Gliosarcoma is a very uncommon primary brain malignancy and is considered a subtype of glioblastoma. It represents approximately 2% of cases classified as glioblastoma. Treatment of gliosarcoma follows the paradigm for glioblastoma with recommendations for a maximal allowable resection followed by post-operative radiation therapy to the operative bed with concurrent temozolomide. Although still being a rare occurrence, gliosarcomas have a relatively higher propensity for transdural growth either through natural disruptions in the dura or through the craniotomy site. We report on a case of a gliosarcoma that grew through a craniotomy site and initially was mistaken for scar formation. Upon realization of the transdural spread, a modification was needed to the post-operative radiation portal. Furthermore, the disease exhibited discontinuous spread along the ipsilateral cerebral hemisphere and ultimately within the contralateral cerebral hemisphere with dural contact.
A 65-year-old female presented with severe headaches and an MRI of the brain revealed a 5.5 cm heterogeneous mass with dural contact in the left frontal lobe with mass effect and a midline shift. Imaging of the chest, abdomen and pelvis failed to reveal an extracranial abnormality (Figure 1).
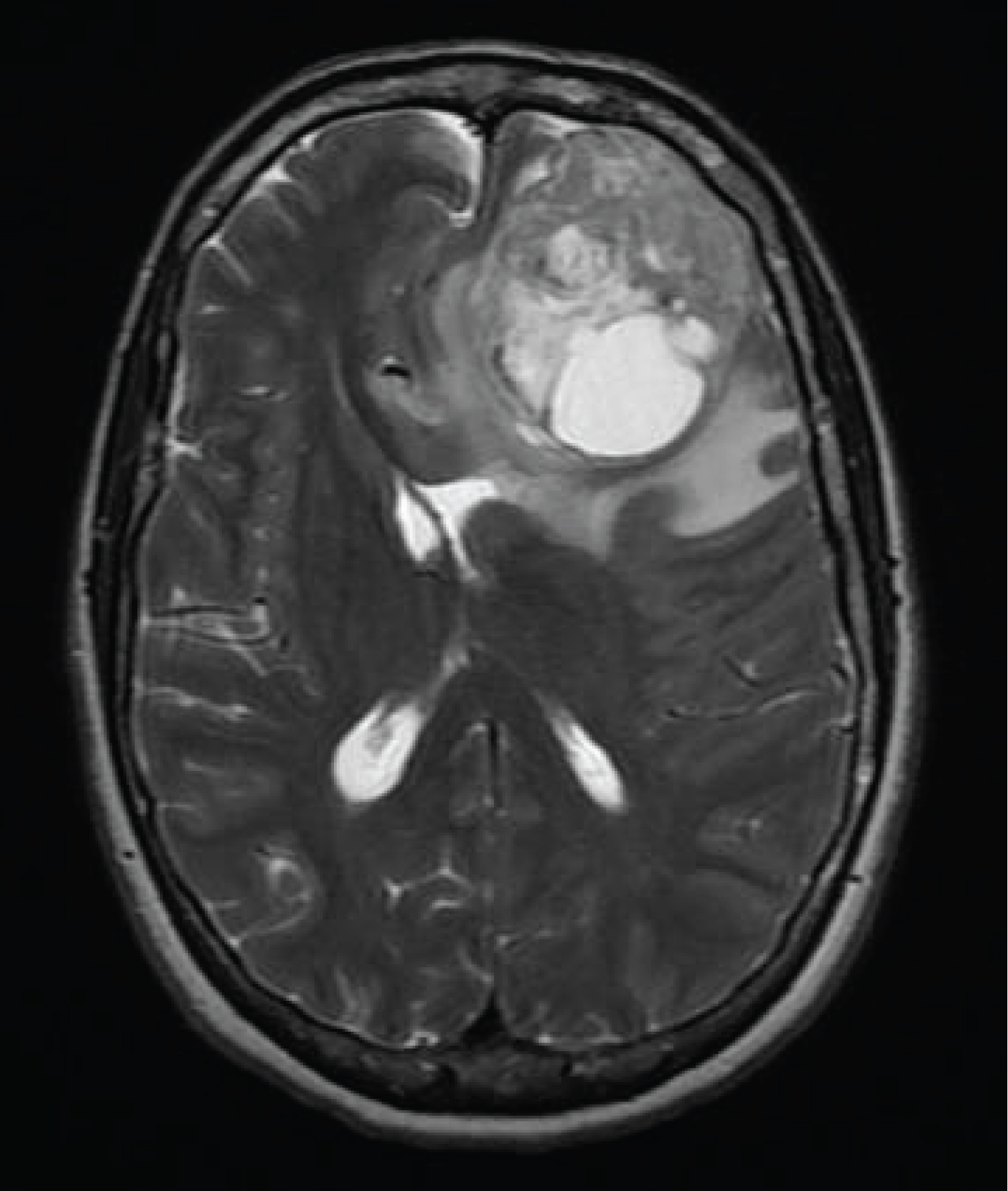 Figure 1: The MRI of the brain at initial presentation revealed a 5 cm heterogeneous mass located in the left frontal lobe with associated vasogenic edema and a 9 mm midline shift to the right. A gross total resection revealed gliosarcoma.
View Figure 1
Figure 1: The MRI of the brain at initial presentation revealed a 5 cm heterogeneous mass located in the left frontal lobe with associated vasogenic edema and a 9 mm midline shift to the right. A gross total resection revealed gliosarcoma.
View Figure 1
A left frontal craniotomy was performed with a gross total removal of the mass. A histopathologic examination of the specimen revealed both gliomatous and sarcomatoid neoplastic elements rendering a diagnosis of gliosarcoma (Figure 2). No IDH1 or IDH2 mutation was identified. Post-operatively, she received a course of radiation therapy delivering a total of 60Gy to the operative site coupled with concurrent temozolomide. She declined maintenance temozolomide.
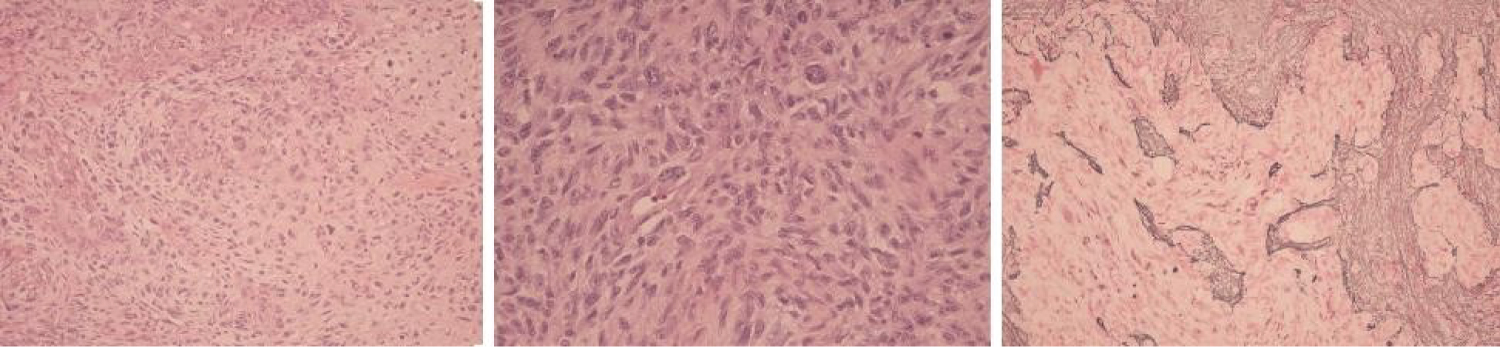 Figure 2: (Left Panel) The removed specimen revealed a WHO grade 4 gliosarcoma. Note the malignant heterologous sarcomatoid elements that in this case are of chrondoid differentiation; (Middle Panel): Note the pleomorphic spindle cell elements with nuclear hyperchromasia and pleomorphism in addition to the neoplastic glial cells with high nuclear grade. Necrosis and endovascular proliferation were also apparent. (Right Panel) Note that the sarcomatous elements stain take up a reticulin stain with and the lack of staining of the gliomatous elements.
View Figure 2
Figure 2: (Left Panel) The removed specimen revealed a WHO grade 4 gliosarcoma. Note the malignant heterologous sarcomatoid elements that in this case are of chrondoid differentiation; (Middle Panel): Note the pleomorphic spindle cell elements with nuclear hyperchromasia and pleomorphism in addition to the neoplastic glial cells with high nuclear grade. Necrosis and endovascular proliferation were also apparent. (Right Panel) Note that the sarcomatous elements stain take up a reticulin stain with and the lack of staining of the gliomatous elements.
View Figure 2
Approximately six months after completion of adjuvant therapy, she developed expressive aphasia and a MRI of the brain revealed a new left sided temporal lobe mass outside the previously treated area (Figure 3).
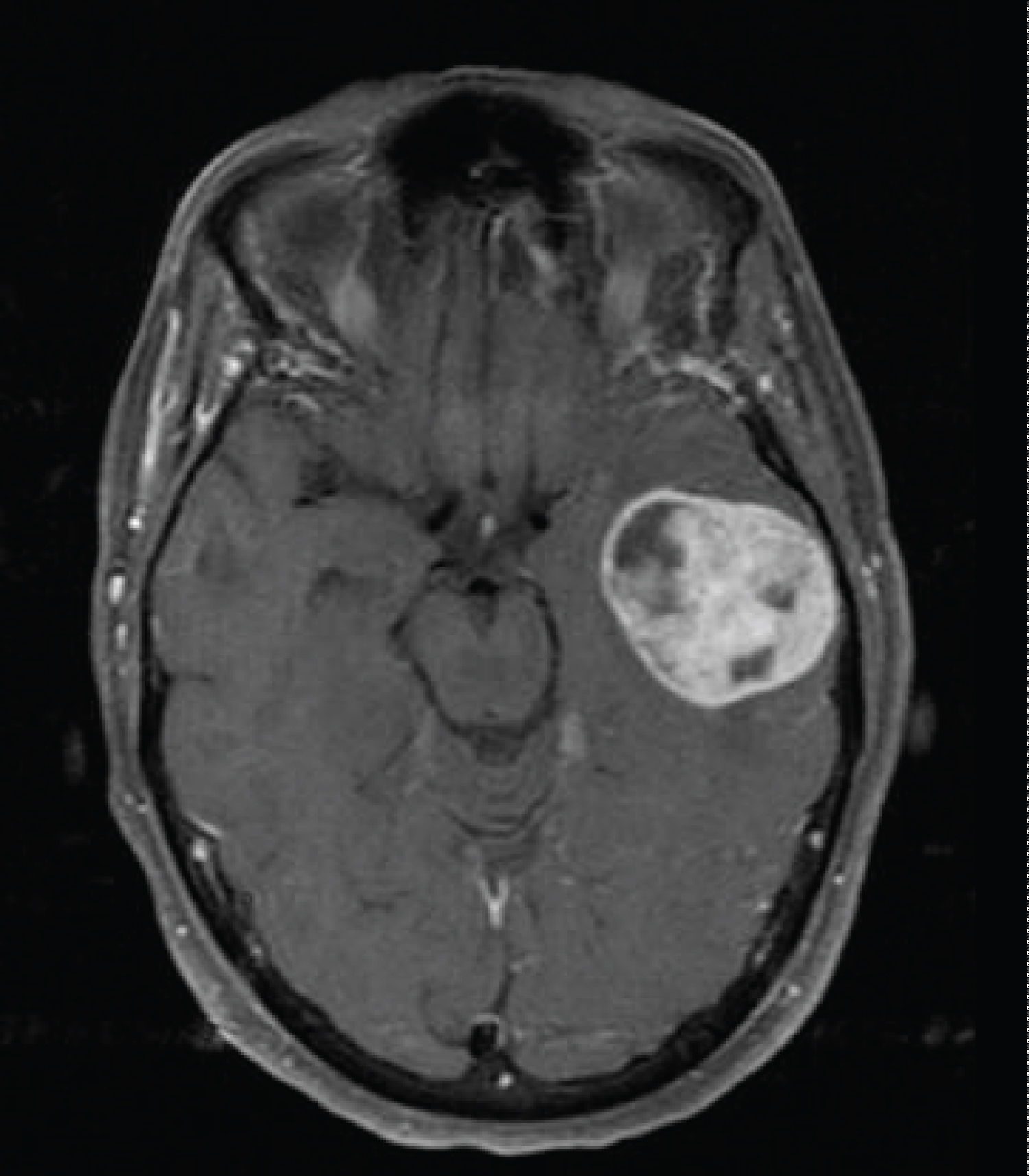 Figure 3: Eight months after her initial resection, an MRI was performed to evaluate new expressive aphasia. Gadolinium enhanced T1-weighted images showed development of a 3 cm new left temporal lobe mass.
View Figure 3
Figure 3: Eight months after her initial resection, an MRI was performed to evaluate new expressive aphasia. Gadolinium enhanced T1-weighted images showed development of a 3 cm new left temporal lobe mass.
View Figure 3
A second resection was performed and a total gross removal was achieved. As before, a histopathologic evaluation of the specimen revealed a gliosarcoma. She embarked on a second course of radiation therapy coupled with temozolomide. During her course of therapy, she was noted to have an enlarging subcutaneous nodule over the left temporal craniotomy site that initially was thought to represent developing scar tissue from her surgery. However, its persistent growth led to a third MRI that revealed a new mass in the left frontal parietal region with transdural extension of the mass through the second craniotomy site (Figure 4).
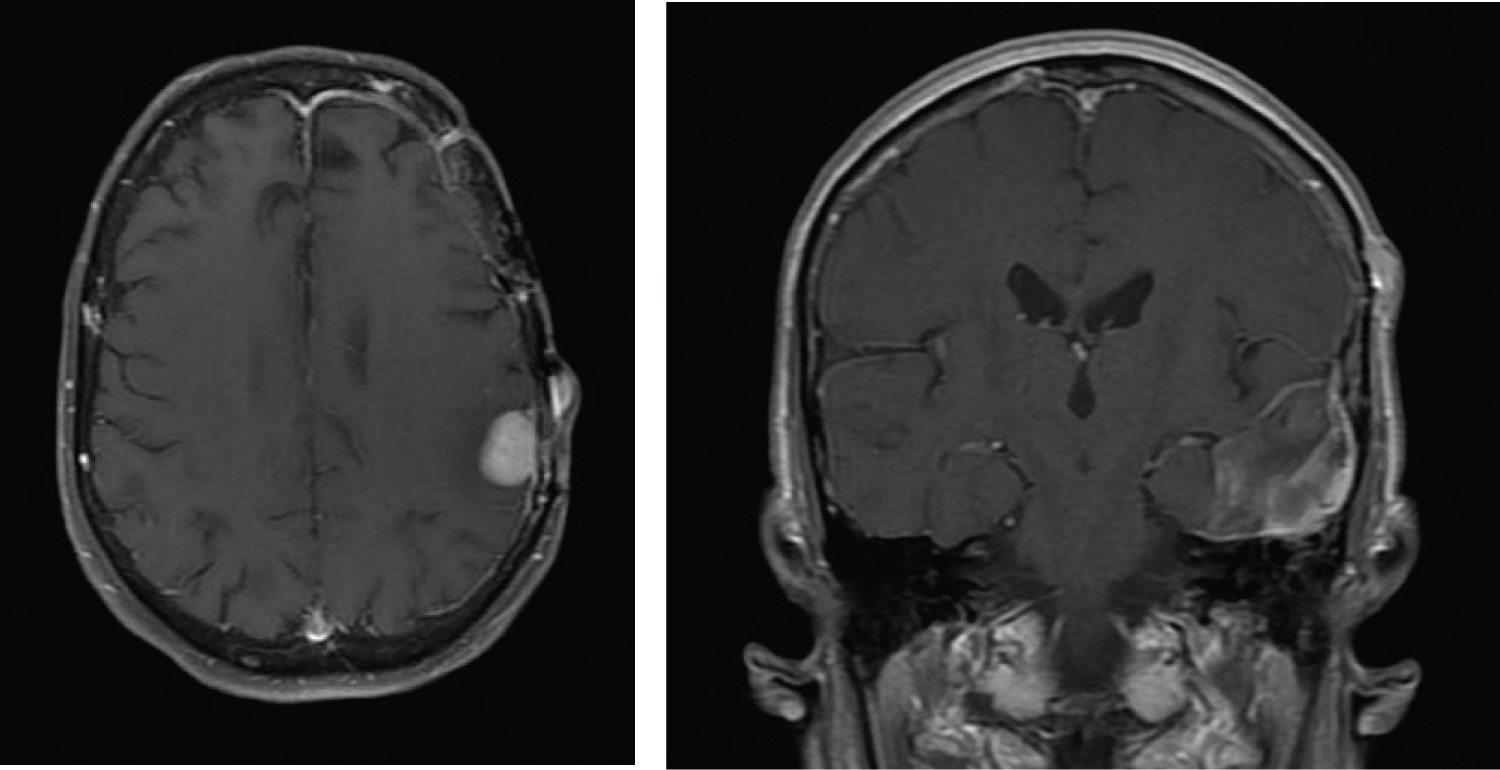 Figure 4: After resection of the second mass (Figure 3), an enlarging palpable mass was noted near the second craniotomy site. Transverse MRI images revealed a new 2.3 cm enhancing lesion contacting the dura in the left frontal parietal region with an associated 14 mm scalp nodule that represented transdural spread of disease through the second craniotomy site. The coronal images show the new enhancing lesion and adjacent scalp nodule remote from the previous intraparenchymal operative bed in the left temporal lobe.
View Figure 4
Figure 4: After resection of the second mass (Figure 3), an enlarging palpable mass was noted near the second craniotomy site. Transverse MRI images revealed a new 2.3 cm enhancing lesion contacting the dura in the left frontal parietal region with an associated 14 mm scalp nodule that represented transdural spread of disease through the second craniotomy site. The coronal images show the new enhancing lesion and adjacent scalp nodule remote from the previous intraparenchymal operative bed in the left temporal lobe.
View Figure 4
This led to modification of the radiation therapy portal in order to incorporate the third lesion as well as the associated scalp lesion. However, two months after completion of her second course of concurrent radiotherapy and temozolomide, an MRI of the brain revealed a fourth mass with dural contact along the left occipital lobe (Figure 5). She chose for further limited field radiation therapy with concurrent Avastin.
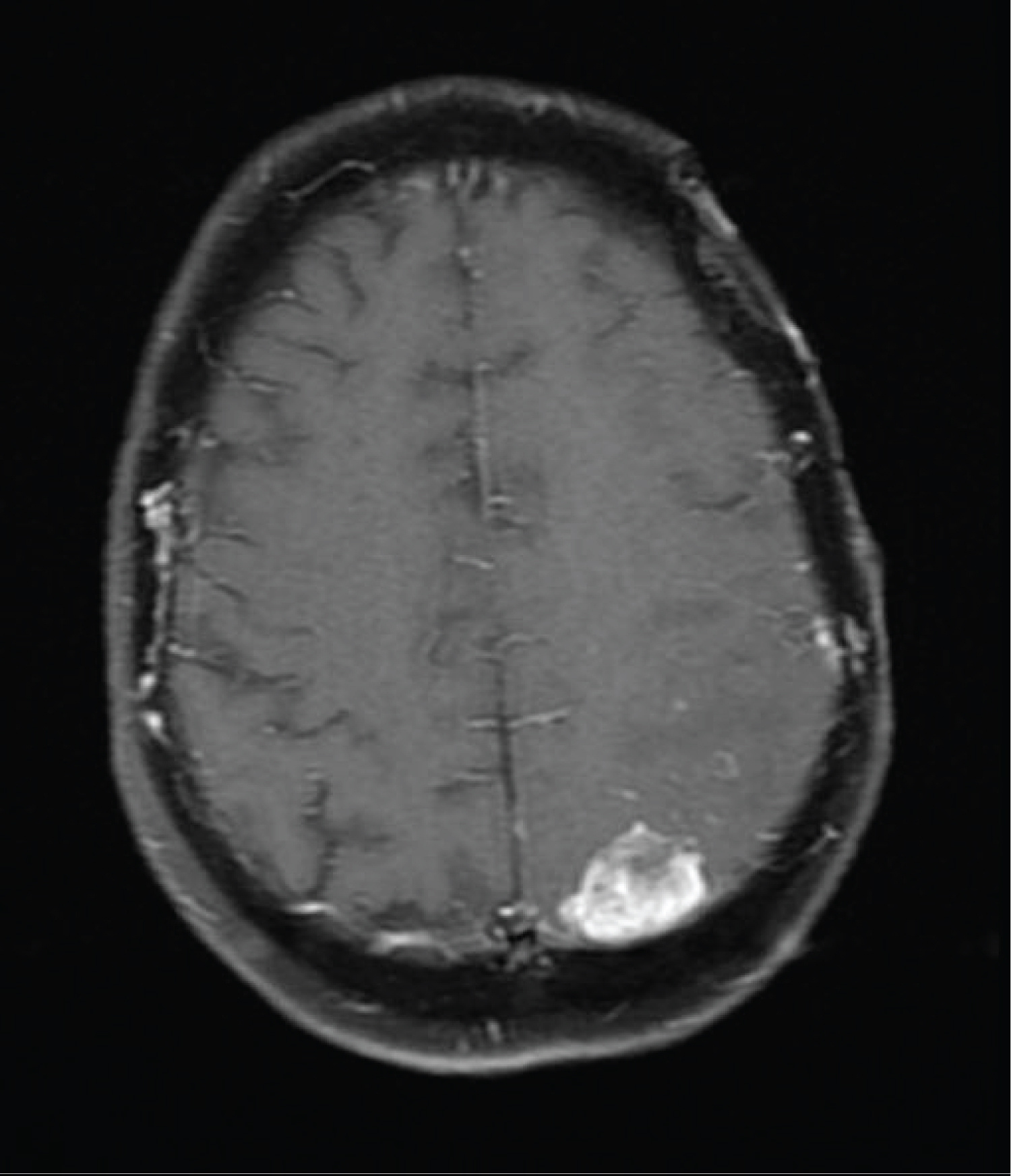 Figure 5: Axial T1 Fat-Suppression images with gadolinium enhancement show a new enhancing mass in the left posterior parietal region with dural contact.
View Figure 5
Figure 5: Axial T1 Fat-Suppression images with gadolinium enhancement show a new enhancing mass in the left posterior parietal region with dural contact.
View Figure 5
Two months after completion of her course of therapy, an MRI of the brain revealed progression of disease in the left temporal lobe and a new focus of signal abnormality in the medial right frontal lobe (Figure 6). Her performance status had markedly deteriorated and no further therapy was administered.
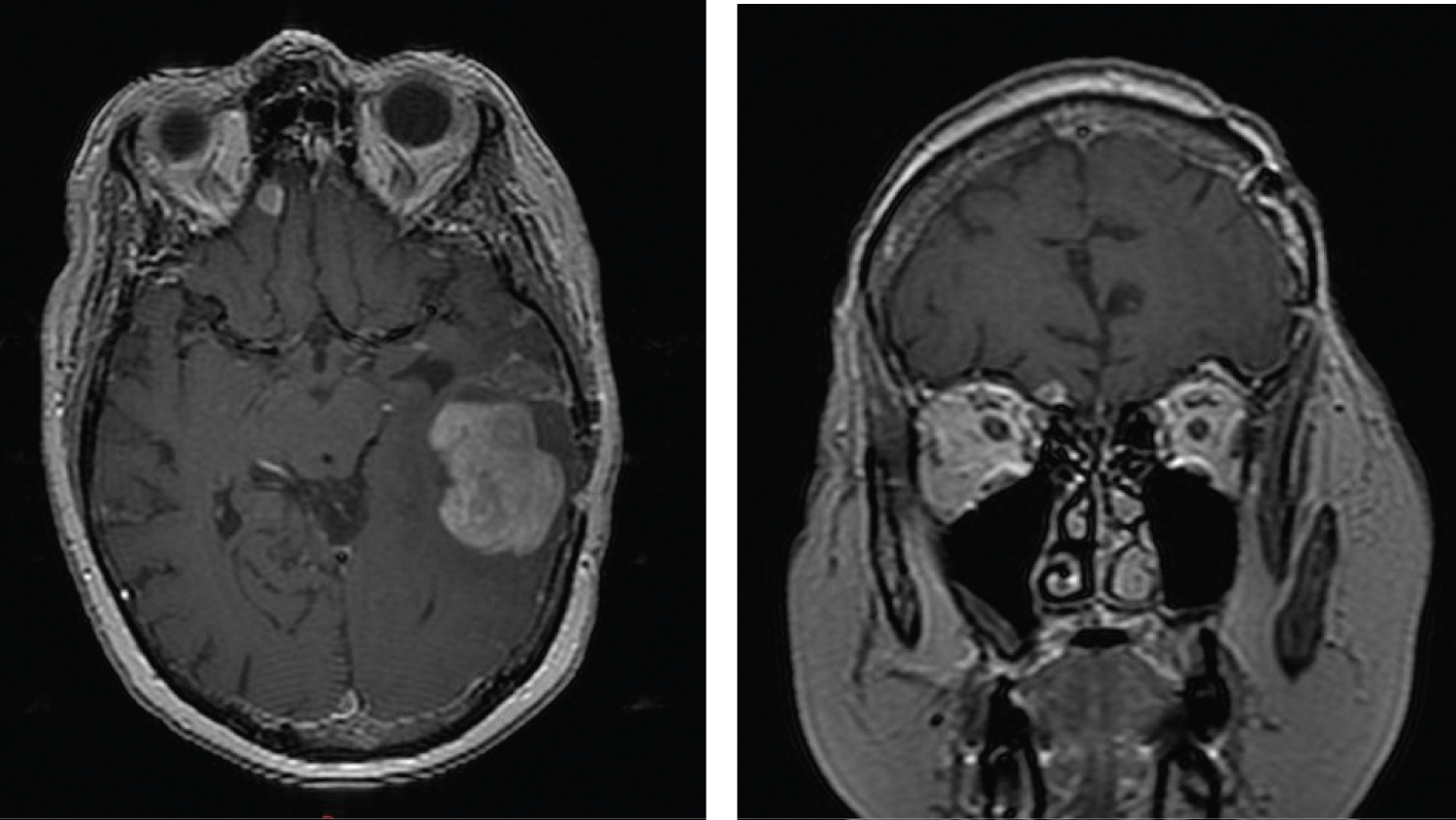 Figure 6: Axial and coronal contrast enhanced T1 images showing growth of the gliosarcoma in the left temporal lobe and development of a new mass in the right frontal lobe with dural contact.
View Figure 6
Figure 6: Axial and coronal contrast enhanced T1 images showing growth of the gliosarcoma in the left temporal lobe and development of a new mass in the right frontal lobe with dural contact.
View Figure 6
One month later, she developed right frontal headaches and a final MRI of the brain revealed progression of the metastatic deposit in the right frontal lobe (Figure 7).
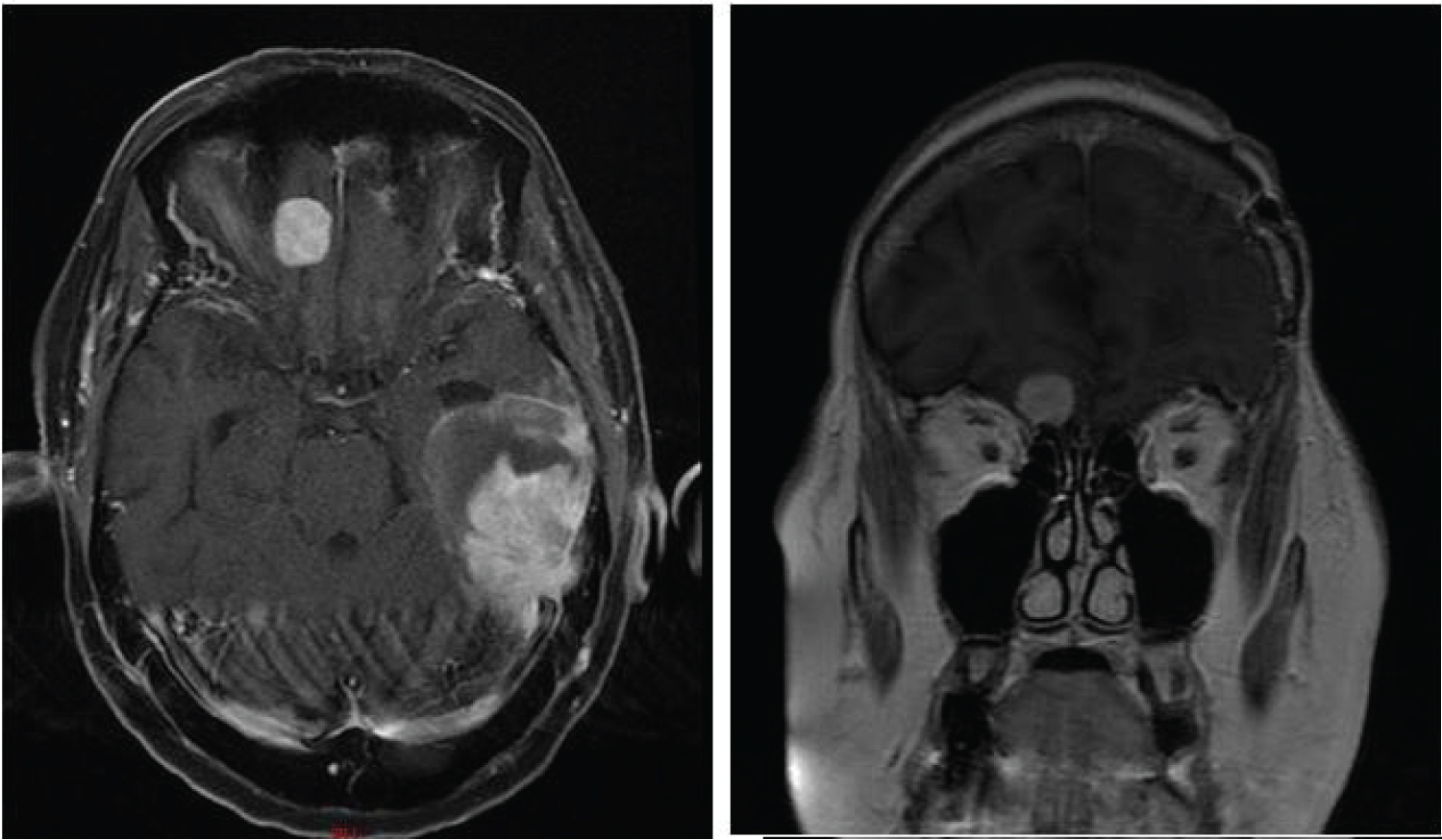 Figure 7: Axial and coronal MRI images showing enlargement of the left temporal mass and right frontal lobe mass.
View Figure 7
Figure 7: Axial and coronal MRI images showing enlargement of the left temporal mass and right frontal lobe mass.
View Figure 7
She succumbed to her disease six weeks after her last scan.
The treatment for gliosarcoma is identical to that of the more common glioblastoma with first- line radiation therapy coupled with Temodar [1,2]. No standard treatment has been described for recurrences with further resection, radiation, and chemotherapy being at the discretion of the treating physician.
Leptomeningeal cells and the associated cellular components of the dura mater may constitute a barrier against glioma cell invasion and could account for the very low incidence of extracranial spread of gliomas [3]. However, the incidence of extracranial metastasis with gliosarcoma has been reported in up to 11% of cases [4]. This is higher than the accepted incidence of < 2% reported in glioblastomas [5,6]. The higher incidence may be due to the sarcomatous elements having a greater propensity for dissemination. Cases of distal extracranial spread from have been reported with distal metastasis most commonly identified in the lungs, liver, and cervical nodes although metastasis have been reported in the bone, kidneys, spleen, pancreas, stomach, and muscle [7].
Our patient developed transdural spread into the overlying galea aponeurotica through the second craniotomy site. Transdural spread with spontaneous extension through natural disruptions of the meninges has been reported but most cases are found in patients that have undergone neurosurgical intervention and this pattern of spread has been reported for all subtypes of glioma. However, only a few case reports are found in the literature specifically addressing the issue of extension in the surrounding scalp for gliosarcomas. Lakhdar and colleagues reported a case of man who developed scalp metastases two months after resection of a left frontal lobe gliosarcoma [8]. Frandsen, et al. reported on two cases with subcutaneous extension after resection of a temporal gliosarcoma [1]. A case of transdural tumor growth through a surgical defect created in the calvarium of a patient undergoing brachytherapy for a glioblastoma with a sarcomatous component has also been reported [9]. In our patient, the growth of disease was initially mistaken as fibrous scar formation. However, upon review of a subsequent MRI of the brain, a dural base metastasis was identified underneath yielding the diagnosis. This required a modification of the radiation treatment fields to encompass this area.
Late in her course of disease, our patient developed frontal headache and occasional blood-tinged rhinorrhea. Due to her marked deterioration, an investigation into the etiology was deemed to be futile and not performed. However, as seen in Figure 7, her last MRI showed enlargement in a metastatic deposit adjacent to the cribriform plate. It is reasonable to assume that there may have been continued growth and extension of disease in the right frontal lobe through the spontaneous defect of the dural in the region of the cribriform plate leading to her symptoms.
In our patient, disease was also noted to have spread in a sequential fashion from the left cerebral hemisphere to the right cerebral hemisphere. Radiographically, the MRI signal abnormalities had dural contact. The authors feel that it is reasonable to assume that the disease spread either through direct extension along the dura or through the cerebrospinal fluid leading to discontinuous metastatic deposits.
Although dural and leptomeningeal disease spread have been reported for gliosarcoma, this mode of dissemination is rare [10-16]. The particular mode of intracranial spread in our patient was most similar to the case reported by Rech, et al. of a man that developed a gliosarcoma due to a delayed transformation from a resected grade II glioma and the case and by Ramos, et al. in a patient with a resected left temporal lobe gliosarcoma [17,18].
Gliosarcomas are aggressive primary brain malignancies that can exhibit transdural spread through either iatrogenic or natural disruptions in the dura mater. This mode of progression through a surgical defect in the cranium and may be mistaken for scar formation in the craniotomy site. The possibility of dural spread of disease needs to be kept in mind when evaluating patients during follow-up visits as patients may present with signs and symptoms distal to the original location of the malignancy.
The authors report no financial assist or conflict of interest.