Several diseases can be monitored by assessing the variation of specific ion concentration in body fluids. This study proposes an alternative analysis, based on the X-ray fluorescence spectrometry technique, for the investigation of iron (Fe concentration determination) in whole blood of the dystrophic animal models. We performed blood analyses from three different strains with Muscular Dystrophy (Dmdmdx/J, SJL/J and A/J) and from healthy (C57BL/6J) mice strain adopted as the control group. The results emphasize that iron levels in whole blood male samples of SJL/J and A/J are significantly higher. Related to the gender for Dmdmdx/J and SJL/J mouse strains the comparison emphasizes that iron level increase for males. The study of Fe behavior in whole blood mice samples with muscular dystrophy contributes to its diagnosis, providing a better understanding of this disease.
Progressive muscular dystrophy, Animal model, Iron, Blood
The Progressive Muscular Dystrophies (PMDs) are genetic diseases that affect all muscular groups. They have a hereditary character and there is no cure. Muscle weakness, cardiac and respiratory problems are the main characteristics of this genetic disease [1]. PMDs are characterized by irreversible progressive degeneration of the skeletal muscle. More than thirty different forms of muscular dystrophy have been identified with different levels of complexity. However, the affected muscles may be different depending on the PMD type [2-4]. There are several types of muscular dystrophies that differ in severity, onset, and genetic causes; the Duchenne Muscular Dystrophy (DMD) is the most aggressive and prevalent type. The estimated prevalence of DMD is 1 in every 7,250 males aged 5-24 years, and the total time of diagnosis is 2.5 years [5]. At the age 2½-years-old the first signs or symptoms are noticed by a parent or caregiver. By the age of 3½-years-old the child is taken to a pediatrician or family practitioner for evaluation and at 5 is diagnosed with DMD based on muscle biopsy or DNA test [6,7]. In general, only males are affected and approximately 10% of female carriers can show some disease manifestations. The disorder is caused by a mutation in the dystrophin gene, located in humans on the X chromosome. Unlike most genes, which come in pairs in both sexes and stay active throughout life, in males there is only one X chromosome: as in the female there are two X chromosomes, if one of them has the defective gene, the other X chromosome will guarantee good functioning muscles, i.e. the female will carry the DMD gene but may or may not develop this disease. However, this does not occur with males who only have one X chromosome. For these reasons this disorder usually affects more boys than girls. Nowadays, in Brazil, the PMDs has been investigated at IPEN, Biosciences Institute and Butantan Institute (Research Centers Research Centers at São city) using dystrophic mice strains: Dmdmdx/J characterized by dystrophin protein deficiency (higher incidence), and SJL/J and A/J, both with dyspherlin protein deficiency. Concentrations of inorganic elements of clinical and nutritional relevance (Ca, Cl, I, K, Mg, Na, P and S) were determined in biological tissue of these species aiming to increase the understanding the role of these element interactions in normal and disturbed metabolism [8-10]. In these investigations, Neutron Activation Analyses Technique (NAA) was adopted due the accessibility that can be associated comparatively to conventional procedure performed in serum or plasma (using at least a 1.0 mL of these body fluids [11]), which requires the sacrifice of this small animal model (the total body blood in mice is around 1.0-1.2 mL). The NAA facilities include the use of whole blood (200 µL) an important condition when the biological material is scarce, simplifications in sample preparation (whole blood pipette in paper) as well as multi-elemental analysis [12,13]. However, for the Fe concentration determination this procedure is not appropriated. It takes several hours of neutron irradiation using a nuclear reactor and, usually, this damages the sample (paper is destroyed). Now, we intend to show that using X-Ray Fluorescence Spectrometry Technique (XRF) [14] the measurements can be performed in few minutes for whole blood Fe determination. In this study, we intend to determine the Fe range in whole blood samples of dystrophic mice strains (Dmdmdx/J, SJL/J and A/J) and in healthy mice strains (C57BL/6J), adopted as the control group, to check differences among these groups. The knowledge of the Fe concentration contributes to studying the behavior of PMD diseases in blood.
The animal models, Dmdmdx/J (n = 18), SJL/J (n = 12), A/J (n = 6) and C57BL/6J (n = 24) of adult mice strain (2-months-old), all came from local breeding facilities (Bioscience and Butantan Research Centers and further inbred at IPEN-CNEN/SP (both located at São Paulo city, Brazil) starting from genitors of the Jackson Laboratory (Maine, USA) [15]. Details of the number of samples collected by gender are presented in Table 1. The whole blood samples (< 200 microliters) were collected by the retro-orbital venous plexus. The term whole blood refers to solid (cells) and liquid (plasma) components in blood. Aliquots of 50 (± 0.5%) microliters (in triplicate samples) of whole blood were transferred to the Whatman filter paper (type 41) (Figure 1a). This sample preparation procedure was adopted because it reduces, significantly, the quantity of whole blood to be collected minimizing the stress in this small animal model. All the samples were collected according to the rules approved by Animal Research Ethics Committee (087/99).
Table 1: Whole blood Fe concentration from control and dystrophic groups using XRF spectrometry. View Table 1
 Figure 1a: Blood sample dimension (left) and (right). View Figure 1a
Figure 1a: Blood sample dimension (left) and (right). View Figure 1a
The Energy Dispersive X-Ray Fluorescence Technique (EDXRF) is based on the material excitation by X-rays from an X-ray tube. The beam interaction (X-ray) with the material (in this specific case, blood sample) causes it to be excited and, in the process of de-excitation X-rays characteristic of the material are emitted [16]. Using specific detectors for X-ray detection (Semiconductor Type) it is possible to detect and quantify the chemical elements present in the analyzed material.
The XRF analysis was performed using portable X-ray spectrometer from Amptek (Figure 1b); model X-123 SDD with Au X-ray mini-tube. The characteristic X-ray fluorescent intensity of Fe kα line (6.34 keV) was measured with a Si Drift detector (25 mm2 × 500 μm) with is window (12.5 μm). The blood samples (Figure 1) were irradiated for 200 s using 30 kV and 5 microampere excitations. Figure 2 shows the whole blood spectrum obtained with PXRFS.
 Figure 1b: Amptek mini X-ray spectrometer. View Figure 1b
Figure 1b: Amptek mini X-ray spectrometer. View Figure 1b
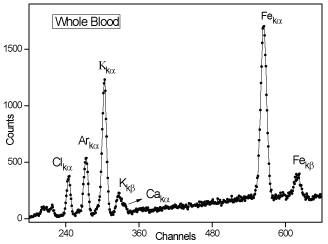 Figure 2: Whole blood PXRFS spectrum. View Figure 2
Figure 2: Whole blood PXRFS spectrum. View Figure 2
For Fe concentration determination, each sample was excited two times and the mean value was adopted. Standard (certified iron solution was prepared following the same blood sample procedure) was irradiated using the same excitation conditions. The quantitative analysis was performed using WINQXAS software (IAEA, version 1.3).
This alternative procedure (XRF) was capable, in a few minutes, to determine whole blood Fe concentrations using an efficient and non-destructive analysis. The Fe concentrations determined in whole blood samples are presented in Table 1. Mean Value (MV) and Standard Deviation (± 1 SD) expressed the results. The confidence interval of 95% (Range) for the control group and the ratios between male and female were also included. The Fe concentration results of the three dystrophic mice strains were compared with the control group (Figure 3) and the Student's t-test was applied for comparison results.
 Figure 3: Whole blood Fe concentrations comparison between control and dystrophic groups. View Figure 3
Figure 3: Whole blood Fe concentrations comparison between control and dystrophic groups. View Figure 3
According to Table 1 the levels are altered for SJL/J male (311 ± 29 mgL-1) and for A/J male (318 ± 18 mgL-1); they are significantly higher even when compared to the control group upper limit estimation (173 mgL-1) for a confidence interval of 95%, usually adopted as reference for clinical practices. The overload of iron can cause joint and abdominal pain, weakness, fatigue or organ damages, and that can eventually trigger another health problem in the PMD patients [17]. For Dmdmdx/J, the results for males (151 ± 22 mgL-1) agree with the control group (137 ± 18 mgL-1, p > 0.05). The only data that present a significant increase in Fe concentration (249 mgL-1) corresponds to the animal model (Dmdmdx/J male) that reached the highest weight (increase of 12% in grams), compared to other dystrophic mice when they reach adulthood (3-month-old). Considering that the DMD muscle generally shows an advanced stage of dystrophy, with muscle fibers abnormally bigger and surrounded by adipose tissues and replacing a healthier muscle, this animal may be severely affected [18]. These data can be seen in Figure 4; they were arranged in ascending order of concentration.
 Figure 4: Whole blood Fe concentrations comparison between Dmdmdx/J mice genders. View Figure 4
Figure 4: Whole blood Fe concentrations comparison between Dmdmdx/J mice genders. View Figure 4
Related to the gender, for the control group the data are statistically equal (p > 0.05) but, for Dmdmdx/J and SJL/J strains the comparison emphases alterations (p < 0.05). In Table 1 the ratios between male and female are presented (last column) showing that an increase in Fe concentration is accentuated in SJL/J males (~1.5). However, there is only one female (SJL/J) with a significant increase in Fe concentration (304 mgL-1) suggesting that this animal may be more likely to develop this disease. These data can be seen in Figure 5; they were arranged in ascending order of concentration.
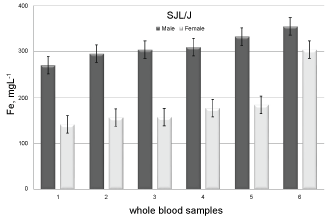 Figure 5: Whole blood Fe concentrations comparison between SJL/J mice genders. View Figure 5
Figure 5: Whole blood Fe concentrations comparison between SJL/J mice genders. View Figure 5
The Fe investigation in whole blood of A/J and SJL/J dystrophic male mice emphasized an increase suggesting that this ion must be constantly evaluated in the PMDs diseases, considering the high prevalence in males and consequent dysfunctions that this increase may cause to the organism such as organ damages, weakness, etc. These data also suggest that the increase of Fe in blood is more pronounced in the absence of dyspherlin protein. Related to the gender, the data obtained in the present study are not conclusive but they suggest that the female that has the Fe blood level increased (SJL/J) may be also developing the disease. The A/J female blood examination should be considered for future investigation to compare with other dystrophic female from other dystrophic mouse strains, confirming or not Fe behavior.
Nowadays, new therapies are being tested in animal models with PMDs. The proposed clinical methodology and the Fe limits in whole blood, established in the investigated animal models, can help health professionals and related areas to evaluate the clinical status of this animal model during tests in new treatments of progressive muscular dystrophies.
We would like to acknowledge for financial support of the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP).