Multiple hereditary exostoses, Proximal tibiofibular joint, Common peroneal nerve palsy, Proximal fibula resection
MHE: Multiple Hereditary Exostoses; PTFJ: Proximal Tibiofibular Joint; PA: Postero-Anterior; BMRC: British Medical Research Council; CPN: Common Peroneal Nerve; LCL: Lateral Collateral Ligament; BFT: Biceps Femoris Tendon
A 37-year-old male presented with progredient weakness of dorsiflexion in his left ankle since 5 months that was accompanied with sensory deficits in the superficial peroneal nerve area at the dorsal aspect of his left foot. Additionally, the patient reported increasing pain around his left Proximal Tibiofibular Joint (PTFJ). There was a history of known Multiple Hereditary Exostoses (MHE) that was primarily diagnosed with patient's age of 8 years. The Postero-Anterior (PA) radiograph of lower leg showed multiple exostoses around his left tibial head that was accompanied with distinctive synostosis of the PTFJ; and, additionally, a distinctive synostosis of the distal tibiofibular joint was present as well (Figure 1A). On physical examination using the scale of the British Medical Research Council (BMRC, 1978, scale 0-5), the weakness of dorsiflexion in patient's ankle was grade 2. Electrodiagnostic confirmed a demyelinating Common Peroneal Nerve (CPN) palsy, and the enlarged surgical revision was detected. The lesion was surgically exposed through a large longitudinal incision starting distally over the lateral aspect of fibula, passing the popliteal fossa with a vertical incision, and extending up longitudinally on the dorsal aspect of the thigh. After that, the Lateral Collateral Ligament (LCL) and the Biceps Femoris Tendon (BFT) were detached from their insertions at the fibula head and proximal fibula (Figure 1B). Intraoperatively, there was a distinctive entrapment of the CPN including the deep and superficial nerve distally. Then, all three nerves were carefully dissected and the enlarged neurolysis of all three nerves were performed (Figure 1B). After that, the proximal fibula including the tibiofibular synostosis and the PTFJ was excised with length of 10 cm (Figure 1C). Finally, LCL and BFT were reattached with sutures at the periost of tibial head, the resulting bony defect of proximal fibula was filled off, including covering of all 3 peroneal nerves, by transposition of the lateral head of gastrocnemius muscles sutured at the periost of tibial bone as well (Figure 1D), and the large surgical incision could be closured primarily (Figure 1E). Histological examination revealed benign exostoses. The postoperative PA radiograph showed correct articulation in knee joint (Figure 2A). Six months after surgery, there was improvement of sensory deficits and motoric weakness regarding dorsiflexion of foot (BRMC) to grade 4+ (Figure 2B). There was no instability in patient's left knee joint in all directions.
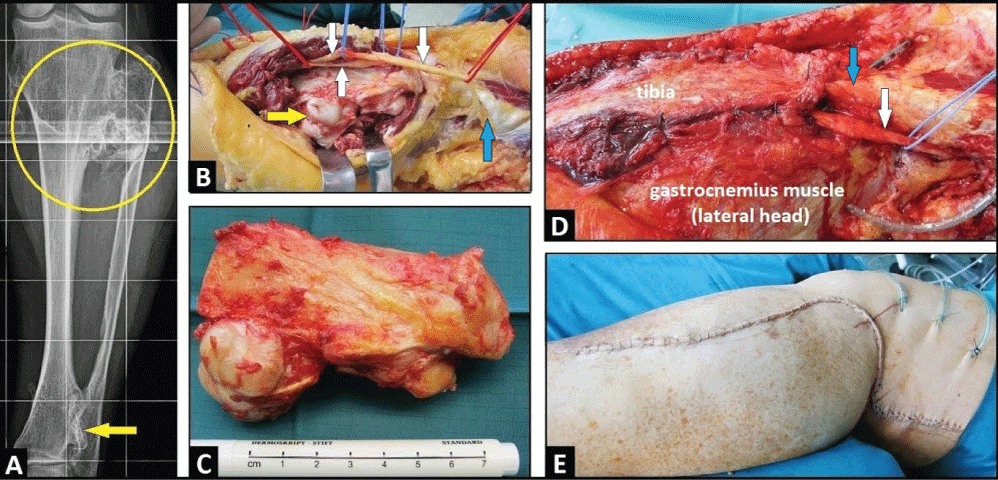 Figure 1: (A) PA radiograph of left lower leg demonstrating multiple exostoses around the proximal third of lower leg and distinctive tibiofibular synostoses (circle), note the additional synostosis of the distal tibiofibular joint (arrow); (B) Clinical photograph showing all three peroneal nerves after its dissections and neurolysis (white arrows) before excision of proximal fibula (yellow arrow), note the detached BFT (blue arrow); (C) Clinical photograph showing the excised proximal fibula including the PTFJ and proximal tibiofibular synostosis with length of 10 cm; (D) Clinical photograph showing filling off the resulting bony defect and covering all three peroneal neves (white arrow) by transposition of the lateral head of gastrocnemius muscle that was sutured at the periost of proximal tibial bone, note that the BFT was reattached at the periost of tibial head (blue arrow); (E) Clinical photograph showing complete closure of surgical incision. View Figure 1
Figure 1: (A) PA radiograph of left lower leg demonstrating multiple exostoses around the proximal third of lower leg and distinctive tibiofibular synostoses (circle), note the additional synostosis of the distal tibiofibular joint (arrow); (B) Clinical photograph showing all three peroneal nerves after its dissections and neurolysis (white arrows) before excision of proximal fibula (yellow arrow), note the detached BFT (blue arrow); (C) Clinical photograph showing the excised proximal fibula including the PTFJ and proximal tibiofibular synostosis with length of 10 cm; (D) Clinical photograph showing filling off the resulting bony defect and covering all three peroneal neves (white arrow) by transposition of the lateral head of gastrocnemius muscle that was sutured at the periost of proximal tibial bone, note that the BFT was reattached at the periost of tibial head (blue arrow); (E) Clinical photograph showing complete closure of surgical incision. View Figure 1
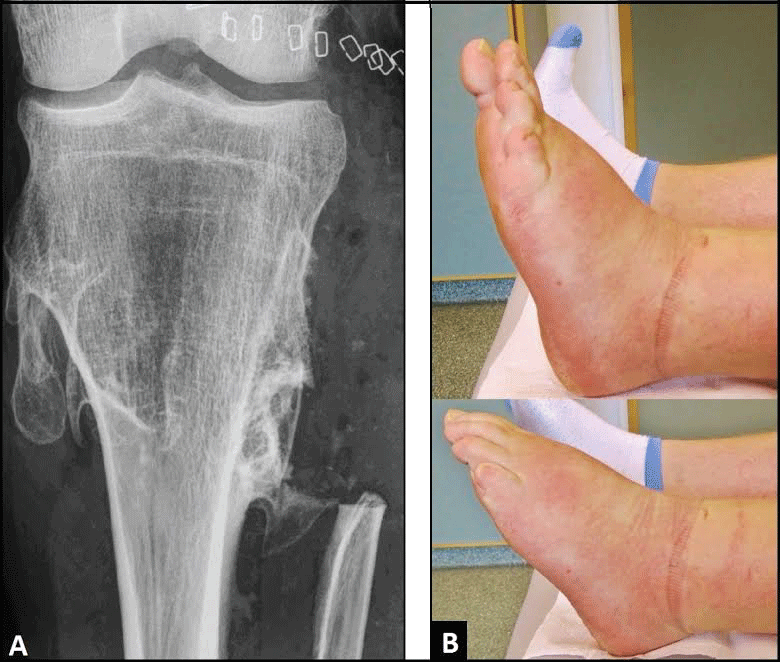 Figure 2: (A) Postoperative PA radiograph demonstrating excision of the proximal fibula, note that there is a correct articulation in knee joint; (B) Clinical photographs six months after surgery demonstrating function of ankle with dorsi- and plantar flexion compared to the unaffected right ankle. View Figure 2
Figure 2: (A) Postoperative PA radiograph demonstrating excision of the proximal fibula, note that there is a correct articulation in knee joint; (B) Clinical photographs six months after surgery demonstrating function of ankle with dorsi- and plantar flexion compared to the unaffected right ankle. View Figure 2
MHE, also known as multiple osteochondromatosis, is a relatively rare autosomal-dominat skeletal disorder that affects approximately one in 50,000 individuals with a slight female predominance in 56% of cases [1,2]. The exostoses typically occur at the arm, forearm, thigh and leg; but they are also commonly found at the pelvis and shoulder [2-4]. The mean age of patients at time of diagnosis is reported to be 5.4 years followed by a mean of 7.3 required surgeries [2]. At time of diagnosis, patients often reported complications such as compression of blood vessels or nerves leading to pain and sensory or motor nerve deficits [5,6]. Larger studies have shown that there is a tendency for transformation in a malignant chondrosarcoma ranging from 0.83% to 7% [7,8], and transformation was observed in patients with a mean age ranging from 29 to 35 years [2,7].
The CPN originates from the sciatic nerve, passes posteriorly and laterally to the biceps femoris muscle in the popliteal fossa, crosses the head of the fibula, and descends to lower leg after dividing in its superficial sensory and deep motor nerve. Most often, CPN palsy occurs at the fibular neck, where the nerve is superficial and vulnerable to direct injury, overstretching, and entrapment. The prognosis of functional recovery in patients with CPN palsy depends on severity of neurological deficit and time of surgical neurolysis. In general, the prognosis for a demyelinating lesion is much more favorable than for an axonal loss lesion [9]. The outcome is usually favorable if surgery is done within 4 months after first presentation of neurological deficits, and less favorable in patients who have neurological symptoms for longer than 12 months [10].
Proximal fibula resection has proven to be a useful and reliable surgical option for treatment of benign or malignant bony tumors [11], and additionally, the proximal fibula is a common donor site for reconstruction of bone loss or joint reconstruction in patients undergoing osteoarticular resections in other anatomic locations [12]. This surgical procedure is generally associated with detachment of LCL and BFT, and in the literature it is still controversial whether the LCL and BFT should be reattached or not. Joint laxity is generally to be considered as predisposition for development of joint osteoarthritis [13,14]. Isolated sectioning of LCL can result in an increase of varus rotation at all angles of knee flexion [15]. The biceps femoris imparts a posteriorly directed force to the proximal tibia and the iliotibial band, leading to ante¬rior stability, thus reducing strain on the anterior cruciate ligament [16]. Although low-grade knee laxity is often unavoidable despite reattachment of LCL and BFT, its reattachment is recommended to prevent or reduce the severity of knee laxity [11,17,18].
The author declares that he has no conflict of interest concerning this article.