Nerve crush injury at focal sites causes rapid muscle atrophy in the connected muscle downstream of the affected motor end plate, and results in profound functional and metabolic deficits. This report characterizes the resultant functional and molecular changes at the post-synapse in an androgen depleted mouse model of reversible nerve injury. Weekly functional phenotyping utilizing Paw Grip Endurance Analysis (PaGE) revealed a significant functional deficit as a result of nerve crush (vs. sham procedure animals). Molecular characterization of the post-synapse after injury (gastrocnemius skeletal muscle) revealed down regulation of key neuromuscular junction genes such as MuSK and Rapsyn. While treatment with Testosterone Propionate (TP) yielded little benefit, this report highlights a Selective Androgen Receptor Modulator (SARM) that accelerates the recovery as evidenced by improved PaGE endpoints. Treatment with the SARM increased the expression of MuSK and AChR-gamma, along with augmented NMJ innervation. Collectively, this report identifies molecular events associated with nerve injury and repair and provides preliminary evidence for the therapeutic use of novel SARMs in such conditions that are associated with nerve injury.
Muscle, Nerve, Nerve crush, Neuromuscular junction, Androgen, SARM
Reversible nerve injury results in significant atrophy in skeletal muscle that receives the post-synaptic motor signals from the injured nerve. The re-formation of the Neuromuscular Junction (NMJ), during the recovery phase of this post-injury event is a dynamic process involving the interplay of several factors in a temporal manner and recapitulates aspects of the early formation of the NMJ during embryonic development. During embryonic development, the nascent NMJ is in a state with acetylcholine receptors distributed throughout the entire muscle fiber and is positioned to receive signals from a range of newly formed network of nerves. The muscle-nerve connection then undergoes a period of pruning, where specific clusters of acetylcholine receptors are selectively activated while the others are lost, resulting in a single dominant innervated muscle fiber with a functional motor endplate [1]. This process is controlled by muscle skeletal receptor tyrosine-protein kinase (MuSK), which responds to the nerve derived signal agrin [2]. MuSK is phosphorylated when exposed to agrin, which leads to a cascade of events that culminates in the formation of the adult NMJ [3].
The MuSK-agrin system is regulated by several proteins that are critical for NMJ formation. These proteins include Docking Protein 7 (Dok-7), a non-catalytic cytoplasmic adaptor protein that reactivates MuSK and low-density Lipoprotein Receptor-Related Protein 4 (LRP4) that binds agrin [4]. Rapsyn, although not directly involved in the MuSK-agrin pathway has also been shown to stabilize Acetylcholine Receptors (AChRs) [5]. An important factor in the maturation of the NMJ is the clustering or accumulation of nicotinic AChRs at the sites of neuromuscular contact near the endplates. Another process is the molecular switch in the composition of AChR subunits from embryonic gamma (γ) to the adult epsilon (ε) sub-types. This switch from the embryonic gamma types to the mature epsilon types occurs between postnatal days five and seven. This timing is similar for the transition of multi-innervated muscle fibers to single innervated fibers [6]. This process of AChR subunit transitions and organization is also observed in the adult muscle in cases of reversible nerve injury.
Upon nerve injury, the nerve begins to de-innervate and lose contact with the muscle fibers. This insult that results in denervation may release neurite outgrowth inhibitory factors that are derived from the insulating axon sheath myelin. One such factor has been identified as reticulon-4 (NOGO-A), which is a membrane bound protein associated with the endoplasmic reticulum [7]. Ultimately, the injury derived NMJ reverts back to an embryonic state, as the muscle begins to lose function, and then begins to recover function as the nerve re-innervates by re-engaging those embryonic processes found during de-novo NMJ formation [8,9].
It is well established that androgens play an important role during development; their many actions include mediating neuromuscular sex differences. Moreover, androgens have been shown to protect against motorneuron death, and display capacity to enhance nerve regeneration following injury [10]. Initial nerve regeneration studies were performed using rodent facial nerve axotomy model [11,12], where androgens exerted dramatic positive effects on the speed of functional recovery [12,13]. Brown, et al. has shown that agonists to the androgen receptor speed up the recovery time after sciatic nerve crush [14]. Furthermore, androgen treatment in the context of peripheral neurotoxicity (using saporin) was shown to be neuroprotective [15].
This study aimed to explore the molecular and functional changes following reversible nerve crush in an Orchidectomized (ORX) mouse model as well as determine the efficacy of androgen receptor agonists. In addition to assessing the effects of Testosterone Propionate (TP), we examined the potential usefulness of a non- Steroidal Selective Androgen Receptor Modulator (SARM). SARMs act as ligands and bind the androgen receptor resulting in tissue selective activity profile which is hypothesized to result from unique conformations induced by binding of the ligand in the C-terminus ligand binding domain of the nuclear hormone receptor [16,17]. In this study we report molecular changes at the post synaptic NMJ after a controlled nerve crush injury which includes a coordinate set of molecular changes in transcripts, such as MuSK, AchR's and Rapsyn. Treatment with a SARM produced a robust acceleration in return to normalization of function which may be mediated by a sub-set of genes involved in the reformation of a functional motor endplate. Immunohistochemical analysis revealed increased NMJ innervation as a result of SARM treatment. Collectively, these point to an accelerated recovery following nerve crush at the postsynapse in the SARM-treated animals.
All animal experiments were approved by the Animal Care and Use Committee of Eli Lilly and Company. ICR mice underwent an Orchidectomy (ORX) at 8-weeks of age at Harlan Laboratories (Indianapolis, IN) and were delivered to Eli Lilly at 10 weeks of age. The treatment and nerve crush procedure in mice initiated 32 weeks post-ORX (40-weeks-old) and remained on treatment for 28 days. Mice were randomized by weight into four groups of six, individually housed in a standard cage with a 12:12-hour light-dark cycle and given ad libitum access to water and food. A second group of ICR underwent a similar protocol, but underwent the treatment and nerve crush procedure at 8 weeks-post ORX (16-weeks-old).
There were four separate groups; Group 1 was the untreated Sham group. These mice were anesthetized and the sciatic nerve was exposed. The wound was glued closed, without the nerve being crushed. Groups 2-4 underwent a unilateral sciatic nerve crush as previously described [18,19]. In brief, these mice were anesthetized and the sciatic nerve was exposed. A hemostat (Fine Science Tools, 13020-12) was modified with a spacer between the handles so the amount of force applied during nerve crush was consistent. Once the nerve was freed from surrounding connective tissue, the jaws of the hemostat were placed around the nerve which was subsequently crushed (holding hemostat jaws closed for 10 seconds). The wound was then glued closed. All mice were subcutaneously dosed with 100 µl of HEC buffer daily (Vehicle; Group 2), 100 µl of a selective androgen receptor modulator (SARM; synthesized at Eli Lilly & Co., and described in World patent application WO2007 002181) [20] at a concentration of 2 mg/kg/day, or 100 µl of testosterone propionate (TP; Sigma T1875) at a concentration of 14 mg/kg/week (starting one day prior to nerve crush procedure). Animals were sacrificed 28 days after undergoing a unilateral sciatic nerve crush injury (see Figure 1 for overview of timeline). At the time of sacrifice the perineal muscles (levator ani and bulbar cavernosus) were isolated as a positive control for a typical anabolic androgen response [21,22].
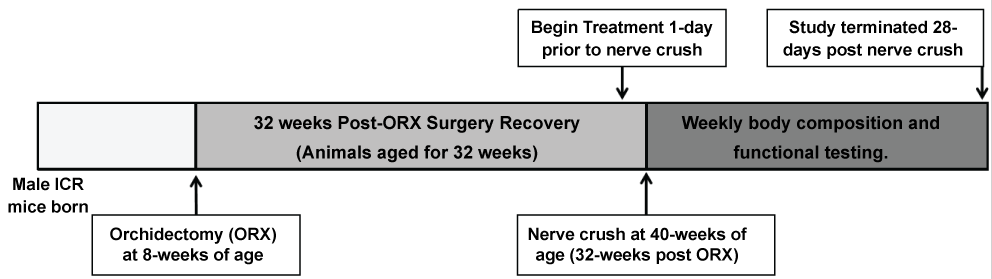 Figure 1: Study timeline. View Figure 1
Figure 1: Study timeline. View Figure 1
Total body weight, muscle and fat mass for each mouse was determined weekly using an EchoMRI-100 scanner (Echo MRI, Houston, TX).
Muscle strength and endurance was measured using the Paw Grip Endurance (PaGE) Test [23-27]. Mice were placed right side up on 1 cm2 grid, the grid was shaken and inverted approximately 45 cm above a padded surface. The animals were given 3 trials to keep their hindlimb paws on the screen for 120 seconds. Their latency to release each hindlimb paw was determined by averaging the release time for each leg from all 3 trials. Leg 1 represented that on the sciatic nerve crushed side, while leg 2 was that on the contralateral (non-nerve crushed) side.
At the time of necropsy, muscles were weighed, flash frozen in liquid nitrogen and stored at -80 °C. Tissue was placed in Trizol and homogenized using fast- prep-24 tubes and homogenizer (MP Biomedicals, Santa Ana, CA). Gastrocnemius RNA extraction was done using the Perfect Pure RNA isolation kit (5 Prime, Gaithersburg, MD). The extracted RNA was quantified using a NanoDrop® Spectrophotometer (Thermo Scientific, Rockford, IL).
Single stranded RNA was converted to cDNA using a High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Grand Island, NY). TaqMan gene analysis was performed on the generated cDNA. The following NMJ related genes were investigated using Taqman probes purchased from Applied Biosystems: Reticulon 4 (NOGO-A; Mm00445861_m1), MuSK (Mm01346929_m1), Dok7 (Mm00554697_m1), Rapsyn (Mm00485539_m1), AChR subunits epsilon (Mm00437411_m1) and gamma (Mm00437419_m1). Each gene was normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH; Mm99999915_g1).
Muscles were dissected and immersed in 0.25 M sucrose for 1 h at 48 °C, then in 0.5 M sucrose for 45 min, and finally in 1.5 M sucrose for 30 min [28]. The muscles were embedded at optimal cutting temperature compound (OCT; Sakura Finetek, Torrance, California) and stored at -80 °C. Tissues were sectioned on a Leica CM3050S cryostat. Sections were fixed with 2% paraformaldehyde, then stained with mouse anti synaptophysin (Dako North America, Carpinteria, California) and rabbit anti-neurofilament-L (Abcam Inc., Cambridge, Massachusetts) to label presynaptic terminals and motorneuron axons, respectively [29]. Postsynaptic terminals containing acetylcholine receptor (AChR) clusters were labeled with a-bungarotoxin (a-BTX; Molecular Probes, Grand Island, New York). Innervated neuromuscular junctions (NMJs) were defined as the histologic colocalization of the presynaptic marker synaptophysin and the AChR marker aBTX [30]. The percentage of innervated NMJs from each diet group was determined by counting at least 100 NMJs from each animal in each diet group [29-31].
Data are represented as the mean and + Standard Error of the Mean (SEM). Statistical analysis was performed using the calculated mean, SEM and N for each group (Graph Pad Prism). Statistical significance for tissue weight, gene expression, and NMJ innervation was determined by one-way ANOVA followed by a Dunnett’s Multiple Comparisons Test. Statistical significance for body composition and PaGE data was determined by two-way ANOVA followed by a Dunnett's Multiple Comparisons Test.
Adult Orchidectomized (ORX) mice underwent nerve crush or sham procedure following ORX surgery recovery period. One day prior to nerve crush, and for the remainder of study, mice were treated with either, 1) Vehicle (hydroxyethycellulose; HEC), 2) Testosterone propionate (TP; 14 mg/kg/wk), or 3) A selective androgen receptor modulator (SARM; 2 mg/kg/day). Specific perineal muscles in the body, such as the Bulbocavernosus (BC) and the levator ani (LA), are particularly androgen sensitive and will significantly atrophy upon loss of androgen signaling (e.g., ORX setting), and hypertrophy in the presence of increased circulating androgen signaling [21,22]. These tissues provide a Pharmacodynamic (PD) marker for confirming the anabolic effects of TP and SARM treatments. The BC and LA from each group were removed and weighed upon study termination (28 days of treatment), and were significantly larger than muscles from vehicle and sham groups (Figure 2D and Figure 2E). These results indicate that the treatment with TP and SARM had shown the expected anabolic effect.
As expected, there were no differences in body weight or composition between animals that underwent the nerve crush versus the sham procedure. However, the gastrocnemius muscles from the nerve crushed leg were significantly smaller than sham procedure muscle (Figure 2F), confirming effectiveness of the nerve crush. While target engagement of TP and SARM was confirmed (via LA & BC weights; Figure 2D and Figure 2E), there were no changes observed in the body weight, fat mass or lean mass as a result of compound treatment (Figure 2A, Figure 2B and Figure 2C). However, consistent with the responsiveness of the perineal LA & BC muscles, the non-crushed (contralateral) gastrocnemius muscles from TP treated mice were significantly larger than vehicle treated. The lack of gastrocnemius response to SARM (in either leg) was not surprising as these muscles often will not display robust hypertrophy in response to short-term androgen signaling.
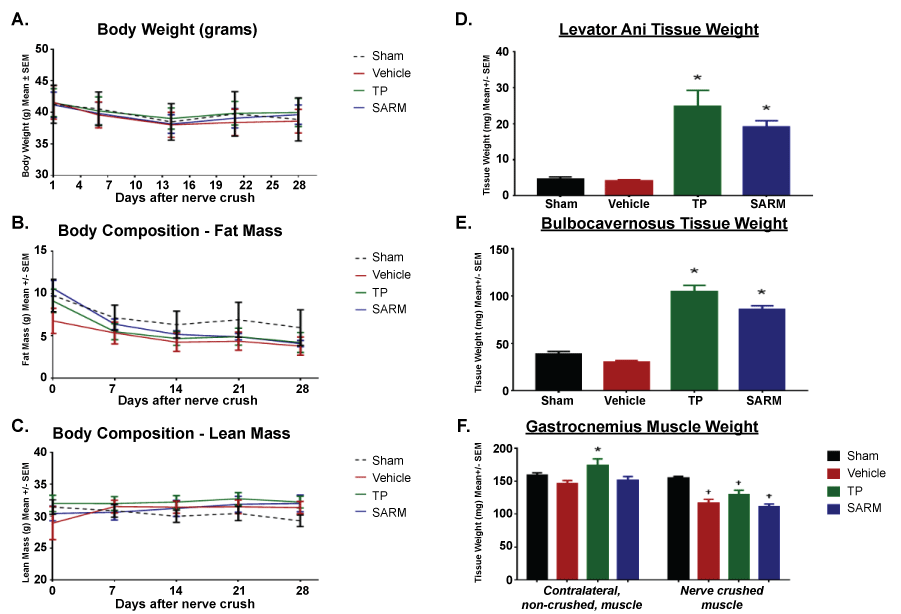 Figure 2: Body Composition and Tissue Weight Analysis A) Body weight; B) Fat Mass; C) Lean Mass were measured weekly. Upon study termination (28 days post nerve crush); D) Levator Ani; E) Bulbocvernosus; F) Gastrocnemius muscles were isolated and weighed. N = 6/group. Data are means ± SEM. *p > 0.05 vs. Vehicle and †p < 0.05 vs. Sham, as determined by ANOVA and Dunnett's post hoc analysis. TP: Testosterone Propionate; SARM: Selective Androgen Receptor Modulator. View Figure 2
Figure 2: Body Composition and Tissue Weight Analysis A) Body weight; B) Fat Mass; C) Lean Mass were measured weekly. Upon study termination (28 days post nerve crush); D) Levator Ani; E) Bulbocvernosus; F) Gastrocnemius muscles were isolated and weighed. N = 6/group. Data are means ± SEM. *p > 0.05 vs. Vehicle and †p < 0.05 vs. Sham, as determined by ANOVA and Dunnett's post hoc analysis. TP: Testosterone Propionate; SARM: Selective Androgen Receptor Modulator. View Figure 2
The relatively short duration of treatment applied in the context of a nerve crush injury did not allow for the phenotypic changes in body composition as measured by EchoMRI. However the more valuable measurement of functional recovery using the Paw Grip Endurance (PaGE) test was investigated and provided effective characterization (Figure 3A and Figure 3B). Immediately following the nerve crush procedure, functional performance, defined as a measure of latency for leg release in the nerve crushed leg (leg 1), was completely lost in all treatment groups. Likewise, the contralateral, non-nerve crushed leg (leg 2), also displayed a decreased latency for leg release, likely a result of the inability to maintain a grip once the nerve crushed leg released its hold. This functional deficit was maintained in all treatment groups until day 14, after which the groups began improving in a treatment dependent manner. At 21 days post-nerve crush, the SARM treated animals showed significantly greater functional capacity with increased latency for leg release in both the nerve crushed and contralateral leg when compared to the vehicle treated nerve crushed animals. By 28 days post-nerve crush, the SARM treated mice displayed significantly greater latency for leg release versus the vehicle nerve crush group, and matched the functional capacity of the Sham group. Somewhat surprising was the lack of efficacy in the PaGE test in the TP treated animals. For the entirety of the 28-day time course, these animals treated with TP tracked with, or were slightly worse, though not significant, than the vehicle group. Collectively, these results suggested that the SARM treatment may have a greater effect in the recovery and repair of the neuromuscular junction compared to TP. In contrast, the anabolic responses seen at the perineal muscles (LA and BC) were comparable for both TP and SARM, and were both significantly increased compared to vehicle control.
 Figure 3: Paw Grip Endurance Analysis (PaGE) in Mice. Weekly PaGE analysis was performed as displayed as latency for leg release for the A) Leg 1 (Nerve crushed) and B) Leg 2 (contralateral, non-crushed). N = 6/group. Data are means ± SEM. *p > 0.05 Sham vs. Vehicle and **p < 0.05 SARM vs. Vehicle, as determined by ANOVA and Dunnett's post hoc analysis at individual time points. TP: Testosterone Propionate; SARM: Selective Androgen Receptor Modulator. View Figure 3
Figure 3: Paw Grip Endurance Analysis (PaGE) in Mice. Weekly PaGE analysis was performed as displayed as latency for leg release for the A) Leg 1 (Nerve crushed) and B) Leg 2 (contralateral, non-crushed). N = 6/group. Data are means ± SEM. *p > 0.05 Sham vs. Vehicle and **p < 0.05 SARM vs. Vehicle, as determined by ANOVA and Dunnett's post hoc analysis at individual time points. TP: Testosterone Propionate; SARM: Selective Androgen Receptor Modulator. View Figure 3
In order to better understand the findings using the function PaGE readout, gene expression analysis on the crushed gastrocnemius muscle was performed (Figure 4). Focus was placed on several key genes involved in the neuromuscular junction. In muscles taken at study end (Day 28 post-nerve crush), AChR-ε levels were not different between any of the compound treatment groups or versus sham. However, the embryonic type AChR-γ subunit (which ultimately transitions to the mature AChR-ε subunit) was significantly increased in all nerve-crush muscles versus the sham. Likewise, SARM treatment significantly augmented this increase in AChR-γ expression versus vehicle, suggesting increased signaling for creation and clustering of NMJs. This process of NMJ clustering is dependent on scaffolding proteins including Dok7, MuSK and Rapsyn. While no differences were observed in Dok7 expression as a result of nerve crush or treatment, a noticeable decline in MuSK expression was observed as a result of nerve crush when comparing sham versus vehicle groups. Consistent with the AChR-γ expression, SARM treatment led to a significant increase in MuSK expression versus vehicle, lending further support towards improved signaling mediated by SARM for increased creation and clustering of NMJs following nerve crush insult. Rapsyn, another key player in assists in the scaffolding of AChR's, was significantly decreased as a result of nerve crush and unlike AChR and MuSK, was not altered with treatment. Finally, Rtn4 (also known as NOGO), which acts to inhibit neuronal outgrowth was not altered in either direction as a result of nerve crush or treatment at day 28 after injury. Together, the gene data findings suggest a positive effect of SARM treatment in the creation and clustering of NMJs following nerve crush injury.
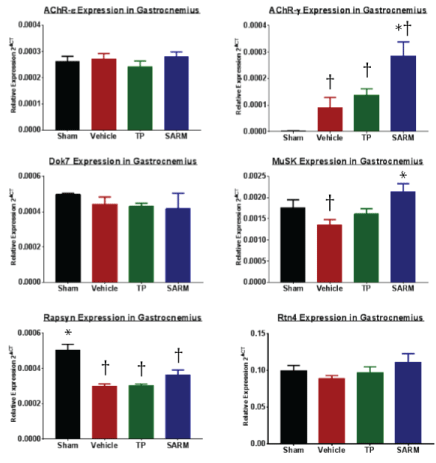 Figure 4: Gastrocnemius muscle gene expression. Gastrocnemius muscles from Leg 1 (Nerve crushed) were isolated upon study termination (28 days post nerve crush). Expression of acetylcholine receptor epsilon (AChR-ε), Acetylcholine Receptor Gamma (AChR-γ), Docking Protein 7 (Dok7), Muscle Associated Receptor Tyrosine Kinase (MuSK), Rapsyn, and Reticulon-4 (RTN4) were measured and normalized to GAPDH (displayed as relative expression). N = 6/group. Data are means ± SEM. *p > 0.05 vs. Vehicle and †p < 0.05 vs. Sham, as determined by ANOVA and Dunnett's post hoc analysis. TP: Testosterone Propionate; SARM: Selective Androgen Receptor Modulator. View Figure 4
Figure 4: Gastrocnemius muscle gene expression. Gastrocnemius muscles from Leg 1 (Nerve crushed) were isolated upon study termination (28 days post nerve crush). Expression of acetylcholine receptor epsilon (AChR-ε), Acetylcholine Receptor Gamma (AChR-γ), Docking Protein 7 (Dok7), Muscle Associated Receptor Tyrosine Kinase (MuSK), Rapsyn, and Reticulon-4 (RTN4) were measured and normalized to GAPDH (displayed as relative expression). N = 6/group. Data are means ± SEM. *p > 0.05 vs. Vehicle and †p < 0.05 vs. Sham, as determined by ANOVA and Dunnett's post hoc analysis. TP: Testosterone Propionate; SARM: Selective Androgen Receptor Modulator. View Figure 4
To fully understand the role of SARM on the NJM, immunohistochemical analysis was performed on the gastrocnemius muscles (from mice undergoing nerve crush 8 weeks post-ORX). Figure 5A presents a representative image in which both innervated and deinnervated NMJs are stained (synaptophysin and neurofilament-L in green and a-bungarotoxin in red). At study termination, the vehicle treated nerve crush mice displayed a significant reduction in innervation (as a % of Sham tissues). Notably, muscles from SARM treated mice displayed significantly greater innervation (vs. Vehicle; Figure 5B). These results corroborate the molecular and functional changes observed.
 Figure 5: Gastrocnemius innervation. A) Representative photomicrographs of an innervated and deinnervated neuromuscular junction in gastrocnemius muscles from Leg 1 (Nerve crushed) stained with synaptophysin (SNP) and Neurofilament-L (NF-L) in green and α-bungarotoxin (α-Btx; label acetylcholine receptors) in red; B) Innervation of gastrocnemius muscles (nerve crushed) measured at study termination. N = 6/group. Data are means ± SEM. *p > 0.05 vs. Vehicle and †p < 0.05 vs. Sham, as determined by ANOVA and Dunnett's post hoc analysis. TP: Testosterone Propionate; SARM: Selective Androgen Receptor Modulator. View Figure 5
Figure 5: Gastrocnemius innervation. A) Representative photomicrographs of an innervated and deinnervated neuromuscular junction in gastrocnemius muscles from Leg 1 (Nerve crushed) stained with synaptophysin (SNP) and Neurofilament-L (NF-L) in green and α-bungarotoxin (α-Btx; label acetylcholine receptors) in red; B) Innervation of gastrocnemius muscles (nerve crushed) measured at study termination. N = 6/group. Data are means ± SEM. *p > 0.05 vs. Vehicle and †p < 0.05 vs. Sham, as determined by ANOVA and Dunnett's post hoc analysis. TP: Testosterone Propionate; SARM: Selective Androgen Receptor Modulator. View Figure 5
The role that androgen plays in regulating adult skeletal muscle mass is well-defined; agonists to the androgen receptor (e.g., testosterone, SARMs) act as anabolic agents and result in muscle hypertrophy [32-34]. In addition, androgen receptor agonists have shown activities as protective, as well as regenerative, and have been considered as therapeutic options in cases of muscle insult (e.g., toxin insult, freeze insult, disuse atrophy) [35-38]. A component of skeletal muscle in such injury situations, whether direct or indirect, often involves nerve denervation. Re-innervation of the nerve can accelerate the functional recovery process and shorten the timeline towards normalizing muscle function. The factors that determine the regenerative ability of an injured neuron are known to include severity and proximity of the injury to the cell body [39], and their interplay with therapeutic agents can augment re-innervation. Androgens have been shown to be effective agents at enhancing nerve regeneration/innervation, though the specific nerve injuries and resultant methods of defining regeneration by androgens (e.g., function, molecular, innervation) show varied effectiveness [10,12-14,40]. Likewise, the mechanism(s) by which androgen's positive effects are driven are not completely understood. This study allowed for the exploration of potential nerve regeneration, measured molecularly and functionally, by androgen receptor activation via two distinct agonists, testosterone propionate or a Selective Androgen Receptor Modulator (SARM).
The Paw Grip Endurance (PaGE) test is an established functional measurement method [26,27], as evidenced in this study with the loss of PaGE activity in animals post nerve crush vs. sham animals. The efficacy of SARM treatment became maximally evident by day 21 (post-nerve crush) and maintained its superior therapeutic effectiveness until study end vs. both vehicle and TP. Notably, the contralateral (non-crushed) leg also displayed a functional deficit post-nerve crush; though this was simply an inability of the animals to hold body weight with a single hind paw. However, as noticed in all groups, as the crushed leg began to regain function, the contralateral leg was able to maintain grip for a longer period of time, as evidence by the increased latency for leg release.
The ability of SARM treatment to completely rescue the nerve-crush induced functional loss by day 28 can be explained, at least in part, through the measured transcript changes at the NMJ. The observed transcript changes seemed to resemble the activity occurring during embryonic NMJ formation [41,42]. Expression of MuSK, a protein critical for NMJ formation, trended down as a result of nerve-crush, while SARM treatment was able to significantly increase MuSK expression versus vehicle. The expression of Rapsyn, a protein shown to stabilize acetylcholine receptors during NMJ (re)-formation [5], was significantly downregulated as a result of nerve crush. However, unlike MuSK, neither androgen agonist modulated Rapsyn expression. Consistent with MuSK expression, the acetylcholine receptor gamma (AChR-γ) mRNA expression levels were increased with SARM treatment. The AChR-γ subunit is typically observed in the embryonic setting though is also observed in settings of regeneration. In both embryonic development and regeneration, the gamma subunit is ultimately replaced by adult acetylcholine receptor epsilon (AChR-ε) subunit through a process of endplate shifting from multi to single-axons [6]. Interestingly, there were no observed differences in AChR-ε expression, suggesting by day 28, a steady state of AChR-ε expression had been reached. However, the AChR-γ and MuSK modulation (vs. sham and upregulated for SARM versus vehicle) on day 28 suggests that an ongoing, dynamic, signaling process for NMJ rebuilding remains present. Together with the function data, this molecular data indicates that regeneration process following nerve crush insult can be augmented with specific pharmacological treatment.
It is likely that the nerve regeneration process is a dynamic event whereby denervation and re-innervation happens repeatedly and simultaneously, and not necessarily in a linear fashion. The healthy NMJ is constantly turning over as it reforms and modifies. A major component of the NMJ's dynamic nature is the process of AChR removal and addition called acetylcholine receptor turnover [43]. Interplay between acetylcholine receptor density and acetylcholinesterase controls the efficacy of neuromuscular junctions. In the nerve crushed setting, with the connection between the nerve and the muscle lost, the system's response is organized, as displayed by the steady state AChR-ε by day 28. However, the increases in AChR-γ (embryonic subunit) could potentially be a compensation method as part of the re- innervation process.
Indeed, SARM treated muscles displayed increased innervation compared to the vehicle group as measured by immunohistochemistry. The role of androgen signaling modulating the NMJ has been established, with several studies showing in castrated models, reduction of muscular activity and atrophy of muscle were accompanied by adaptations of the neuromuscular junctions [44,45]. Furthermore, the beneficial effects of androgen signaling in various nerve crush models (e.g., facial and sciatic) has been described [13,46-48]. The innervation results described herein are important as they pull together a strong set of molecular and functional data to highlight the potential efficacy of the SARM molecule. However, important future experiments should aim to investigate the regeneration process at earlier time points in order to understand the dynamic nature of this event.
While this study highlighted efficacy of the SARM treatment, interestingly, TP treatment did not result in significant alterations in function, and lacked the connected robust molecular signature. The study design was organized so that treatment began prior to nerve injury in order to get therapy on board during the important temporal window described by others. Kujawa & Jones had previously shown that the first week following injury represented the “critical period” [47], and moreover Tanzer and Jones subsequently showed the importance within the first 6-hours of injury [46]. So our lack of T efficacy cannot by traced to dose timing, and the positive changes seen at the LA muscle clearly confirmed target engagement with TP. One possibility for lack of efficacy could be route of administration. Swallow, et al. previously described a model of nerve injury whereby injected testosterone was able to enhance axon regeneration, but lacked benefit on functional recovery [48]. However using a sustained-release testosterone pellet, Kujawa, et al. was able to show functional recovery following facial nerve axotomy, while the injected T in this study lacked functional efficacy [13]. The superior efficacy of sustained-release test implants has also been shown when comparing nerve injury recovery in normal vs castrated + implant rats; where the castrated + implant displayed greater recovery from an axotomy-induced hind limb paralysis [49]. Future studies in the mouse will be needed to gain understanding of the impact of the route of administration (injected vs. implant) for testosterone, as well as recognizing the novel applications for the growing field of SARMs.
In addition, while the PaGE test proved a valuable functional output, it does provide a relatively holistic view of musculoskeletal function. Thus, additional future studies should aim to further understand muscle performance associated with the nerve crush and treatments including weight bearing (footprint analysis) and muscle stimulation. While each test has definite benefits, with each additional function test, there remain caveats, e.g., the quantitative analysis of nerve functions through footprint measurements can be compromised by the variations in print length parameters with gait velocity or smearing of prints due to the dragging of tail [50,51]. While a more muscle specific technique would also prove valuable, because of the nerve crush, in situ/vivo twitch response measurements [52] are not feasible as they require nerve stimulation. However, an ex-vivo muscle stimulation protocol would be an important study to understand the direct role of muscle motor endplates and force generation [53]. In addition, future studies will need to consider the inflammatory response associated with nerve crush. Neural-immune interactions are known to occur in response to disease and/or trauma of the nervous and immune system [54]. Immune activation (i.e., inflammation) and the subsequent release of immune mediators into the peripheral nervous system play a critical role during nerve degeneration and regeneration. Various peripheral immune cells, including mast cells, macrophages, and lymphocytes, reside in peripheral nerves and/or are recruited to sites of peripheral nerve injury [55-57].
In conclusion, this study explored a nerve injury model to study both the molecular and functional changes after sciatic nerve crush in ICR mice. Comparing a treatment regimen of testosterone versus a Selective Androgen Receptor Modulator (SARM) in the nerve injury model provided evidence for SARM's therapeutic potential. SARM treatment was able to rescue the function effects of nerve crush. The molecular and immunohistochemical changes appear to correspond with events occurring in embryonic NMJ development and SARM's efficacy, suggesting they are modulated by androgen regulation.
All authors are current or past employees of Eli Lilly and Company; all research was funded and conducted by Eli Lilly and Company.
All animal experiments were approved by the Animal Care and Use Committee of Eli Lilly and Company.