Sinonasal Teratocarcinosarcomas (SNTCS) are rare tumors of the anterior skull base that are typically diagnosed in the fifth decade of life, with only 5 reported cases in children. Treatment typically involves surgical resection and radiotherapy. We present two cases of SNTCS, one involving a 70-year-old male and one involving a 16-year-old male, the latter having the first endoscopic-only resection of this skull base tumor. Given that pathologic diagnosis is difficult due to the mixed cell lines on specimens, high clinical suspicion in the male patient when pathologists struggle with obtaining a diagnosis, is key.
Sinonasal teratocarcinosarcoma, Endonasal approach, Anterior skull base, Pediatrics
Combining the histopathological features of carcinosarcoma and malignant teratomas, Sinonasal Teratocarcinosarcomas (SNTCS) are rare, aggressive tumors of the anterior skull base and sinonasal cavity. Patients are usually male and are diagnosed in their fifth decade of life [1]. Diagnosis is based on the presence of malignant epithelial elements and two or more malignant mesenchymal components, such as fibroblasts, cartilage, bone and smooth muscle [1]. However, given its infrequency and phenotypic diversity, diagnosis remains challenging and the presence of "fetal-appearing" clear-cell squamous epithelium and organoid structures are key [1].
Treatment is usually multimodal involving surgical resection and radiotherapy, but may be difficult secondary to aggressive tumor spread, intracranial extension, and high rate of recurrence. Reports of adjuvant chemotherapy are limited and its role it not well defined [1,2].
There have been less than 100 cases of SNTCS described in the literature [1,2], and of these, five have been reported in children. We present two cases of SNTCS, one involving a 70-year-old male and one involving a 16-year-old male, the latter having the first endoscopic-only resection of this skull base tumor. Both of these cases also demonstrate successful use of both adjuvant radiation and chemotherapy.
A 70-year-old male presented with a several month history of epistaxis, anosmia, and vision changes in the right eye after being seen at his local Emergency Department (ED) with massive epistaxis. Head and neck exam was normal except for a proptotic right eye with restricted motion. Nasal endoscopy was limited due to nasal packing but showed an obvious friable right-sided tumor of the entire superior nasal cavity involving the superior septum ethmoids. A Computed Tomography (CT) scan with contrast of the maxillofacial region demonstrated a large lytic right-sided sinonasal mass filling the right nasal cavity, ethmoid sinuses and frontal sinuses (Figure 1a). The tumor demonstrated erosion of the cribriform plate and right ethmoid roof into the paramedian anterior cranial fossa and right olfactory sulcus as well as extension into the nasopharynx, right pterygopalatine fossa and right sphenoid bone with involvement of the right foramen rotundum, vidian canal and anterior and inferolateral right sphenoid sinus. Subsequent CT neck demonstrated right-sided lymphadenopathy of level 2b suggestive of nodal metastatic disease. Subsequent MRI confirmed intracranial extension with leptomeningeal and parenchymal involvement of the right gyrus rectus with surrounding vasogenic edema (Figure 1b).
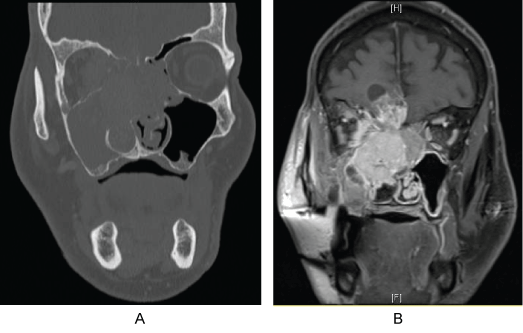 Figure 1: a) Coronal CT; b) MRI depicting intranasal mass extending into frontal lobe.
View Figure 1a
Figure 1: a) Coronal CT; b) MRI depicting intranasal mass extending into frontal lobe.
View Figure 1a
Given the concern for malignancy, the patient was taken to the operating room for nasal endoscopy with biopsies that returned as poorly differentiated adenocarcinoma with necrosis and sinonasal adenocarcinoma (colonic type) with neuroendocrine differentiation/component. Because of the uncertainty with the diagnosis, labs were sent out to Mayo Clinic and returned as SNTCS (Figure 2). A PET scan confirmed activity within the right neck giving him a staging of cT4bN2bMx.
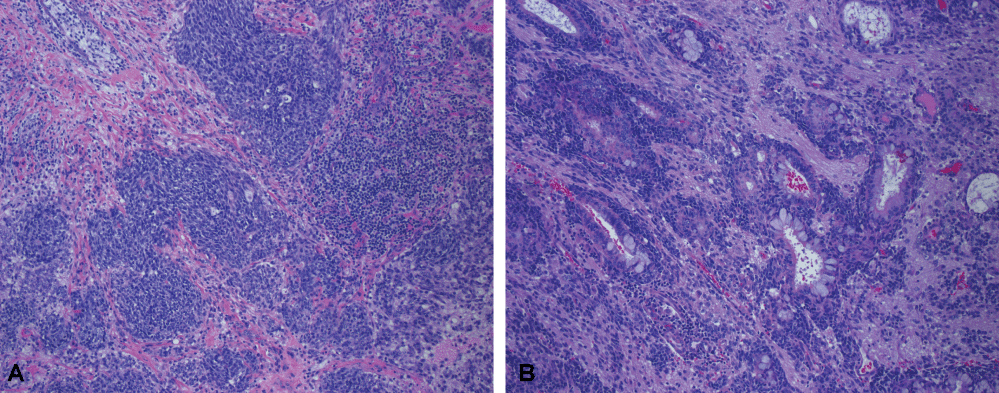 Figure 2: Pathology slides showing both a) Neuroendocrine; b) Glandular differentiation.
View Figure 2a
Figure 2: Pathology slides showing both a) Neuroendocrine; b) Glandular differentiation.
View Figure 2a
The patients case was discussed at the weekly tumor board in conjunction with radiation oncology, hematology/oncology, pathology and head and neck oncology. Surgery with adjuvant chemoradiation was recommended. Once deemed suitable for surgery, the patient underwent bifrontal craniotomy with combined transnasal endoscopic approach and resection of the tumor, right orbital exenteration, and radical neck dissection with sacrifice of cranial nerve XII secondary to tumor invasion. Frozen margins were negative for malignancy and the patient's skull base defect was reconstructed using a pericranial flap, Alloderm, and sealant. The patient was admitted to the ICU without unexpected neurologic deficits or a cerebrospinal fluid leak. He was later transferred to the floor and discharged after 6 days.
The patient underwent radiation 6 weeks after surgery without issue and was then started chemotherapy. Surveillance scans 4 months after surgery demonstrated a right level II lymph node concerning for malignancy, and he underwent revision selective neck dissection. The patient was disease free as of 14 months after surgery, 13 months after radiation, and 2 months after chemotherapy but passed away due to unrelated reasons.
A 16-year-old Hispanic male otherwise healthy was referred to the otolaryngology clinic from the allergy clinic for evaluation of nasal obstruction and congestion refractory to maximal medical therapy. Head and neck exam was normal except for hyponasal voice. Nasal endoscopy revealed a large, well mucosalized, a pulsatile lesion that appeared to be coming from the face of the sphenoid bilaterally causing complete nasal obstruction. Maxillofacial CT and MRI with contrast demonstrated a large, partially cystic, expansile sinonasal mass with peripheral heterogenous calcification centered in the posterior sinonasal region. The mass was associated with bony remodeling of the overlying basisphenoid bone, and posterior ethmoid sinus, nasal septum ad nasal cavity bony margins. The mass measured 4.6 × 3.9 × 5.8 cm and protruded into the right sphenoid sinus and was indeterminate if the mass invades the posterior left ethmoid and sphenoid sinuses (Figure 3).
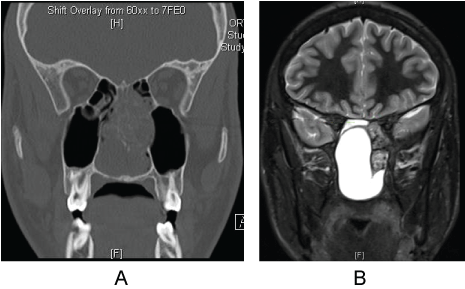 Figure 3: a) Coronal CT; b) MRI demonstrating large sinonasal mass with cystic components and no skull base invasion.
View Figure 3a
Figure 3: a) Coronal CT; b) MRI demonstrating large sinonasal mass with cystic components and no skull base invasion.
View Figure 3a
The patient was taken to the OR for endoscopic biopsy, which was inconclusive on frozen and permanent section. Outside pathology consult remained inconclusive and returned as low-grade epithelial lesion. The decision was made to take the patient for tumor debulking to obtain greater tissue for diagnosis, but to do it with minimal morbidity given the ambiguous diagnosis. Bilateral sphenoid, maxillary, and ethmoid sinuses were opened and solid tumor was largely removed via transnasal endoscopic approach. Residual tumor was left within the sphenoid and the left base of the pterygoids. Permanent pathology, with the assistance of Beth Israel Hospital, returned as SNTCS. After receiving a conclusive diagnosis and discussion at the weekly tumor board, the patient was taken to the OR for complete endoscopic resection of disease with negative margins. Post-operatively, the patient did not develop a CSF leak or any neurologic issues.
He was treated with four cycles of chemotherapy with doxorubicin, ifosfamid and mesna and 6 weeks of radiation therapy during the second chemotherapy cycle. He is currently 38 years out from surgery, 36 months out from radiation therapy, and 32 months out from chemotherapy. He remains disease free on exam and surveillance imaging, and has appropriate neurocognitive functioning.
To date, less than 100 cases have been published in the literature. Typically, these tumors present in the 4th to 5th decade of life and have a 7:1 male predilection [1]. Only 60% of patients survive beyond 3 years [3]. The most common symptoms at presentation are nasal obstruction, epistaxis, and headache [1]. Treatment usually entails surgical resection with radiotherapy. In a retrospective systematic review, patients receiving chemotherapy and radiation faired better, but the study is limited by poor power and limited follow up [1]. Both of our patients underwent surgical resection with chemoradiation. Metastatic disease is common to the neck, as was seen in our first patient. A single institution review demonstrated metastatic nodal disease in 23% of patients [4]. Metastatic disease to the lung, brain, and dura has also been reported [5-7].
Our second patient was sixteen-years-old at the time of his diagnosis making him the fifth pediatric patient diagnosed with SNTCS [8]. All of these patients were male, and reportedly treated with surgery with a combination of radiation and/or chemotherapy. Given the aggressive nature of these tumors, and the difficulty in obtaining truly negative margins, adjuvant chemoradiation seems most appropriate despite the long-term effects of these therapies.
One of the difficulties we had in treating these patients was obtaining a suitable pathologic diagnosis despite obtaining adequate specimens. This is understandable given both benign and malignant epithelial, mesenchymal, and neuroepithelial components. SNTCS is thought to have a 50% initial identification rate [1]. A previous case series demonstrated adenocarcinoma as the primary epithelial component in all tumors in their study [5]. This was similar to our study, as our first case was initially diagnosed as adenocarcinoma, and our second case also had components of adenocarcinoma on final specimen. Stains were of little use, as they are bound to the corresponding cell line within the tumor. The distinguishing factor of these tumors is that they lack germ cell elements [9].
SNTCS is a rare, invasive sinonasal tumor with a poor prognosis. Pathologic diagnosis is difficult given mixed cell lines on specimens. High clinical suspicion in the male patient, particularly when pathologists struggle with obtaining a diagnosis, is key. Metastatic work up is warranted, and aggressive resection with adjuvant chemoradiation is necessary.