Lymphoma of the nasal cavity represent only 0.2-2% of head and neck lymphomas. Treatment is medical with an overall low 5-years survival between 40% and 59%. There are high rates of relapse as well as radiotherapy and chemotherapy resistance. We present a 44-year-old male, who was admitted to our department due to chronic sinonasal infection, complicated by a large septal perforation and nasal tissue necrosis, which developed over several months following an elective septoplasty surgery performed elsewhere. Nasal biopsy revealed granulation tissue and ANCA titers were negative. A repeated biopsy revealed nasal type NK/T Cell Lymphoma. Sinonasal lymphoma is very rare and challenging to diagnose. Presenting signs may mimic benign inflammatory or infectious conditions, such as rhinosinusitis or granulomatous diseases, which often lead to late diagnosis and an advanced disease stage. Surgeons have a responsibility to be aware of any red flags or early warning signs to insure that a proper and thorough medical investigation had been performed prior to any elective surgery. A high index of suspicion is required and fresh tissue biopsies should be taken to rule out malignancy in any case if symptoms persist despite adequate conservative treatment.
Nasal lymphoma, Septal perforation
More than one-third of extranodal lymphomas originate in the head and neck region. The nasal cavity and paranasal sinuses are rarely involved, representing only 0.2-2% of head and neck lymphomas [1]. When the sinonasal system is involved, the disease tends to be locally destructive with invasion into surrounding vasculature and tissues and subsequent extensive necrosis. Treatment includes chemoradiation with an overall low 5-years survival between 40% and 59% [1]. There are high rates of relapse as well as radiotherapy and chemotherapy resistance.
We present a 44-year-old-male suffering from chronic sinonasal infection who was admitted to our department for further investigation and treatment. The patient had undergone an elective septoplasty operation elsewhere. He stated that the septoplasty operation was postponed several times due to fever, large crusts and purulent nasal discharge, and that he was treated preoperatively with antibiotics (PO Ciprofloxacin) and nasal saline. Following the surgery a large septal perforation with crusting and nasal tissue necrosis had developed over several weeks. The patient was treated repeatedly with antibiotics (IV Amoxicillin/clavulanic acid and IV Ciprofloxacin) and nasal ointments (Fusidic acid, Chloramphenicol and Mupirocin). Nasal cultures grew Pseudomonas Aeruginosa sensitive to Ceftazidime. Due to insufficient response, he was admitted again in the same hospital and was treated with IV Ceftazidime, nasal douche and topical antibiotic ointments. A first nasal biopsy was taken and revealed granulation tissue only, without any signs of malignancy. Despite prolonged medical treatment, there was no improvement in his condition. He suffered from severe pain, headaches and developed high body temperature. Therefore, he was transferred to our department.
Physical examination upon admission revealed extensive nasal cellulitis. Endoscopic evaluation exposed thickened nasal mucosa and a large perforation of the nasal septum with substantial crusting (Figure 1). Laboratory CBC tests showed mild anemia with hemoglobin of 12.7 GR/DL with normal WBC (70% Neutrophils) and platelets count. Chemistry tests showed mildly elevated CRP of 27 MG/DL and LDH of 576 U/L.
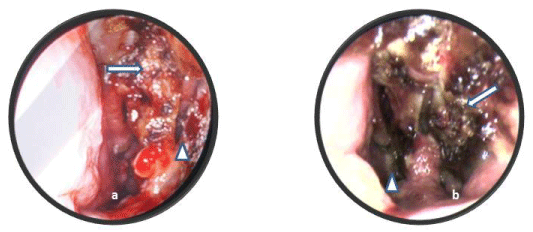 Figure 1: Right (a) and Left (b) Nasal endoscopy showing thickened nasal mucosa with crusting (arrow) and a large septal perforation (arrow head). View Figure 1
Figure 1: Right (a) and Left (b) Nasal endoscopy showing thickened nasal mucosa with crusting (arrow) and a large septal perforation (arrow head). View Figure 1
At this point, the working diagnosis was a severe case of post-operative atrophic rhinitis [2], keeping in mind other differential diagnostic possibilities such as granulomatous diseases or malignancy of the sinonasal tract.
In consultation with infectious diseases unit, the patient was treated with high dose IV Ciprofloxacin and topical antibiotic ointment (Gentamicin-Betamethasone). In office local debridement of crusting and necrotic tissue was periodically performed. CT scan of the sinuses was performed and showed a large septal perforation and diffuse pansinusitis with mucosal thickening (Figure 2). A second nasal biopsy was taken from the nasal septum and lateral nasal wall. Nasal biopsy revealed granulation tissue and ANCA titers were negative. Local debridement and treatment was continued with only minor relief. Therefore, a third very wide excisional biopsy was obtained and revealed mucosal tract covered by ulcerated transitional and pseudostratified squamous epithelium infiltrated by T-cell lymphocytes positive for CD3 and CD56 on immunohistochemistry (IHC) staining (Figure 3) highly suggestive for nasal type NK/T Cell Lymphoma [3]. Serology for HIV and EBV were negative and PCR for EBV was slightly positive. Pathology slides were examined for in situ hybridization for EBV encoded RNA-EBER that was positive, providing us with the final confirmation on the pathology result [4].
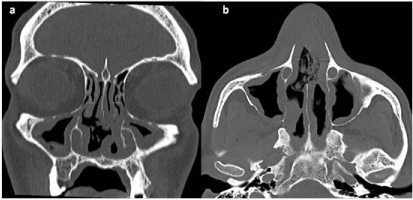 Figure 2: Coronal (a) and Axial (b) CT scan of the sinuses demonstrating a large septal perforation and diffuse pansinusitis mucosal thickening. View Figure 2
Figure 2: Coronal (a) and Axial (b) CT scan of the sinuses demonstrating a large septal perforation and diffuse pansinusitis mucosal thickening. View Figure 2
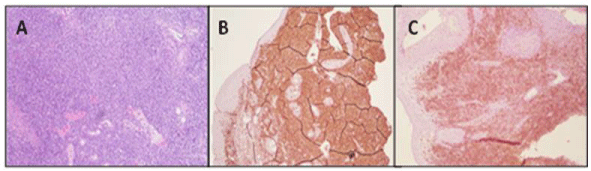 Figure 3: IHC slides: A) Hematoxylin & Eosin staining demonstrate polymorphic lymphoid infiltrate made up of mature, immature, and atypical lymphocytes, plasma cells, histiocytes, eosinophils and macrophages; B) Positive staining for cytoplasmic CD3 epsilon (Unlike T-cell large granular lymphocytes witch present only surface CD3); C) Positive staining for CD56. View Figure 3
Figure 3: IHC slides: A) Hematoxylin & Eosin staining demonstrate polymorphic lymphoid infiltrate made up of mature, immature, and atypical lymphocytes, plasma cells, histiocytes, eosinophils and macrophages; B) Positive staining for cytoplasmic CD3 epsilon (Unlike T-cell large granular lymphocytes witch present only surface CD3); C) Positive staining for CD56. View Figure 3
PET-CT was done and excluded any local or distant metastasis. NK/T cell lymphoma prognostic index was high-intermediate due to high LDH values with B symptoms (Table 1). According to Ann Arbor staging system (Table 2) the patient was considered to be a stage IIE (invading the nasal septum and lateral nasal wall) and was scheduled for sequential chemoradiation therapy.
Table 1: NK/T Cell lymphoma prognostic index. View Table 1
Table 2: ANN-ARBOR staging system. View Table 2
Lymphomas of the paranasal sinuses and nasal cavity are rare. In Asian and South American populations, nasal lym¬phomas are more common and are predominantly of the natural killer NK/T-cell subtype [5], previously referred to as lethal midline granuloma or angiocentric lymphoma. NK/T-cell subtype affects young to middle-aged adults, has a male predilection in most series, and commonly manifests with a nasal mass or obstruction, epistaxis, palatal destruction, erythema and swelling of the face [6]. Infiltration of the tumor cells is frequently perivascular, and vascular occlusion with massive necrosis of the tissue is one of the presenting features. Because of tissue necrosis, it is some¬times difficult to make a diagnosis, and multiple biopsies are often needed. Virtually all cases of NK/T-cell lymphoma are associated with EBV, and in situ hybridization with EBER-1 or EBER-2 may aid in the histologic diagnosis of nasal lympho¬mas. Circulating plasma EBV DNA level can be used to monitor disease status and predict prognosis [4]. IHC stains are positive for CD2, CD3, and CD56. CD56 staining should be done in any patient with a sus¬pected diagnosis of nasal lymphoma, because CD56- and EBV-positive lymphocytes are rarely observed in normal or inflammatory nasal mucosa. Treatment is based on the Ann Arbor staging system, the performance status, and whether or not the patient is fit for chemotherapy [7,8]. It involves radiation alone for patients unfit for chemotherapy, or chemoradiation which can be given in several ways and regimens. More than 80% of patients come to medical attention with stage I to IIE disease. Despite a high response rate (60% to 80%), the relapse rate is also high (50%) [9]. The International Prognostic Index (IPI) for NHL has predictive power in NK/T-cell lymphoma. However, models that include B symptoms, stage, LDH, regional node involvement, local tumor invasiveness, and the presence of extranasal disease improve prognostic discrimination. In most series, 40% to 50% of patients are alive and disease free 5 years after diagnosis. In the case of our patient, his surgery was postponed several times due to high fever, large crusts and purulent nasal discharge. Despite this, his surgeon did not question the diagnosis and did not perform any further examination prior to the surgery. His symptoms worsened significantly following his nasal surgery. Therefore the initial suspicion was that it was a surgical complication, rather than a primary disease, which led to a delay in the diagnosis. Nevertheless, his disease was still local and he was treated with the SMILE chemotherapy regimen (Dexamethasone, Methotrexate, Ifosfamide, L-asparaginase, and Etoposide radiation) and radiotherapy. At one year post-treatment he is doing well and free of disease.
Sinonasal lymphoma is very rare and challenging to diagnose. Presenting signs may mimic benign inflammatory or infectious conditions, such as rhinosinusitis or granulomatous diseases, which often lead to late diagnosis and an advanced disease stage. Surgeons have a responsibility to be aware of any red flags or early warning signs to insure that a proper and thorough medical investigation had been performed prior to any elective surgery. A high index of suspicion is required and fresh tissue biopsies should be taken to rule out malignancy in any case if symptoms persist despite adequate conservative treatment.
The authors have no funding, financial relationships, or conflicts of interest to disclose.