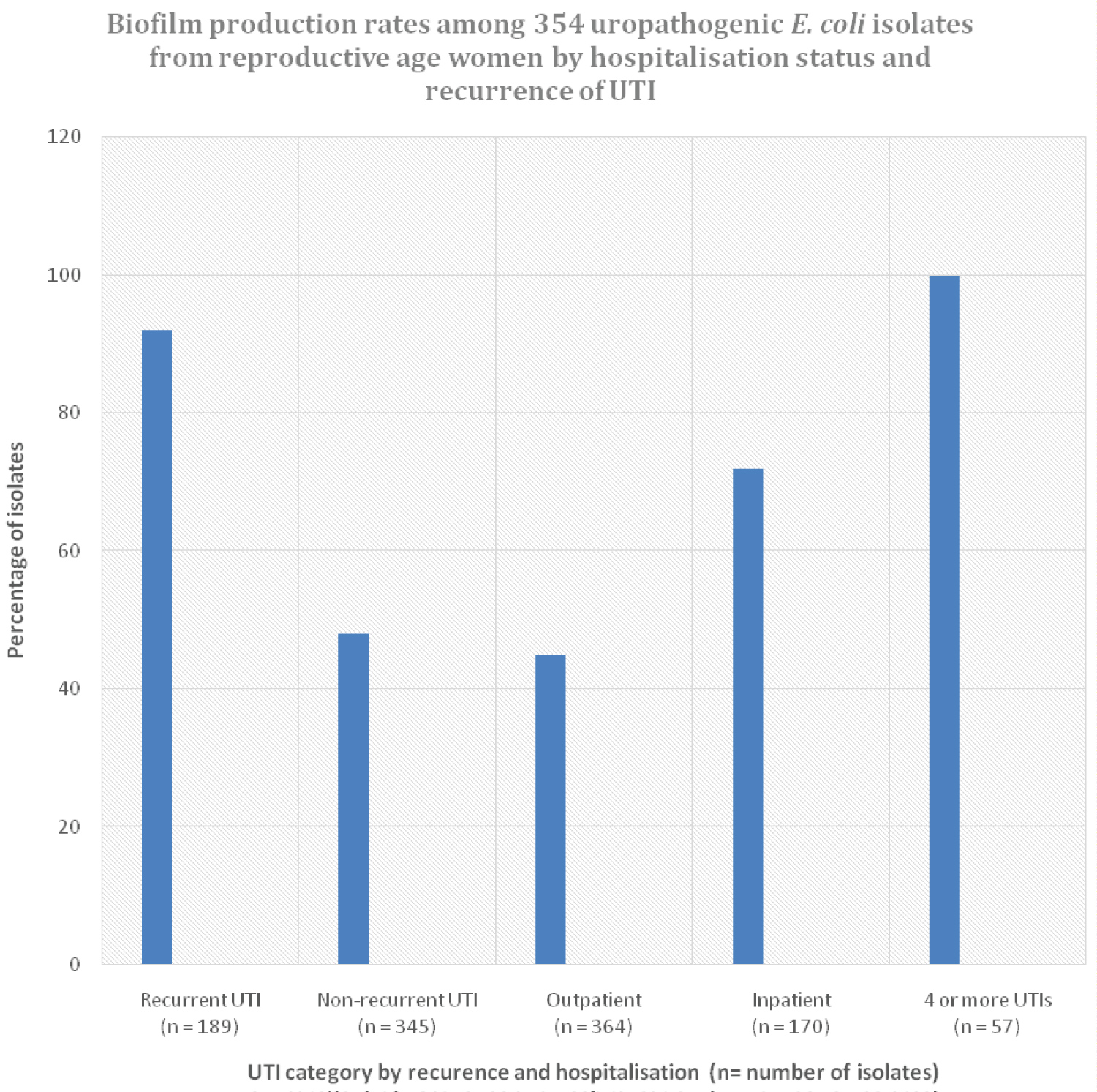 Figure 2: Biofilm production rates by hospitalisation status and recurrence of UTI amongst uropathogenic E. coli isolates from reproductive-age women.
Figure 2: Biofilm production rates by hospitalisation status and recurrence of UTI amongst uropathogenic E. coli isolates from reproductive-age women.
*Recur UTI: recurrent UTI; #Non-Rec UTI: non-recurrent UTI
View Figure 2
The rate of antibiotic resistance amongst the 534 reproductive age women cystitis isolates was high, with 324 (61%) of the isolates resistant to more than one antimicrobial agent, and hence classified as multidrug resistant (Figure 3). Among these isolates, ESBL production was observed in 22 (4%) of the isolates. The prevalence of biofilm production amongst the different antibiotics differed significantly by antibiotic type and susceptibility phenotype. However, for each antibiotic (save for nalidixic acid), the rate of biofilm production was higher in the resistant isolates vs. the susceptible ones, and most of the differences were statistically significant. Specifically, in the majority of antibiotics, the rate of biofilm production among the resistant isolates was twice that of susceptible ones. However, even among the antibiotic susceptible isolates in each antibiotic category, at least 40% of the isolates were biofilm producers, up to a maximum of 70% (nalidixic acid), but it was on average 42% for each antibiotic (Figure 3). Among the resistant isolates, the rate of biofilm production for each antibiotic ranged from 42% (nalidixic acid) to 100%. However, on average, the rate of biofilm production amongst the resistant isolates was above 75%. In 8 of the 14 antibiotics studied, the rate of biofilm production was above 80%, with rates of 100% observed in four antibiotics (nitrofurantoin, gentamicin, amikacin, imipenem, and ceftazidime). Furthermore, all the ESBL producers (n = 22) exhibited biofilm producing capacity, and so were 95% of multidrug resistant isolates (n = 208). Further analysis of biofilm production by different susceptibility categories based on the number of drugs an isolate was resistant to concurrently, revealed that as the number of antibiotics increased, so did the rate of biofilm production (Figure 4). For example, only 46% of isolates resistant to 1-3 drugs (n = 304) were biofilm producers, compared to 100% for those resistant to more than 11 drugs (n = 48) (P < 0.001).
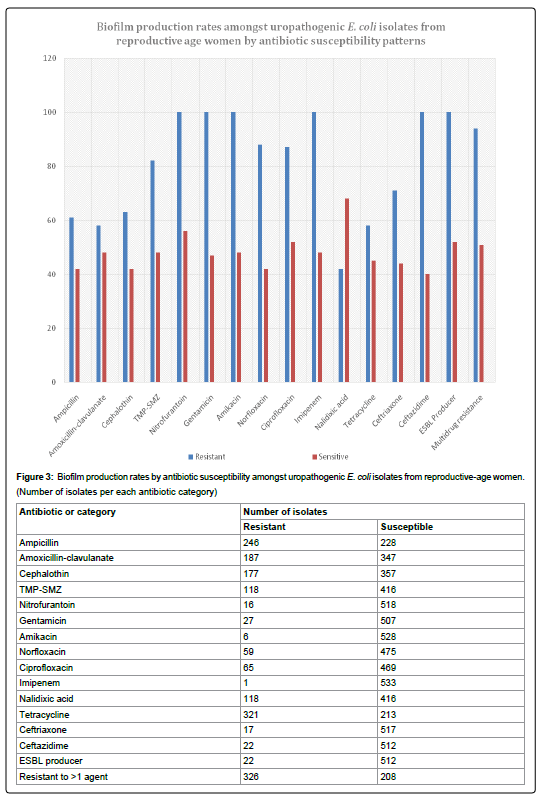 Figure 3: Biofilm production rates by antibiotic susceptibility amongst uropathogenic E. coli isolates from reproductive-age women.
Figure 3: Biofilm production rates by antibiotic susceptibility amongst uropathogenic E. coli isolates from reproductive-age women.
(Number of isolates per each antibiotic category)
View Figure 3
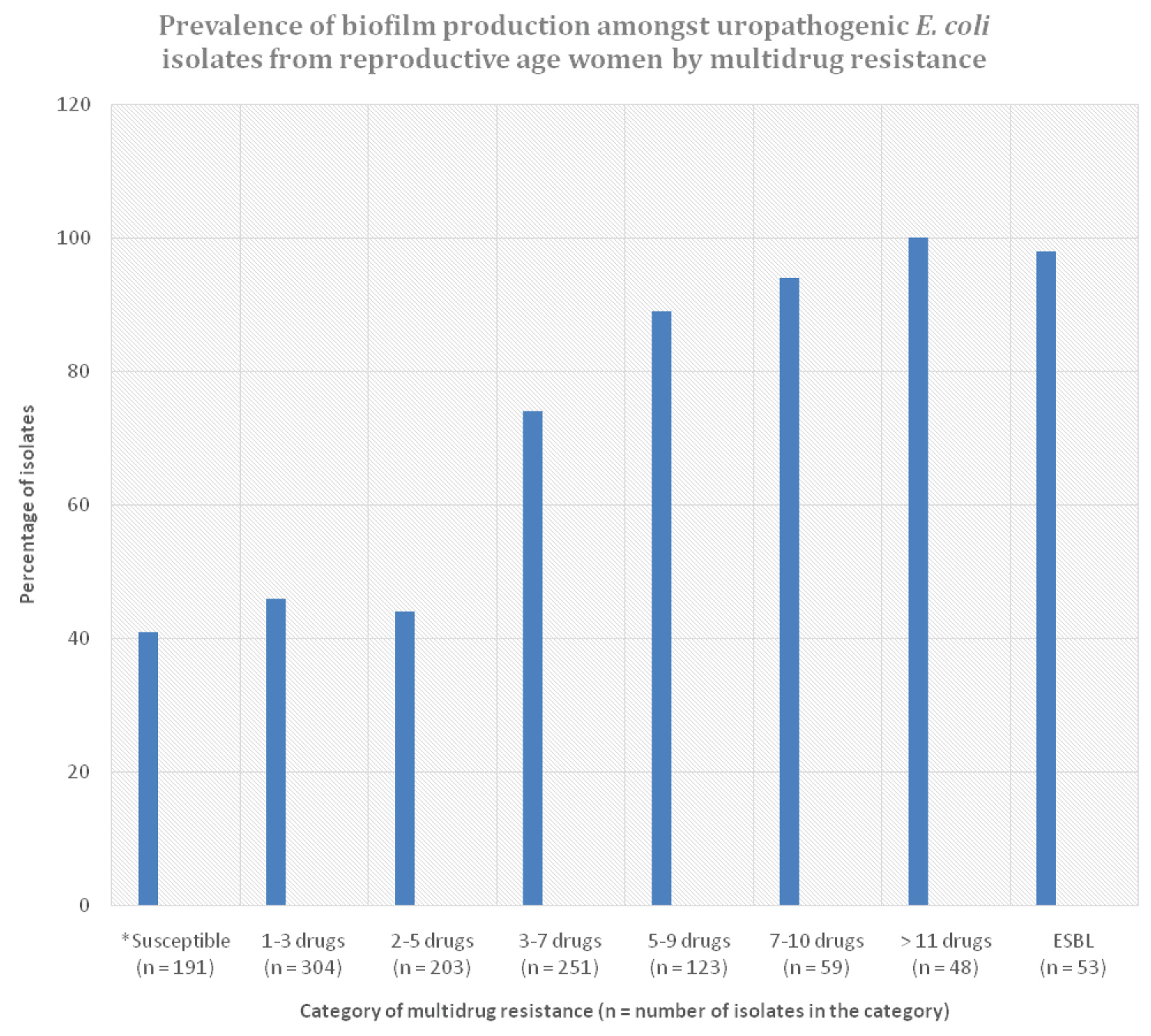 Figure 4: Prevalence of biofilm production by multidrug resistance amongst 534 uropathogenic E. coli isolates from reproductive-age women.
Figure 4: Prevalence of biofilm production by multidrug resistance amongst 534 uropathogenic E. coli isolates from reproductive-age women.
*Susceptible: Susceptible to all antibiotics tested.
View Figure 4
The majority of the cystitis isolates (298, 56%) belonged to group B2, followed by group D (n = 117, 22%). Groups E (9, 2%) and F (9, 2%), were least represented (Figure 5). Likewise, rates of biofilm production by phylogenetic group distribution followed the prevalence levels of the groups, with higher rates of 78% amongst phylogenetic group B2 and 64% for group D. Save for group B1 with a biofilm production rate of 42%, the rest of the remaining groups had an almost similar rate of biofilm production ranging from 25-32%.
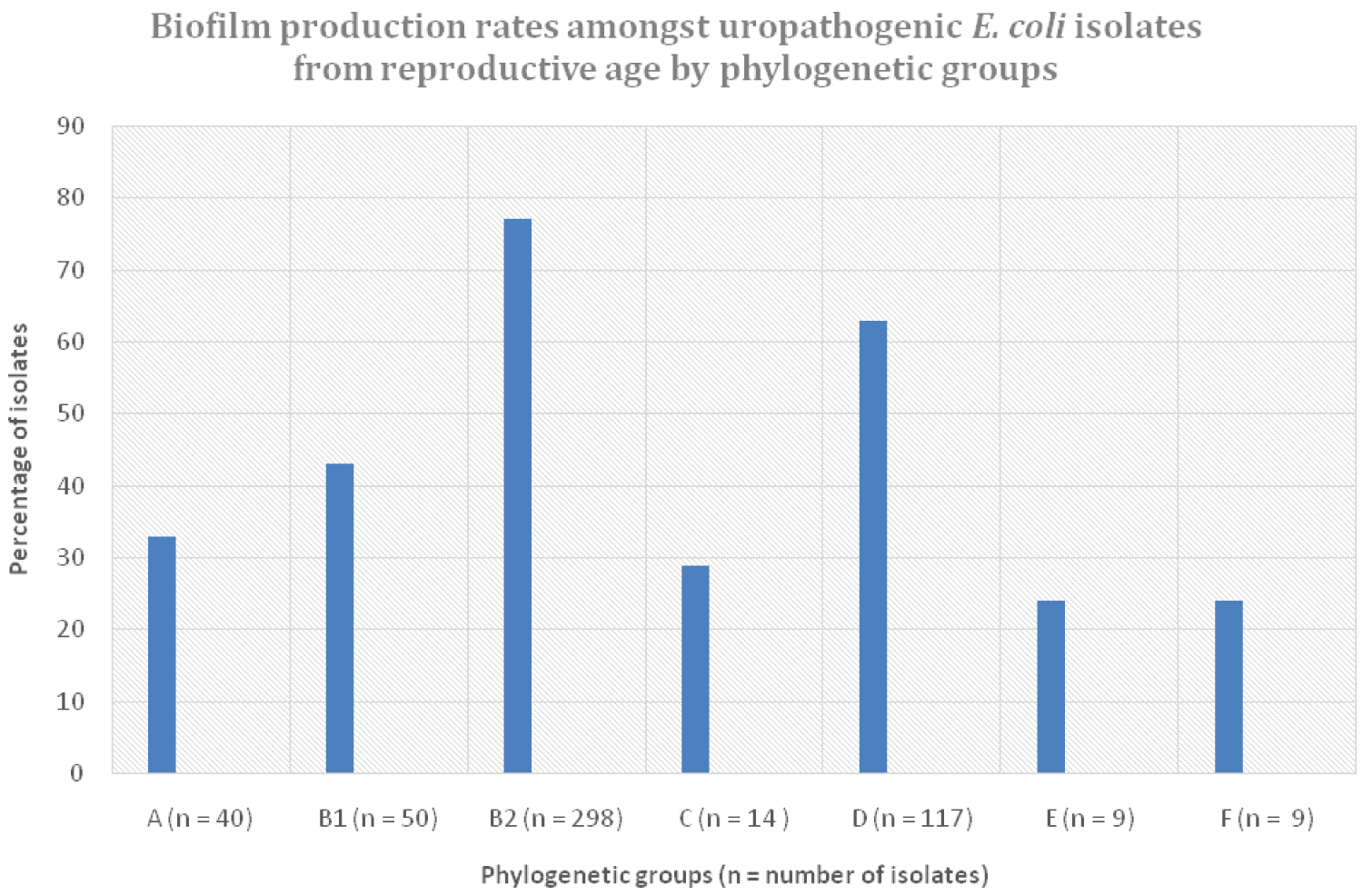 Figure 5: Biofilm production rates amongst uropathogenic E. coli isolates by phylogenetic group, from reproductive-age women.
View Figure 5
Figure 5: Biofilm production rates amongst uropathogenic E. coli isolates by phylogenetic group, from reproductive-age women.
View Figure 5
The aim of this study was to understand the distribution of biofilm production by several host- and non-host factors, including age, recurrence of UTI, phylogenetic group and antibiotic susceptibilities, among uropathogenic E. coli (UPEC) isolates from reproductive age women. We reasoned that understanding how biofilm production is associated with some of these factors would shed light into possible solutions for halting the spread of UTI.
We found out that biofilm production was significantly higher in specific age groups, namely the 21-25 years (51%), 26-30 years (72%), and 31-35 years (60%). Incidentally, these are the age groups in which sexual activity is at highest as per previous studies [14], which is a contributory factor in the transmission of urinary pathogens. Consequently, UTI tends to occur much more frequently in these age groups as previously observed [15], increasing the likelihood of recurrent UTIs and the acquisition of capacity to form biofilms. So, in essence, the association of biofilm production with specific age groups may be indirectly related to increased prevalence of recurrent UTIs in these age groups, as observed in our isolates (data not shown).
There is considerable data in literature on the association between biofilm production and recurrence of UTI, with a significant number of articles indicating a positive correlation between the two [16]. Our findings are in strong agreement with this, as the majority of the recurrent UTI isolates (92%) were biofilm producers, being more than twice the rate in non-recurrent UTIs. This is not a surprising finding as previous studies have concluded that biofilm formation in UPEC is a complicating factor for treatment and recurrence of UTIs [17]. Bacterial strains that have previously caused UTI in another individual, or have remained in the same individual for a long period of time due to inadequate treatment or no treatment, are more likely to have acquired the capacity to produce biofilm, through the well described phenomenon of quorum sensing [18].
The pathogenesis of recurrent UTI is premised on the ability of UPEC to persist in the uroepithelium, which is dependent on the ability to form biofilms, and the presence of various specific VFs [19]. Thus, persistence of bacterial strains in the urogenital tract is most likely multifactorial, hence the present findings must be interpreted with caution as the capacity to produce biofilms per se doesn't necessarily imply that such isolates have the capacity to persist in the urogenital tract. Other VFs are important, including presence of a wide range of adhesins as previously noted [10]. Although we did not characterise most of our isolates for other VFs, the limited available data shows a high prevalence of adhesin VFs amongst some of our recurrent UTI isolates (data not shown). In males, indications are that biofilm formation may give a bacterial strain an increased potential to cause prostatitis, relapse and recurrent UTIs, with several studies observing that 50-70% isolates collected from patients with relapse infections tend to be biofilm producers [17,20]. Unfortunately, in most of these studies, characterization of the isolates for other VFs was not performed, which would have shed more light on any associations.
Having mentioned about the high prevalence of biofilm production amongst the recurrent UTI isolates, it is important to balance this out by analysing the reasons for the relatively high prevalence of biofilm production among non-recurrent UTI isolates of 46%. This could be due to erroneous inclusion of recurrent UTI isolates in this cohort as information about this was self-reported by the study subjects. It is highly possible that recall bias could have resulted in some patients being classified as non-recurrent UTI. It is also plausible that the biofilm producing isolates from the non-recurrent UTI cases may not possess other factors that also contribute to the persistence of the strains in the urogenital tract, as previously suggested.
In agreement to several studies [21], the majority of the UTI cases (69%) in the present study were confined to the outpatient setting compared to the hospital setting, as most UTI cases are relatively clinically mild and hence treated in the outpatient setting. However, when it came to biofilm production, a greater majority (72%) of the isolates from hospitalised patients were biofilm producers compared to 45% for outpatients. This is to be expected as a greater proportion of hospitalised patients would have most likely acquired the infection in the hospital rather than the community environment. According to current European Centre for Disease Prevention and Control (ECD) studies, around 31% of nosocomial infections are UTIs [22]. Several studies have shown that most of bacterial isolates from catheter infections are biofilm producers. It is therefore logical to argue that some of the nosocomial UTIs in our patients may have been transmitted from catheter infections, as evidenced by the fact that between 15% and 25% of hospitalised patients may receive short-term indwelling urinary catheters at one stage during their hospital stay [23]. Furthermore, the hospital environment is more likely to harbour antibiotic resistant isolates, which are known to be associated with biofilm production [24].
Many studies have been carried out to understand the link between antibiotic resistance and biofilm production amongst UPEC from several cohorts, with contrasting conclusions [9]. Some investigations have highlighted that biofilm formation can be induced by low concentration of certain antibiotics [25]. In the present study, a significantly higher proportion of antibiotic resistance isolates (94%) were biofilm producers compared to susceptible ones (51%), and this was pronounced in all the specific antibiotics tested, save for nalidixic acid with opposite results, which confirms previous findings [26]. These findings confirm previous observations in several jurisdictions, suggesting that biofilms are associated with increased resistance to antibiotics [27]. A study by Manuela, et al. reported that biofilm production was significantly associated with fluoroquinolone resistance [28]. However, in another study, no relationship was observed between multi-drug resistance and biofilm formation, which probably is linked to the method used to demonstrate biofilm production [29].
On another note, the difference in the rate of biofilm production between the resistant and susceptible isolates was less marked in three antibiotics, namely ampicillin, augmentin, and tetracycline, which to the best of our knowledge have not been described before. Furthermore, all the isolates resistant to antibiotics in which UPEC resistance is normally very low, including nitrofurantoin, gentamicin, amikacin, imipenem, and ceftazidime, were all biofilm producers as previously noted by other researchers [30], most likely because such isolates are normally multidrug resistant. Overall, these findings demonstrate a strong link between biofilm formation and antibiotic resistance. However, the challenge lies in ascribing biofilm production to specific antibiotics or vice versa. The confounding nature of having an isolate being resistant to more than one antibiotic adds a high level of complexity when interpreting the causal relationship between biofilm production and specific antibiotics. So, it may be very difficult to ascribe the biofilm producing capacity to specific antibiotics.
In an effort to deal with the confounding effect of multiple antibiotics on biofilm production, we studied the distribution of biofilm production by the number of antibiotics an isolate was concurrently resistant to vs. isolates that were susceptible to all antibiotics. We discovered that as the number of antibiotics that an isolate was resistant to increased, so did the capacity to produce biofilms, a phenomenon that has been previously described [31], which seem to suggest that exposure to certain antibiotics may induce or confer the capacity to produce biofilms through a currently unknown mechanism. Indeed, high rates of biofilm production have been observed in multidrug resistant strains in several bacterial species, including Klebsiella pneumoniae in which ESBLs harboring the blaCTX-M gene were biofilm producers [32]. This also agrees with our finding in which an overwhelming majority of the ESBL producers had capacity to produce biofilms. Indeed, it has been demonstrated that biofilm formation and beta-lactamases synergistically contribute for extensive dissemination of multi-drug resistant strains of Gram-negative bacilli. They are responsible for implicating chronicity, persistence, and relapse of infections leading to high morbidity and mortality; thus, posing a serious health crisis [33].
Several studies have demonstrated that low doses of certain antibiotics can induce biofilm formation, indicating that the presence of antibiotics regulates biofilm formation [34]. Some studies have demonstrated a relationship between the acquisition of resistance determinants (specifically resistance to quinolones) and the ability to form biofilm among UPEC isolates [26]. A study involving the antibiotic erythromycin confirmed that sub-MIC of the drug resulted in increase in the expression of biofilm-related genes, including atlE (encoding a major autolysin), fruA (fructose-specific permease), pyrR (pyrimidine regulatory protein), sarA (global regulator) and sigB (sigma factor) [35]. Furthermore, a decrease in biofilm formation has also been directly linked to a decrease of type 1 fimbriae expression [36]. It is also assumed that the biofilm matrix induces antibiotic resistance through several processes, including expression of chromosomally encoded resistant genes, restricting antibiotic diffusion, reduction in bacterial growth rate, and even counteracting the host immunity.
Although UPEC phylogenetic background plays a significant role in virulence, with group B2, and to a less extent Group D, being associated with extraintestinal infections [8], few studies have been carried out to assess the relation between phylogeny and biofilm production which may provide insights needed for establishing strategies for controlling UTIs. Biofilm production was highest (%) in phylogenetic group B2, which incidentally was the most abundant (55%) amongst the isolates. This was followed by group D with 65% being biofilm producers, and also this group was the second most abundant at 22%. These results confirm previous findings worldwide [8], highlighting a relationship between biofilm production and phylogenetic groups B2 and D. As previously argued, it is very challenging to work out how this relationship works as there several other competing factors. For example, multidrug resistance tends to be confined to these phylogenetic groups [37,38] as well as a multiplicity of other VFs, all of which have been linked to biofilm production). We have previously shown that group B2 strains on average carry more VF genes than other groups, and hence argue that probably the phylogeny forms the basis for virulence in UPEC [39]. It would be interesting to compare the rate of biofilm production amongst B2 isolates by multidrug resistance, and infection status to have a better understanding of the relationship. The rate of biofilm production amongst phylogenetic groups normally associated with commensal strains was on average low at about 25%. On combining all data together, majority of strong biofilm producers belonged to phylogenetic group B2, while in group D isolates were found to be moderate biofilm producers. Moreover, phylogenetic group B2 is the most commonly associated with persistent infections.
In conclusion, we demonstrated a high tendency among the UPEC isolates to form a biofilm, specifically by specific age groups, recurrent UTIs, hospitalisation, and phylogenetic groups B2 and D, which appeared linked to the high prevalence of antibiotic resistance amongst the isolates. However, this association is still debatable, and may be influenced by the origin and phylogenetic group of the isolates. Comparing results from other investigations is challenging due to different methodologies used for measuring biofilm production, as well as the patient cohorts from which the isolates originated, resulting in conflicting findings [40].
We acknowledge help from the Charles Sturt University laboratory personnel at the Orange campus.